Decentralized Trial Wearable Market Report 2026

Decentralized Trial Wearable Market Report 2026
Global Outlook – By Product Type (Smartwatches, Fitness Trackers, Biosensors, Smart Clothing, Other Product Types), By Distribution Channel (Online, Offline), By Application (Remote Patient Monitoring, Clinical Trials, Disease Management, Other Applications), By End-User (Pharmaceutical Companies, Contract Research Organizations, Healthcare Providers, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Decentralized Trial Wearable Market Overview
• Decentralized Trial Wearable market size has reached to $2.43 billion in 2025 • Expected to grow to $6.18 billion in 2030 at a compound annual growth rate (CAGR) of 20.5% • Growth Driver: Increasing Demand For Real-Time Patient Monitoring Driving The Market Growth Due To Rising Chronic Disease Burden And Need For Continuous Health Surveillance • Market Trend: Ultra-Compact Wearable Devices Revolutionize Remote Monitoring In Clinical Trials • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Decentralized Trial Wearable Market?
A decentralized trial wearable is a digital device designed to capture and transmit real-time health, physiological, or activity data from participants in decentralized or hybrid clinical trials. It facilitates remote monitoring and data collection without requiring in-person site visits. The use of such wearables improves data reliability, participant convenience, and overall trial efficiency by enabling continuous data flow, reducing logistical burdens, and enhancing engagement throughout the study duration. The main product types of decentralized trial wearables are smartwatches, fitness trackers, biosensors, smart clothing, and other product types. These wearables are distributed through online and offline channels. They are used for various applications, including remote patient monitoring, clinical trials, disease management, and other applications, and are used by various end-users, including pharmaceutical companies, contract research organizations (CROs), healthcare providers, and other end-users.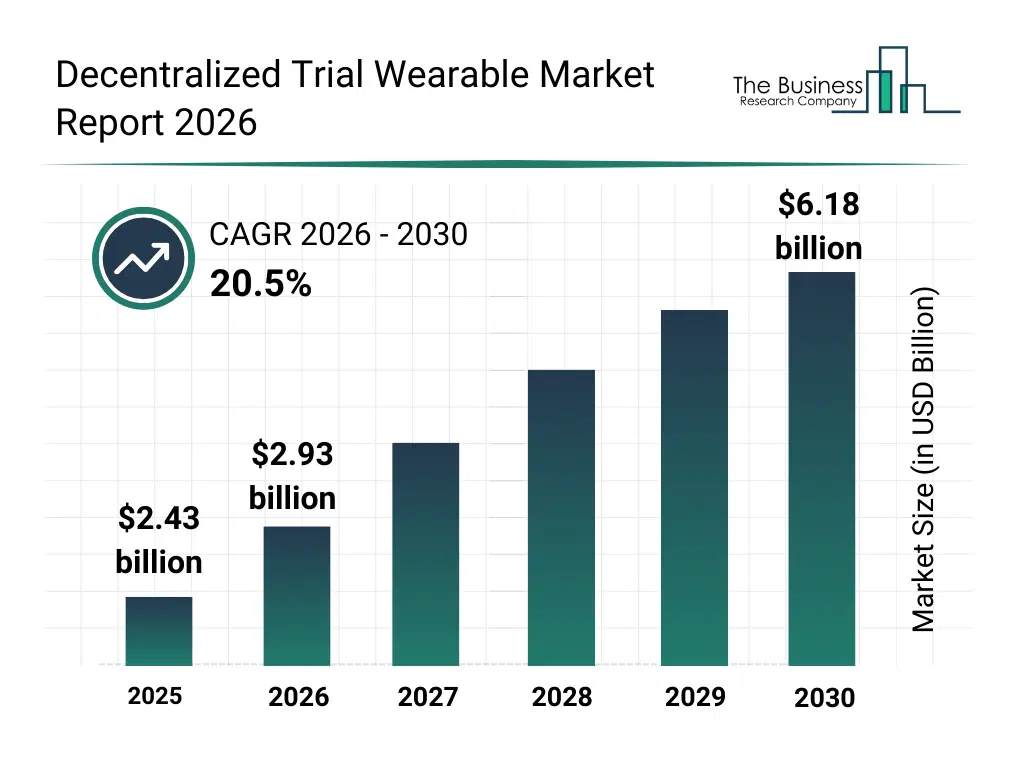
What Is The Decentralized Trial Wearable Market Size and Share 2026?
