
Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Report 2026
Global Outlook – By Component (Software, Services), By Deployment Mode (Cloud-Based, On-Premises), By Application (Site Selection, Patient Recruitment, Protocol Design, Performance Analytics, Other Applications), By End-User (Pharmaceutical Companies, Contract Research Organizations, Academic Research Institutes, Hospitals, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Overview
• Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility market size has reached to $1.53 billion in 2025 • Expected to grow to $4.38 billion in 2030 at a compound annual growth rate (CAGR) of 23.3% • Growth Driver: The Growing Clinical Trials Are Fueling The Growth Of The Market Due To The Rising Demand For Optimized Trial Locations And Enrollment • Market Trend: Advancement In AI-Powered Platforms Transforming Clinical Trial Site Feasibility And Patient Recruitment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market?
Artificial intelligence (AI)-powered clinical trial site feasibility refers to services and platforms that leverage advanced algorithms to evaluate and select optimal clinical trial sites. It analyzes large datasets to predict site performance and patient recruitment potential. The solution enhances decision-making speed and accuracy, reducing the risk of trial delays. It streamlines the feasibility process while improving overall operational efficiency. The main component types of artificial intelligence (AI)-powered clinical trial site feasibility are software and services. Software refers to AI-powered digital platforms and tools that analyze trial data, site performance, and patient demographics to optimize clinical trial site selection and feasibility. The deployment modes are cloud-based and on-premises for various applications such as site selection, patient recruitment, protocol design, performance analytics, and others, and the key end-users include pharmaceutical companies, contract research organizations, academic research institutes, hospitals, and others.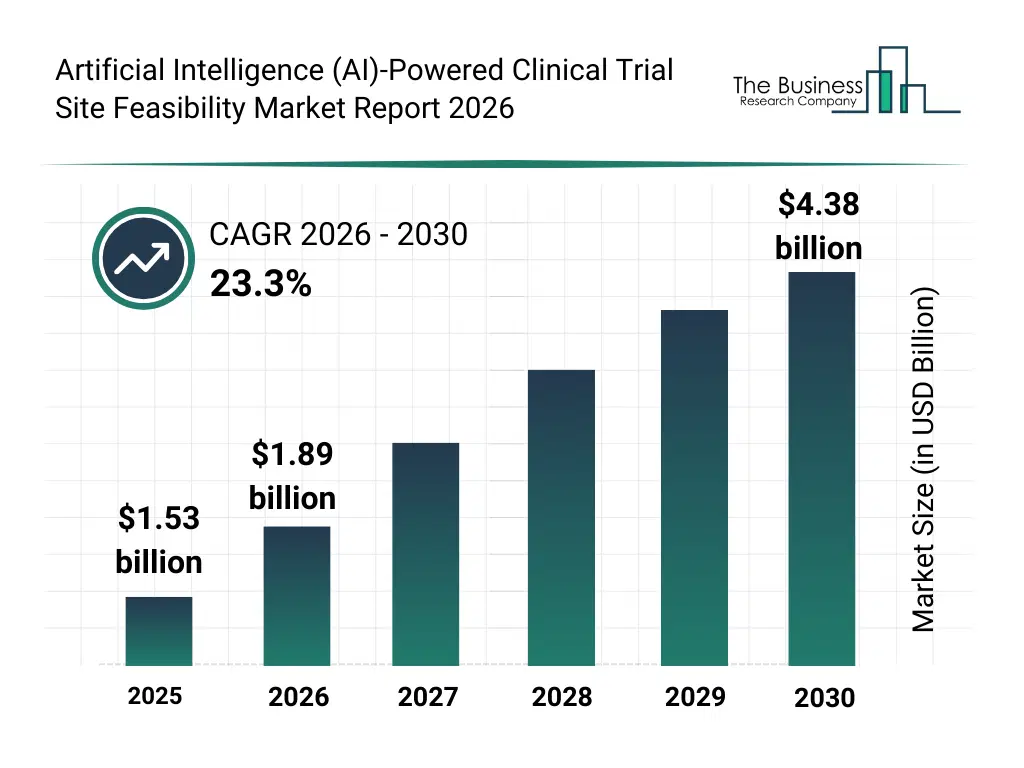
What Is The Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Size and Share 2026?
The artificial intelligence (ai)-powered clinical trial site feasibility market size has grown exponentially in recent years. It will grow from $1.53 billion in 2025 to $1.89 billion in 2026 at a compound annual growth rate (CAGR) of 23.6%. The growth in the historic period can be attributed to increasing complexity of clinical trial designs, rising cost pressures on drug development timelines, growing reliance on data-driven trial planning, expansion of global multi-site clinical trials, early adoption of predictive analytics in trial feasibility.What Is The Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Growth Forecast?
The artificial intelligence (ai)-powered clinical trial site feasibility market size is expected to see exponential growth in the next few years. It will grow to $4.38 billion in 2030 at a compound annual growth rate (CAGR) of 23.3%. The growth in the forecast period can be attributed to increasing adoption of decentralized clinical trials, rising demand for faster site activation, growing use of real-world evidence in feasibility decisions, expansion of ai-enabled trial planning platforms, increasing emphasis on trial risk mitigation. Major trends in the forecast period include increasing adoption of AI-driven site selection models, rising use of predictive patient recruitment analytics, growing integration of real-world data in feasibility assessment, expansion of cloud-based feasibility platforms, enhanced focus on trial performance forecasting.Global Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Segmentation
1) By Component: Software, Services 2) By Deployment Mode: Cloud-Based, On-Premises 3) By Application: Site Selection, Patient Recruitment, Protocol Design, Performance Analytics, Other Applications 4) By End-User: Pharmaceutical Companies, Contract Research Organizations, Academic Research Institutes, Hospitals, Other End-Users Subsegments: 1) By Software: Predictive Analytics Software, Machine Learning (ML) Platforms, Natural Language Processing (NLP) Tools, Data Visualization Software, Clinical Trial Management System (CTMS) Integration Software, Real-World Data (RWD) Analytics Software, Risk-Based Monitoring (RBM) Software 2) By Services: Feasibility Assessment Services, Site Selection Services, