
Antimicrobial Resistance Diagnostics Market Report 2026
Global Outlook – By Product Type (Kits And Reagents, Instruments And Consumables), By Pathogen (Drug-Resistant Streptococcus Pneumoniae (DRSP), Drug Resistant Campylobacter (DRC), Clostridium Difficile (CD), Methicillin Resistant Staphylococcus Aureus (MRSA), Drug Resistant Neisseria Gonorrhoeae (DRNG), Drug Resistant Salmonella (DRntS), Other Pathogens), By Technology (Microbiology Culture, Immunoassay, PCR (Polymerase Chain Reaction), NGS (Next-Generation Sequencing), Mass Spectrometry, Rapid And Point Of Care, Other Technologies), By End-User (Hospitals And Clinics, Diagnostics Laboratories, Research Institutes, Pharmaceutical And Biotechnology Companies, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Antimicrobial Resistance Diagnostics Market Overview
• Antimicrobial Resistance Diagnostics market size has reached to $4.41 billion in 2025 • Expected to grow to $6.23 billion in 2030 at a compound annual growth rate (CAGR) of 7.1% • Growth Driver: Rising Drug-Resistant Infections Driving The Growth Of The Market Due To The Need For Early Detection And Targeted Treatment • Market Trend: Technological Advancement In Rapid Point-Of-Care Diagnostics • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Antimicrobial Resistance Diagnostics Market?
Antimicrobial resistance (AMR) diagnostics are tests used to detect the presence of microorganisms that have developed resistance to antibiotics or other antimicrobial agents. These diagnostics help identify the specific pathogen and determine which drugs it is resistant or susceptible to, enabling targeted and effective treatment. Antimicrobial resistance (AMR) diagnostics are essential for controlling the spread of resistant infections and guiding appropriate antimicrobial use in both clinical and public health settings. The main product types in the antimicrobial resistance diagnostics market include kits and reagents, instruments, and consumables. Kits and reagents are standardized sets of chemicals and biological materials used to detect or measure specific pathogens or substances in diagnostic tests. These diagnostics are used to detect various pathogens such as drug-resistant Streptococcus pneumoniae (DRSP), drug-resistant Campylobacter (DRC), Clostridium difficile (CD), methicillin-resistant Staphylococcus aureus (MRSA), drug-resistant Neisseria gonorrhoeae (DRNG), drug-resistant Salmonella (DRNTS), and other resistant pathogens. A range of technologies is employed, including microbiology culture, immunoassay, polymerase chain reaction (PCR), next-generation sequencing (NGS), mass spectrometry, rapid and point-of-care testing, and others. The key end-users are hospitals and clinics, diagnostic laboratories, research institutes, pharmaceutical and biotechnology companies, and others.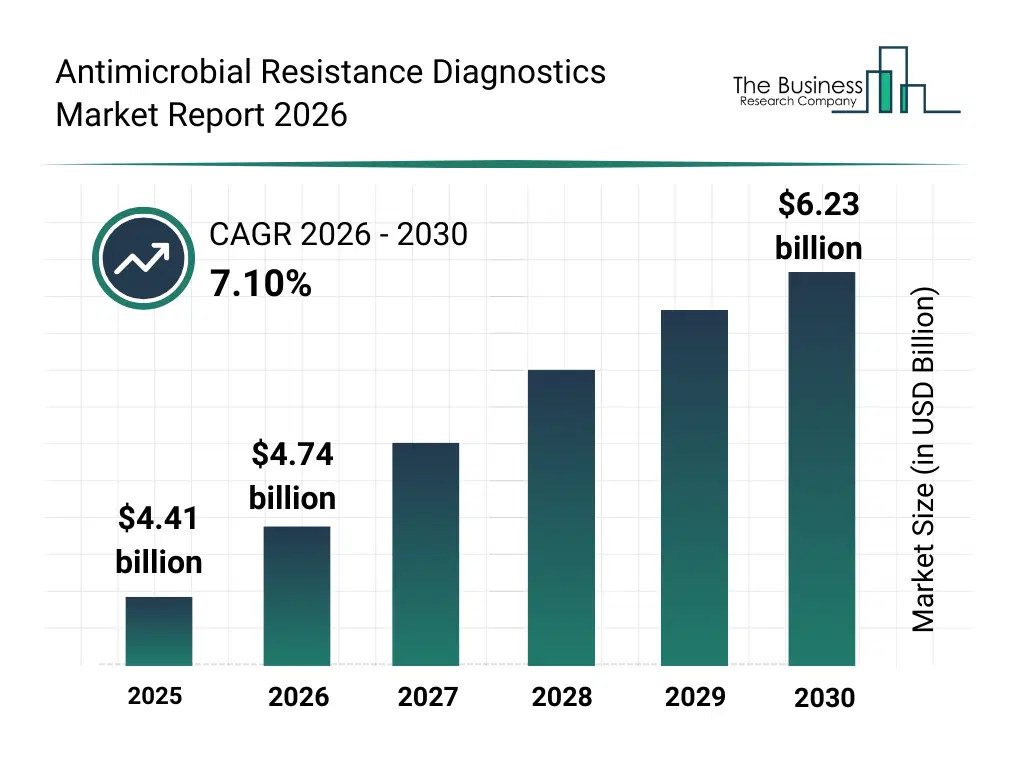
What Is The Antimicrobial Resistance Diagnostics Market Size and Share 2026?
