
Aptamer Diagnostic Kits Market Report 2026
Global Outlook – By Product Type (Deoxyribonucleic Acid (DNA) Aptamer Kits, Ribonucleic Acid (RNA) Aptamer Kits, Peptide Aptamer Kits), By Technology (Electrochemical, Optical, Colorimetric, Fluorescent, Other Technologies), By Application (Infectious Disease Diagnosis, Cancer Diagnosis, Cardiovascular Disease Diagnosis, Neurological Disorders, Other Applications), By End-User (Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Aptamer Diagnostic Kits Market Overview
• Aptamer Diagnostic Kits market size has reached to $1.85 billion in 2025 • Expected to grow to $3.97 billion in 2030 at a compound annual growth rate (CAGR) of 16.5% • Growth Driver: The Increasing Prevalence Of Infectious Diseases Is Fueling The Market Due To The Growing Need For Rapid, Accurate, And Early Detection Of Pathogens And Disease Biomarkers • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Aptamer Diagnostic Kits Market?
Aptamer diagnostic kits are analytical tools that use synthetic single-stranded DNA or RNA molecules, called aptamers, which can specifically bind to target molecules with high affinity. They detect the presence of biomolecules, pathogens, or chemical compounds by generating measurable signals upon binding. These kits offer rapid, sensitive, and selective diagnostic capabilities for various medical, environmental, and research applications. The main product types of aptamer diagnostic kits include deoxyribonucleic acid (DNA) aptamer kits, ribonucleic acid (RNA) aptamer kits, and peptide aptamer kits. Deoxyribonucleic acid (DNA) aptamer kits refer to kits that use synthetic deoxyribonucleic acid sequences to specifically bind to target molecules for accurate and rapid diagnostics. These solutions are available with electrochemical, optical, colorimetric, fluorescent, and other technologies. The various applications involved are infectious disease diagnosis, cancer diagnosis, cardiovascular disease diagnosis, neurological disorders, and other applications, and they are used by several end users such as hospitals and clinics, diagnostic laboratories, research institutes, and other end-users.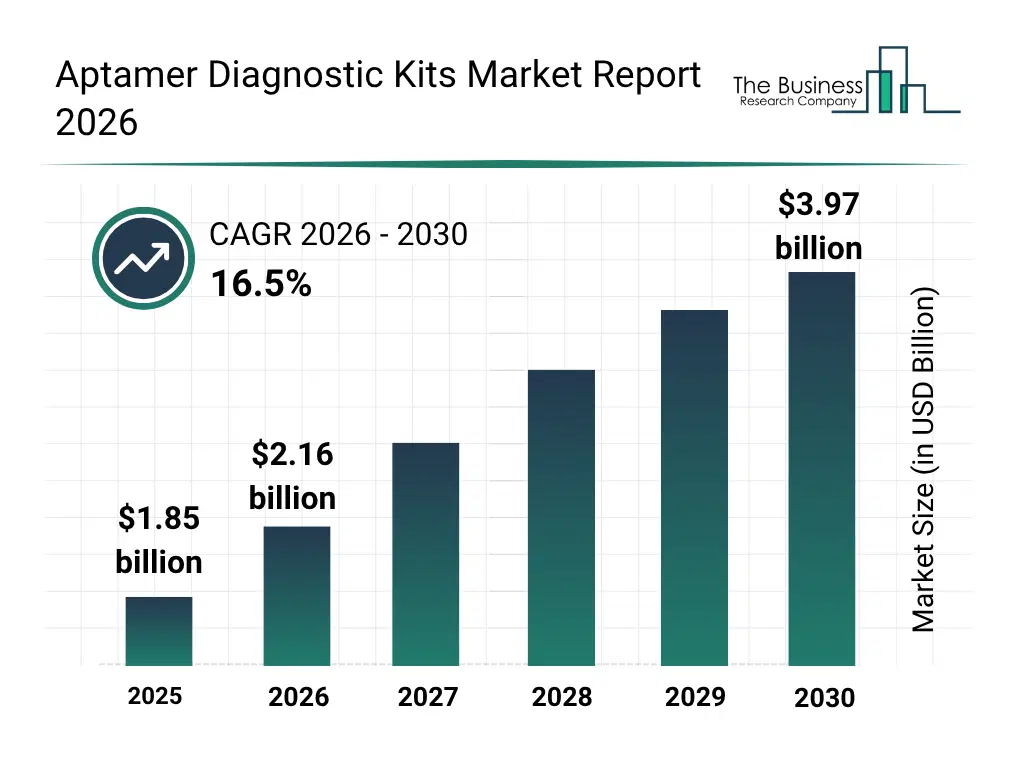
What Is The Aptamer Diagnostic Kits Market Size and Share 2026?
The aptamer diagnostic kits market size has grown rapidly in recent years. It will grow from $1.85 billion in 2025 to $2.16 billion in 2026 at a compound annual growth rate (CAGR) of 16.2%. The growth in the historic period can be attributed to increasing prevalence of infectious diseases, rising demand for sensitive diagnostic tools, growth in hospital and laboratory infrastructure, adoption of enzyme-linked and colorimetric kits, technological advances in aptamer synthesis.What Is The Aptamer Diagnostic Kits Market Growth Forecast?
The aptamer diagnostic kits market size is expected to see rapid growth in the next few years. It will grow to $3.97 billion in 2030 at a compound annual growth rate (CAGR) of 16.5%. The growth in the forecast period can be attributed to growing use of RNA and peptide aptamer kits, increasing integration with AI-based diagnostic analytics, rising demand for personalized medicine applications, expansion of point-of-care and rapid diagnostic testing, growing adoption in cancer and cardiovascular disease diagnosis. Major trends in the forecast period include increasing adoption of high-sensitivity aptamer kits, rising demand for multiplex diagnostic capabilities, growing integration of fluorescence and electrochemical technologies, expansion of infectious disease diagnostic applications, rising focus on rapid point-of-care testing.Global Aptamer Diagnostic Kits Market Segmentation
1) By Product Type: Deoxyribonucleic Acid (DNA) Aptamer Kits, Ribonucleic Acid (RNA) Aptamer Kits, Peptide Aptamer Kits 2) By Technology: Electrochemical, Optical, Colorimetric, Fluorescent, Other Technologies 3) By Application: Infectious Disease Diagnosis, Cancer Diagnosis, Cardiovascular Disease Diagnosis, Neurological Disorders, Other Applications 4) By End-User: Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End-Users Subsegments: 1) By Deoxyribonucleic Acid (DNA) Aptamer Kits: Enzyme-Linked Deoxyribonucleic Acid (DNA) Aptamer Kits, Fluorescence-Based Deoxyribonucleic Acid (DNA) Aptamer Kits, Colorimetric Deoxyribonucleic Acid (DNA) Aptamer Kits, Electrochemical Deoxyribonucleic Acid (DNA) Aptamer Kits 2) By Ribonucleic Acid (RNA) Aptamer Kits: Fluorescence-Based Ribonucleic Acid (RNA) Aptamer Kits, Electrochemical Ribonucleic Acid (RNA) Aptamer Kits, Colorimetric Ribonucleic Acid (RNA) Aptamer Kits, High-Sensitivity Ribonucleic Acid (RNA) Aptamer Kits 3) By Peptide Aptamer Kits: Fluorescence-Based Peptide Aptamer Kits, Enzyme-Linked Peptide Aptamer Kits, Electrochemical Peptide Aptamer Kits, Multiplex Peptide Aptamer KitsWhat Are The Drivers Of The Aptamer Diagnostic Kits Market?
