
Allergy Clinical Trial Services Market Report 2026
Global Outlook – By Service Type (Study Design And Protocol Development, Site Management And Patient Recruitment, Clinical Operations And Monitoring, Bioanalytical And Laboratory Services, Data Management And Biostatistics, Regulatory Affairs And Medical Writing), By Clinical Trial (Phase I, Phase II, Phase III, Phase IV or Post-Marketing), By Therapeutic Indication (Allergic Rhinitis, Allergic Asthma, Atopic Dermatitis, Food Allergies, Drug Hypersensitivity), By End User (Pharmaceutical Companies, Biotechnology Firms, Academic Research Institutions, Government Organizations) – Market Size, Trends, Strategies, and Forecast to 2035
Allergy Clinical Trial Services Market Overview
• Allergy Clinical Trial Services market size has reached to $1.15 billion in 2025 • Expected to grow to $1.66 billion in 2030 at a compound annual growth rate (CAGR) of 7.5% • Growth Driver: Increasing Incidence Of Allergic Diseases Driving The Market Growth Due To Rising Prevalence Among Children And Adults • Market Trend: Advancements In Decentralized And Hybrid Models Accelerating Allergy Clinical Research Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Allergy Clinical Trial Services Market?
Allergy clinical trial services are specialized research programs that evaluate the safety, efficacy, and dosing of new treatments for allergic conditions, including asthma, rhinitis, and food or drug allergies. These services involve carefully designed protocols, patient monitoring, and data collection to generate scientifically robust results that support regulatory approval and clinical use. The main service types of allergy clinical trial services include study design and protocol development, site management and patient recruitment, clinical operations and monitoring, bioanalytical and laboratory services, data management and biostatistics, and regulatory affairs and medical writing. Study design and protocol development refer to the planning and structuring of clinical trials to ensure scientific validity, regulatory compliance, and patient safety. They are conducted across Phase I, Phase II, Phase III, and Phase IV or post-marketing clinical trials for allergic rhinitis, allergic asthma, atopic dermatitis, food allergies, and drug hypersensitivity therapeutic indications. The key end users include pharmaceutical companies, biotechnology firms, academic research institutions, and government organizations.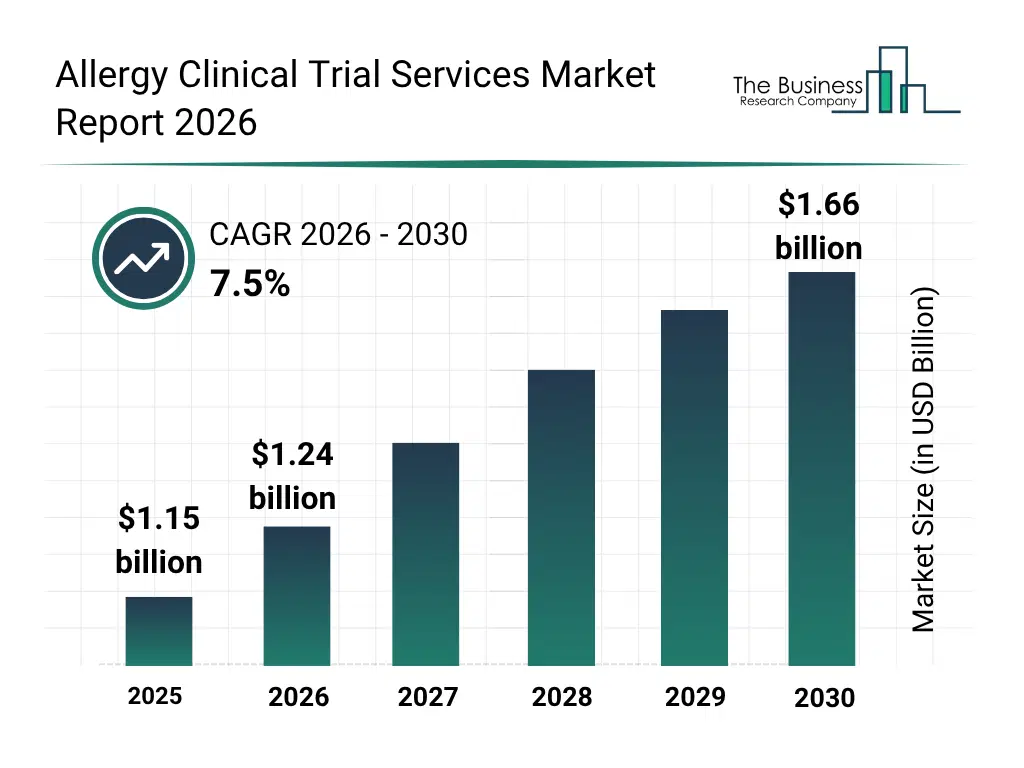
What Is The Allergy Clinical Trial Services Market Size and Share 2026?
The allergy clinical trial services market size has grown strongly in recent years. It will grow from $1.15 billion in 2025 to $1.24 billion in 2026 at a compound annual growth rate (CAGR) of 7.3%. The growth in the historic period can be attributed to increasing prevalence of allergic diseases, rising investment in clinical research, adoption of structured study protocols, expansion of laboratory testing infrastructure, growth of pharmaceutical and biotechnology companies.What Is The Allergy Clinical Trial Services Market Growth Forecast?
The allergy clinical trial services market size is expected to see strong growth in the next few years. It will grow to $1.66 billion in 2030 at a compound annual growth rate (CAGR) of 7.5%. The growth in the forecast period can be attributed to growing focus on precision medicine approaches, increasing integration of ai-driven analytics, rising demand for remote monitoring and decentralized trials, expansion of phase ii and iii allergy studies, growing emphasis on regulatory compliance and data management. Major trends in the forecast period include increasing adoption of remote clinical monitoring services, rising demand for patient recruitment and retention solutions, growing integration of electronic data capture (edc) systems, expansion of bioanalytical and laboratory testing capabilities, rising focus on regulatory compliance and medical writing services.Global Allergy Clinical Trial Services Market Segmentation
1) By Service Type: Study Design And Protocol Development, Site Management And Patient Recruitment, Clinical Operations And Monitoring, Bioanalytical And Laboratory Services, Data Management And Biostatistics, Regulatory Affairs And Medical Writing 2) By Clinical Trial: Phase I, Phase II, Phase III, Phase IV or Post-Marketing 3) By Therapeutic Indication: Allergic Rhinitis, Allergic Asthma, Atopic Dermatitis, Food Allergies, Drug Hypersensitivity 4) By End User: Pharmaceutical Companies, Biotechnology Firms, Academic Research Institutions, Government Organizations Subsegments: 1) By Study Design And Protocol Development: Clinical Protocol Development Services, Trial Design Consulting Services, Feasibility Assessment Services, Endpoint Definition And Selection Services 2) By Site Management And Patient Recruitment: Investigator Site Management Services, Patient Recruitment And Retention Services, Site Training And Support Services, Clinical Site Feasibility Assessment Services 3) By Clinical Operations And Monitoring: On-Site Clinical Monitoring Services, Remote Clinical Monitoring Services, Study Progress Tracking Services, Compliance And Quality Assurance Services 4) By Bioanalytical And Laboratory Services: Pharmacokinetic And Pharmacodynamic Analysis Services, Immunogenicity Testing Services, Biomarker Assay Development Services, Sample Processing And Analysis Services 5) By Data Management And Biostatistics: Electronic Data Capture (EDC) Services, Data Cleaning And Validation Services, Statistical Analysis And Modeling Services, Report Generation And Interpretation Services 6) By Regulatory Affairs And Medical Writing: Regulatory Submission Preparation Services, Clinical Study Reports Writing Services, Investigator Brochure Development Services, Regulatory Consulting And Compliance ServicesWhat Is The Driver Of The Allergy Clinical Trial Services Market?
