
Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Report 2026
Global Outlook – By Treatment Type (Symptomatic Drug Therapy, Supportive Care Therapy, Palliative Care), By Symptom Management (Seizure Management, Liver Dysfunction Management, Neurodevelopmental Support, Metabolic and Nutritional Support), By Drug Class (Antiepileptic Drugs (excluding valproate), Hepatoprotective and Liver Support Medications, Metabolic Cofactor and Vitamin Therapies, Supportive Adjunct Medications), By Route of Administration (Oral, Injectable), By End User (Hospitals, Specialty Neurology and Metabolic Clinics, Palliative Care Centers) - Market Size, Trends, And Global Forecast 2026-2035
Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Overview
• Alpha-1 Antitrypsin Deficiency Augmentation Therapy market size has reached to $1.8 billion in 2025 • Expected to grow to $2.68 billion in 2030 at a compound annual growth rate (CAGR) of 8.5% • Growth Driver: The Rising Number Of Clinical Trials Is Driving The Market Due To Expanding Research Efforts And Therapeutic Innovation • Market Trend: Expanded Vial Options Enhance Flexibility In AAT Augmentation Therapy • North America was the largest region in 2025.What Is Covered Under Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
Alpha-1 antitrypsin deficiency augmentation therapy is a medical treatment that involves infusing purified alpha-1 antitrypsin protein into individuals with deficient levels to protect the lungs from damage caused by an imbalance of protease and antiprotease enzymes. It aims to slow the progression of emphysema and improve lung function in affected patients. The main types of products in alpha-1 antitrypsin deficiency augmentation therapy are glassia, aralast NP, prolastin C, zemaira and respreeza. Glassia is a medicine containing human Alpha1-Proteinase inhibitors (Alpha1-PI). It is used to treat adults with lung disease (emphysema) caused by severe alpha1-antitrypsin (Alpha1) deficiency. They are administered through oral, injection and inhalations by hospitals, specialty clinics and others.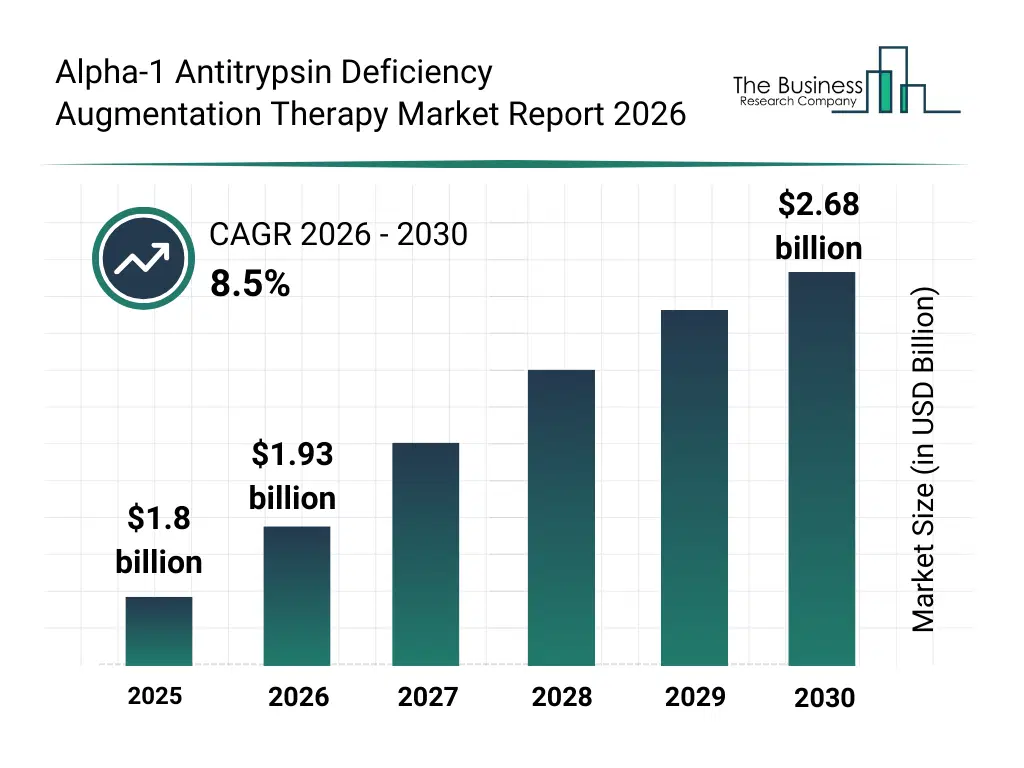
What Is The Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Size and Share 2026?
The alpha-1 antitrypsin deficiency augmentation therapy market size has grown strongly in recent years. It will grow from $1.8 billion in 2025 to $1.93 billion in 2026 at a compound annual growth rate (CAGR) of 7.3%. The growth in the historic period can be attributed to limited treatment options for alpers disease, increasing awareness of rare genetic disorders, reliance on hospital-based care, growing pediatric patient population, expansion of supportive care programs.What Is The Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Growth Forecast?