The decentralized trial wearable market size has grown exponentially in recent years. It will grow from $2.43 billion in 2025 to $2.93 billion in 2026 at a compound annual growth rate (CAGR) of 20.8%. The growth in the historic period can be attributed to increasing adoption of decentralized clinical trial models, rising use of wearable health devices in research, growing demand for remote patient engagement, advancements in sensor miniaturization, expansion of digital clinical trial infrastructure.What Is The Decentralized Trial Wearable Market Growth Forecast?
The decentralized trial wearable market size is expected to see exponential growth in the next few years. It will grow to $6.18 billion in 2030 at a compound annual growth rate (CAGR) of 20.5%. The growth in the forecast period can be attributed to increasing investments in virtual trial platforms, rising demand for real-world evidence generation, expansion of hybrid clinical trial adoption, growing emphasis on regulatory-compliant remote monitoring, increasing use of ai-driven trial analytics. Major trends in the forecast period include increasing adoption of continuous remote health monitoring wearables, rising integration of multi-parameter biosensing devices, growing use of real-time data transmission in clinical trials, expansion of patient-centric trial design enablement, enhanced focus on data accuracy and compliance.Global Decentralized Trial Wearable Market Segmentation
1) By Product Type: Smartwatches, Fitness Trackers, Biosensors, Smart Clothing, Other Product Types 2) By Distribution Channel: Online, Offline 3) By Application: Remote Patient Monitoring, Clinical Trials, Disease Management, Other Applications 4) By End-User: Pharmaceutical Companies, Contract Research Organizations, Healthcare Providers, Other End-Users Subsegments: 1) By Smartwatches: Health Monitoring Smartwatches, Electrocardiogram (ECG) Smartwatches, Blood Oxygen Tracking Smartwatches, Multi-Sensor Smartwatches 2) By Fitness Trackers: Heart Rate Monitoring Fitness Trackers, Activity Tracking Fitness Trackers, Sleep Tracking Fitness Trackers, Multi-Metric Fitness Trackers 3) By Biosensors: Glucose Monitoring Biosensors, Electrocardiogram (ECG) Biosensors, Temperature Sensing Biosensors, Multi-Parameter Biosensors 4) By Smart Clothing: Sensor-Embedded Smart Shirts, Smart Pants With Biometric Sensors, Compression Wear Smart Clothing, Smart Athletic Wear 5) By Other Product Types: Hybrid Wearable Devices, Remote Patient Monitoring Wearables, Medical Grade Wearables, Innovative Material WearablesWhat Is The Driver Of The Decentralized Trial Wearable Market?
The increasing demand for real-time patient monitoring is expected to propel the growth of the decentralized trial wearable market going forward. Real-time patient monitoring refers to the continuous collection and transmission of health data through connected medical devices that automatically relay physiological measurements to healthcare providers for immediate assessment and intervention. The demand for real-time patient monitoring is rising because the increasing prevalence of chronic diseases such as diabetes, cardiovascular disorders, and respiratory illnesses requires continuous tracking of patients’ health to enable early intervention and better disease management. Decentralized trial wearable kit supports real-time patient monitoring by continuously collecting and transmitting health data from patients in their natural environments to centralized databases, enabling researchers and healthcare professionals to remotely track patient health, detect anomalies early, and make timely, data-driven clinical decisions. For instance, in August 2023, according to Vivalink, a US-based healthcare technology company, a recent survey revealed that 84% of current remote patient monitoring (RPM) users intend to expand their use in 2024, 45% of healthcare providers are utilizing RPM for acute monitoring in hospital-at-home programs, and 77% believe that RPM-based care will overtake traditional inpatient hospital care within the next five years. Therefore, the increasing demand for real-time patient monitoring is driving the growth of the decentralized trial wearable industry.Key Players In The Global Decentralized Trial Wearable Market
Major companies operating in the decentralized trial wearable market are Oracle Corporation, IQVIA Holdings Inc., LabCorp Drug Development Inc., ICON Public Limited Company, Garmin Ltd., Clario Inc., Medidata Solutions Inc., BioTelemetry Inc., Signant Health Holding Corp, Withings France SA, Medable Inc., Science 37 Holdings Inc., THREAD Research Inc., Huma Therapeutics Ltd., Empatica S.r.l., Clinical Ink Inc., VivaLNK Inc., Sempulse Ltd., Activinsights Ltd., Mahalo Health Inc.Global Decentralized Trial Wearable Market Trends and Insights