The increasing prevalence of infectious diseases is expected to propel the growth of the aptamer diagnostic kits market going forward. Infectious diseases refer to illnesses caused by pathogenic microorganisms, such as bacteria, viruses, fungi, or parasites, that can be transmitted from one person, animal, or environment to another. Infectious diseases are illnesses caused by pathogenic microorganisms, including bacteria, viruses, fungi, and parasites, that can spread between individuals, animals, or through environmental exposure. Aptamer diagnostic kits help in managing infectious diseases by enabling rapid, highly sensitive, and specific detection of pathogens or disease biomarkers in patient samples, allowing for early diagnosis, timely treatment, and improved disease monitoring and control. For instance, in March 2024, according to the Centers for Disease Control and Prevention, a US-based government agency, the number of tuberculosis cases increased from 8,320 in 2022 to 9,615 in 2023, representing a rise of 1,295 cases. Therefore, the increasing prevalence of infectious diseases is driving the growth of aptamer diagnostic kits industry. The increasing investments in healthcare is expected to propel the growth of the aptamer diagnostic kits market going forward. Investments in healthcare refers to the allocation of financial resources by governments, institutions, or private entities to improve medical infrastructure, services, technologies, and access to healthcare. Investments in healthcare are growing due to governments and institutions increasing funding to strengthen medical infrastructure, expand access to healthcare services, and support the adoption of advanced medical technologies. Investments in healthcare support the development and adoption of aptamer-based diagnostic kits, enabling faster, more precise, and cost-effective disease detection while improving patient outcomes. For instance, in January 2026, according to the Centers for Medicare & Medicaid Services, a US-based government agency, national health expenditure (NHE) is projected to grow at an average rate of 5.8% between 2024 and 2033, outpacing the average gross domestic product (GDP) growth of 4.3% and raising the health spending share of GDP from 17.6% in 2023 to 20.3% by 2033. Therefore, the increasing investments in healthcare is driving the growth of the aptamer diagnostic kits industry.Key Players In The Global Aptamer Diagnostic Kits Market
Major companies operating in the aptamer diagnostic kits market are Kaneka Corporation, Maravai LifeSciences, Nexus Dx Inc, Creative Biolabs, Aptitude Medical Systems Inc, Aptamer Group Limited, Base Pair Biotechnologies Inc, Aptamer Sciences Inc, Aptagen LLC, Vivonics Inc, AptaMatrix Inc, Aptus Biosciences, AuramerBio Ltd, Neoncorte Bio, Bio‑Connect B.V, Aptarion Biotech AG, SB Bioscience Co. Ltd, NeoVentures Biotechnology Inc, AM Biotechnologies LLC, NeoVentures Biotechnology Inc.What Are Latest Mergers And Acquisitions In The Aptamer Diagnostic Kits Market?
In May 2025, Aptamer Sciences Co. Ltd., a South Korea-based biotechnology company that specializes in providing aptamer-based diagnostic products and kits, partnered with Apidi Healthcare Pvt. Ltd. to introduce a lung cancer diagnostic kit in India. With this partnership, Aptamer Sciences Co., Ltd. and Apidi Healthcare Pvt. Ltd. aim to expand access to early and accurate lung cancer detection, leverage advanced aptamer-based biomarker technology, and improve diagnostic outcomes for patients. Apidi Healthcare Pvt. Ltd. is an India-based manufacturing company that specializing in diagnostic solutions and healthcare services.Regional Insights
North America was the largest region in the aptamer diagnostic kits market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Aptamer Diagnostic Kits Market?
The aptamer diagnostic kits market consists of sales of next generation ligand kits, multiplexed detection assay kits, and custom aptamer assay development kits. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Aptamer Diagnostic Kits Market Report 2026?
The aptamer diagnostic kits market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the aptamer diagnostic kits industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Aptamer Diagnostic Kits Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.16 billion |
| Revenue Forecast In 2035 | $3.97 billion |
| Growth Rate | CAGR of 16.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Kaneka Corporation, Maravai LifeSciences, Nexus Dx Inc, Creative Biolabs, Aptitude Medical Systems Inc, Aptamer Group Limited, Base Pair Biotechnologies Inc, Aptamer Sciences Inc, Aptagen LLC, Vivonics Inc, AptaMatrix Inc, Aptus Biosciences, AuramerBio Ltd, Neoncorte Bio, Bio‑Connect B.V, Aptarion Biotech AG, SB Bioscience Co. Ltd, NeoVentures Biotechnology Inc, AM Biotechnologies LLC, NeoVentures Biotechnology Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