The alpha-1 antitrypsin deficiency augmentation therapy market size is expected to see strong growth in the next few years. It will grow to $2.68 billion in 2030 at a compound annual growth rate (CAGR) of 8.5%. The growth in the forecast period can be attributed to development of gene-targeted therapies, rising investment in rare disease research, increasing collaboration between hospitals and biotech firms, adoption of innovative drug delivery technologies, expansion of specialty clinics for pediatric neurology. Major trends in the forecast period include rising demand for symptomatic and supportive care treatments, increasing focus on rare genetic disorder research, growth in pediatric neurology therapeutics, expansion of specialty clinics and hospital services, adoption of novel drug formulations and delivery methods.Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Segmentation
1) By Treatment Type: Symptomatic Drug Therapy, Supportive Care Therapy, Palliative Care 2) By Symptom Management: Seizure Management, Liver Dysfunction Management, Neurodevelopmental Support, Metabolic and Nutritional Support 3) By Drug Class: Antiepileptic Drugs (excluding valproate), Hepatoprotective and Liver Support Medications, Metabolic Cofactor and Vitamin Therapies, Supportive Adjunct Medications 4) By Route of Administration: Oral, Injectable 5) By End User: Hospitals, Specialty Neurology and Metabolic Clinics, Palliative Care Centers Subsegments: 1) By Symptomatic Drug Therapy: Antiepileptic Drug Therapy (non-valproate based), Acute Seizure Control Medications, Management of Movement Disorders and Spasticity, Supportive Medications for Gastrointestinal Symptoms, Infection Management and Prophylactic Medications 2) By Supportive Care Therapy: Liver Function Monitoring and Support, Nutritional Support and Metabolic Supplementation, Respiratory Support and Assisted Ventilation, Physical, Occupational, and Speech Therapy, Management of Metabolic Decompensation 3) By Palliative Care: Pain and Symptom Relief Management, End-of-Life Supportive Care, Psychological and Family CounselingWhat Is The Driver Of The Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
The rising number of clinical trials is expected to propel the growth of the alpha-1 antitrypsin deficiency augmentation therapy market going forward. Clinical trials are biomedical research studies conducted in human participants to evaluate the safety, efficacy, and optimal use of therapeutic interventions, including new recombinant therapies, gene therapies, and long-acting augmentation products. The increase in clinical trials is primarily due to growing research investment, heightened interest in next-generation treatment modalities, and the urgent need to address the significant unmet medical burden associated with alpha-1 antitrypsin deficiency. Alpha-1 antitrypsin deficiency augmentation therapy helps by restoring functional AAT protein levels in the blood and lungs, slowing disease progression, reducing lung tissue damage, and improving long-term respiratory outcomes in affected individuals. For instance, in December 2024, according to ClinicalTrials.gov, a US-based government clinical trial registry, clinical trial registrations rose from 477,203 at the start of 2024 to 558,474 by late 2025, reflecting tens of thousands of new studies added each year. Therefore, the surge in clinical trial activity is driving the growth of the alpha-1 antitrypsin deficiency augmentation therapy industry.Key Players In The Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market
Major companies operating in the alpha-1 antitrypsin deficiency augmentation therapy market are Pfizer Inc., Sanofi S.A., Takeda Pharmaceutical Company Limited, Biogen Inc., UCB S.A., Eisai Co. Ltd., Jazz Pharmaceuticals plc, GW Pharmaceuticals plc, BioMarin Pharmaceutical Inc., Ultragenyx Pharmaceutical Inc., PTC Therapeutics Inc., Alexion Pharmaceuticals Inc., Ionis Pharmaceuticals Inc., Vertex Pharmaceuticals Incorporated, Roche Holding AG, Novartis AG, Merck & Co. Inc., AbbVie Inc., Bristol Myers Squibb Company, Lundbeck A/SGlobal Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Trends and Insights
Major companies operating in the alpha-1 antitrypsin deficiency augmentation therapy market are focusing on developing innovative products such as augmentation therapy vials to simplify dosing, optimize infusion workflows, and improve patient treatment experiences. An augmentation therapy vial is a plasma-derived alpha-1 antitrypsin product packaged in standardized quantities to support accurate weight-based dosing and sustained AAT protein replacement. For instance, in February 2024, Grifols, a Spain-based producer of plasma-derived medicines, launched PROLASTIN 4-gram and 5-gram vials. These larger vial formats enable clinicians to match weekly dosing requirements more efficiently while reducing packaging waste and storage needs. The expanded options support more streamlined treatment preparation and strengthen the long-established PROLASTIN portfolio for patients with severe alpha-1 antitrypsin deficiency.What Are Latest Mergers And Acquisitions In The Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
In January 2023, GlaxoSmithKline PLC, a UK-based pharmaceutical and biotechnology company, acquired an 11% Stake in Wave Life Sciences Ltd. for $50 million. This acquisition aims to capitalize on GSK's expertise in human genetics, worldwide development and commercial capabilities in conjunction with Wave's exclusive discovery and drug development platform, PRISMTM. The aim is to accelerate the progress of oligonucleotide therapeutics for genetically determined diseases. Wave Life Sciences Ltd. is a US-based clinical-stage RNA medicine company, including the development of drug for Alpha-1 Antitrypsin Deficiency (AATD).Regional Outlook
North America was the largest region in the alpha-1 antitrypsin deficiency augmentation therapy market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
The alpha-1 antitrypsin deficiency augmentation therapy market consists of sales of Fazirsiran and RNA interference (RNAi) therapy. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Report 2026?
The alpha-1 antitrypsin deficiency augmentation therapy market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the alpha-1 antitrypsin deficiency augmentation therapy industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.93 billion |
| Revenue Forecast In 2035 | $2.68 billion |
| Growth Rate | CAGR of 7.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Type, Symptom Management, Drug Class, Route of Administration, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Sanofi S.A., Takeda Pharmaceutical Company Limited, Biogen Inc., UCB S.A., Eisai Co. Ltd., Jazz Pharmaceuticals plc, GW Pharmaceuticals plc, BioMarin Pharmaceutical Inc., Ultragenyx Pharmaceutical Inc., PTC Therapeutics Inc., Alexion Pharmaceuticals Inc., Ionis Pharmaceuticals Inc., Vertex Pharmaceuticals Incorporated, Roche Holding AG, Novartis AG, Merck & Co. Inc., AbbVie Inc., Bristol Myers Squibb Company, Lundbeck A/S |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