Major companies operating in the decentralized trial wearable market are focusing on developing innovative products such as ultra-compact health monitoring devices to enhance patient comfort and improve remote data collection. Ultra-compact health monitoring devices are small, wearable gadgets designed to continuously track physiological and behavioral data with minimal intrusion, helping improve patient compliance, enable remote monitoring, and provide high-quality data for clinical trials. For instance, in June 2025, Empatica Inc., a US-based medical technology company, launched EmbraceMini, the world’s smallest wristworn actigraphy device for clinical research. Measuring just 12mm thick and 14mm wide, comparable in size to a USB flash drive or AA battery, it is designed for ultra-comfortable, non-invasive wear, enabling continuous monitoring of over 200 digital measures, including activity, sleep, gait, and light exposure. The device wirelessly transmits data to Empatica’s FDA-cleared Health Monitoring Platform, supporting remote data collection, digital biomarker analysis, and integration with existing clinical trial systems, all while prioritizing participant experience and adherence. It is purpose-built for studies on sleep disorders, movement disorders, obesity, depression, and pain, among other conditions, providing high-resolution insights into participants’ physiological and behavioral patterns. Its long battery life allows at least seven days of uninterrupted monitoring, reducing the burden on participants and enhancing compliance. Moreover, the modular design allows it to be worn on the wrist, ankle, leg, or waist, and multiple units can be used simultaneously for richer datasets.What Are Latest Mergers And Acquisitions In The Decentralized Trial Wearable Market?
In May 2024, Medable Inc., a US-based biotechnology company, partnered with Masimo Corporation to bring best-in-class medical-grade wearable devices to decentralized clinical research. With this partnership, Medable Inc. and Masimo Corporation aim to enable decentralized clinical trials by integrating medical-grade wearable devices to capture both objective and subjective patient data, reduce patient burden, and improve data quality. Masimo Corporation is a US-based medical technology company that specializes in providing wearable technology used in decentralized clinical trials.Regional Outlook
North America was the largest region in the decentralized trial wearable market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Decentralized Trial Wearable Market?
The decentralized trial wearable market consists of sales of smart rings, wearable patches, glucose monitors, respiratory sensors, and smart clothing. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Decentralized Trial Wearable Market Report 2026?
The decentralized trial wearable market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the decentralized trial wearable industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Decentralized Trial Wearable Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.93 billion |
| Revenue Forecast In 2035 | $6.18 billion |
| Growth Rate | CAGR of 20.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Oracle Corporation, IQVIA Holdings Inc., LabCorp Drug Development Inc., ICON Public Limited Company, Garmin Ltd., Clario Inc., Medidata Solutions Inc., BioTelemetry Inc., Signant Health Holding Corp, Withings France SA, Medable Inc., Science 37 Holdings Inc., THREAD Research Inc., Huma Therapeutics Ltd., Empatica S.r.l., Clinical Ink Inc., VivaLNK Inc., Sempulse Ltd., Activinsights Ltd., Mahalo Health Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Decentralized Trial Wearable market was valued at $2.43 billion in 2025, increased to $2.93 billion in 2026, and is projected to reach $6.18 billion by 2030.
request a sample hereThe global Decentralized Trial Wearable market is expected to grow at a CAGR of 20.5% from 2026 to 2035 to reach $6.18 billion by 2035.
request a sample hereSome Key Players in the Decentralized Trial Wearable market Include, Oracle Corporation, IQVIA Holdings Inc., LabCorp Drug Development Inc., ICON Public Limited Company, Garmin Ltd., Clario Inc., Medidata Solutions Inc., BioTelemetry Inc., Signant Health Holding Corp, Withings France SA, Medable Inc., Science 37 Holdings Inc., THREAD Research Inc., Huma Therapeutics Ltd., Empatica S.r.l., Clinical Ink Inc., VivaLNK Inc., Sempulse Ltd., Activinsights Ltd., Mahalo Health Inc. .
request a sample hereMajor trend in this market includes: Ultra-Compact Wearable Devices Revolutionize Remote Monitoring In Clinical Trials. For further insights on this market.
request a sample hereNorth America was the largest region in the decentralized trial wearable market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the decentralized trial wearable market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here