The antimicrobial resistance diagnostics market size has grown strongly in recent years. It will grow from $4.41 billion in 2025 to $4.74 billion in 2026 at a compound annual growth rate (CAGR) of 7.5%. The growth in the historic period can be attributed to increasing prevalence of antibiotic-resistant infections, reliance on conventional culture-based diagnostics, rising hospital-acquired infections, growing awareness of antimicrobial stewardship, expansion of clinical microbiology laboratories.What Is The Antimicrobial Resistance Diagnostics Market Growth Forecast?
The antimicrobial resistance diagnostics market size is expected to see strong growth in the next few years. It will grow to $6.23 billion in 2030 at a compound annual growth rate (CAGR) of 7.1%. The growth in the forecast period can be attributed to advancement in pcr and ngs diagnostic technologies, increasing government funding for amr surveillance programs, rising adoption of rapid diagnostic tests, growth in pharmaceutical and biotechnology research, integration of data-driven decision support tools. Major trends in the forecast period include rising adoption of rapid and point-of-care amr diagnostics, growing demand for targeted and personalized antimicrobial therapy, increased surveillance of drug-resistant pathogens, expansion of molecular and genomic-based diagnostic testing, strengthening of hospital infection control and stewardship programs.Global Antimicrobial Resistance Diagnostics Market Segmentation
1) By Product Type: Kits And Reagents, Instruments And Consumables 2) By Pathogen: Drug-Resistant Streptococcus Pneumoniae (DRSP), Drug Resistant Campylobacter (DRC), Clostridium Difficile (CD), Methicillin Resistant Staphylococcus Aureus (MRSA), Drug Resistant Neisseria Gonorrhoeae (DRNG), Drug Resistant Salmonella (DRntS), Other Pathogens 3) By Technology: Microbiology Culture, Immunoassay, PCR (Polymerase Chain Reaction), NGS (Next-Generation Sequencing), Mass Spectrometry, Rapid And Point Of Care, Other Technologies 4) By End-User: Hospitals And Clinics, Diagnostics Laboratories, Research Institutes, Pharmaceutical And Biotechnology Companies, Other End Users Subsegments: 1) By Kits And Reagents: Deoxyribonucleic Acid (DNA) Extraction Kits, Ribonucleic Acid (RNA) Extraction Kits, Polymerase Chain Reaction (PCR) Reagents, Immunoassay Reagents, Stains And Dyes, Culture Media 2) By Instruments And Consumables: Polymerase Chain Reaction (PCR) Machines, Ultraviolet-Visible (UV-Vis) Spectrophotometers, Next-Generation Sequencing (NGS) Systems, Microarray Scanners, Automated Liquid Handlers, Sample Collection Tubes And SwabsWhat Is The Driver Of The Antimicrobial Resistance Diagnostics Market?