The increasing prevalence of allergic diseases is expected to propel the growth of the allergy clinical trial services market going forward. Allergic diseases are immune system–mediated hypersensitivity conditions, including seasonal allergic rhinitis, eczema, and food allergies. The prevalence of these conditions is rising, largely due to greater exposure to environmental allergens such as pollen, dust, and air pollutants, which are widely associated with heightened immune system hypersensitivity in susceptible individuals. Allergy clinical trial services support the management and treatment of allergic diseases by enabling the development, testing, and evaluation of new therapies and interventions, thereby helping healthcare providers identify effective solutions and improve patient outcomes. For instance, according to the 2024 National Health Interview Survey (NHIS) by the Centers for Disease Control and Prevention, a US-based government agency, approximately 29.5% of children in the United States had been diagnosed with a seasonal allergy, eczema, or a food allergy, up from 20.6% in the previous survey year. Therefore, the increasing incidence of allergic diseases is driving the growth of the allergy clinical trial services industry.Key Players In The Global Allergy Clinical Trial Services Market
Major companies operating in the allergy clinical trial services market are Johnson & Johnson, Merck & Co. Inc., Sanofi S.A., Novartis International AG, GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd, Stallergenes Greer International AG, Torii Pharmaceutical Co. Ltd., ALK‑Abelló A/S, Allergopharma GmbH & Co. KG, Circassia Group plc, Inmunotek S.L., Biomay AG, Laboratorios LETI S.A., Alerje Inc., Inimmune Corporation, Siolta Therapeutics Inc., Allovate Therapeutics Inc., DBV Technologies S.A., Camallergy S.L.Global Allergy Clinical Trial Services Market Trends and Insights
Major companies operating in the allergy clinical trial services market are focusing on conducting clinical trials to develop innovative products such as, novel anti-inflammatory drugs to improve symptom control and reduce disease severity in allergy patients. Novel anti-inflammatory drugs are newly developed medications designed to target specific immune pathways that cause inflammation, helping to reduce allergic reactions, relieve symptoms, and prevent disease flare-ups more effectively than traditional treatments. For instance, in December 2023, Innovaderm Research, a Canada-based clinical research organization, highlighted its role as a key collaborator in the clinical and study development of a recently published Phase 1 atopic dermatitis study. The study evaluated the safety, tolerability, and preliminary efficacy of an investigational therapy in patients with moderate-to-severe atopic dermatitis, a chronic inflammatory skin condition commonly associated with allergic responses. Innovaderm contributed to protocol development, regulatory strategy, site selection, patient recruitment, and overall operational management, ensuring adherence to clinical and scientific standards. The company leveraged its dermatology-focused expertise, specialized investigator network, and integrated data management capabilities to support accurate data collection and efficient trial execution.What Are Latest Mergers And Acquisitions In The Allergy Clinical Trial Services Market?
In March 2024, eMed Inc., a US-based healthcare technology company, acquired Science 37 Holdings, Inc. for an undisclosed amount. With this acquisition, eMed, Inc. aims to expand its digital clinical trial capabilities, enhance patient access to decentralized trials, accelerate clinical research timelines, and strengthen its technological infrastructure to support a broader range of therapeutic studies globally. Science 37 Holdings Inc. is a US-based healthcare technology company that specializes in providing clinical trial services for allergy-related conditions.Regional Insights
North America was the largest region in the allergy clinical trial services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Allergy Clinical Trial Services Market?
The allergy clinical trial services market includes revenues earned by entities by providing services such as clinical trial design and management, patient recruitment and screening, allergy testing and diagnostics, drug administration and monitoring, data collection and analysis, regulatory compliance support, and biomarker and immunological assessments. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Allergy Clinical Trial Services Market Report 2026?
The allergy clinical trial services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the allergy clinical trial services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Allergy Clinical Trial Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.24 billion |
| Revenue Forecast In 2035 | $1.66 billion |
| Growth Rate | CAGR of 7.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Clinical Trial, Therapeutic Indication, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Johnson & Johnson, Merck & Co. Inc., Sanofi S.A., Novartis International AG, GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd, Stallergenes Greer International AG, Torii Pharmaceutical Co. Ltd., ALK‑Abelló A/S, Allergopharma GmbH & Co. KG, Circassia Group plc, Inmunotek S.L., Biomay AG, Laboratorios LETI S.A., Alerje Inc., Inimmune Corporation, Siolta Therapeutics Inc., Allovate Therapeutics Inc., DBV Technologies S.A., Camallergy S.L. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