The rising prevalence of drug-resistant infections is expected to propel the growth of the antimicrobial resistance diagnostics market going forward. Drug-resistant infections occur when bacteria, viruses, fungi, or parasites evolve mechanisms to survive exposure to medications that were previously effective against them, making standard treatments ineffective and infections harder to control. The growing prevalence of drug-resistant infections is due to the overuse of antibiotics in agriculture, where low-dose antimicrobials are routinely added to animal feed, promoting resistance that can spread to humans through the food chain. Antimicrobial resistance diagnostics, such as rapid genetic assays and culture-based sensitivity tests, play a pivotal role in combating the growing prevalence of drug-resistant infections by enabling early identification of resistant pathogens, guiding targeted therapies, and reducing the misuse of broad-spectrum antibiotics. For instance, in November 2024, according to the UK Health Security Agency (UKHSA), a UK-based government agency, the agency reported that the number of serious antibiotic-resistant infections in England rose from 58,224 in 2022 to an estimated 66,730 in 2023, while antibiotic use increased to 2.4% in 2023 from 2022. Therefore, the rising prevalence of drug-resistant infections is driving the growth of the antimicrobial resistance diagnostics industry.Key Players In The Global Antimicrobial Resistance Diagnostics Market
Major companies operating in the antimicrobial resistance diagnostics market are F Hoffmann La Roche AG, Thermo Fisher Scientific Inc, Abbott Laboratories, Danaher Corporation, Becton Dickinson And Company, bioMerieux SA, Sysmex Corporation, Bio Rad Laboratories Inc, QIAGEN NV, Seegene Inc, Alifax S r l, Accelerate Diagnostics Inc, Molsid SAS, Genetic Signatures Limited, iFAST Diagnostics Ltd, Astek Diagnostics Inc, OpGen Inc, T2 Biosystems, Jiangsu Medomics Medical Technology Co Ltd, Cepheid, Luminex Corporation, Bruker Corporation, Hologic IncGlobal Antimicrobial Resistance Diagnostics Market Trends and Insights
Major companies operating in the antimicrobial resistance (AMR) diagnostics market are focusing on leveraging innovative technologies such as point-of-care antimicrobial-susceptibility testing (AST) technologies to enhance diagnostic speed, precision, and antimicrobial-stewardship outcomes. Point-of-care antimicrobial-susceptibility testing (AST) refers to rapid, near-patient tests that identify which antibiotics will work against an infection, helping clinicians choose effective, targeted treatment quickly. For instance, in June 2023, Sysmex Corporation, a Japan-based provider of in-vitro diagnostic solutions, launched the PA-100 AST System, a point-of-care rapid AST platform designed to detect bacterial presence and determine antimicrobial susceptibility in urine samples within approximately 30 minutes. The system features microfluidic single-cell analysis, near-patient testing capability, and automated reporting of susceptibility outcomes. The PA-100 AST System improves clinical decision-making, reduces unnecessary antibiotic prescription, and supports timely, targeted therapy to combat antimicrobial resistance.What Are Latest Mergers And Acquisitions In The Antimicrobial Resistance Diagnostics Market?
In June 2025, bioMérieux, a France-based provider of in vitro diagnostic (IVD) solutions and infectious disease testing technologies, acquired the assets of Day Zero Diagnostics for an undisclosed amount. With this acquisition, bioMérieux gains access to Day Zero Diagnostics’ next-generation sequencing (NGS) workflows, NGS chemistries, bioinformatics pipeline, and artificial intelligence (AI)-driven antimicrobial resistance (AMR) prediction technologies, thereby strengthening its next-generation antimicrobial resistance diagnostics capabilities while enhancing its innovation pipeline in rapid pathogen identification and antimicrobial susceptibility testing (AST). Day Zero Diagnostics is a US–based developer of sequencing-based infectious disease diagnostic technologies.Regional Outlook
North America was the largest region in the antimicrobial resistance diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Antimicrobial Resistance Diagnostics Market?
The antimicrobial resistance diagnostics market consists of revenues earned by entities by providing services such as pathogen identification, resistance gene detection, rapid screening, antimicrobial susceptibility testing, and diagnostic data interpretation. The market value includes the value of related goods sold by the service provider or included within the service offering. The antimicrobial resistance diagnostics market includes sales of polymerase chain reaction (PCR) instruments, immunoassay kits, point-of-care testing devices, sequencing reagents, and microbiology culture consumables. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Antimicrobial Resistance Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.74 billion |
| Revenue Forecast In 2035 | $6.23 billion |
| Growth Rate | CAGR of 7.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Pathogen, Technology, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F Hoffmann La Roche AG, Thermo Fisher Scientific Inc, Abbott Laboratories, Danaher Corporation, Becton Dickinson And Company, bioMerieux SA, Sysmex Corporation, Bio Rad Laboratories Inc, QIAGEN NV, Seegene Inc, Alifax S r l, Accelerate Diagnostics Inc, Molsid SAS, Genetic Signatures Limited, iFAST Diagnostics Ltd, Astek Diagnostics Inc, OpGen Inc, T2 Biosystems, Jiangsu Medomics Medical Technology Co Ltd, Cepheid, Luminex Corporation, Bruker Corporation, Hologic Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
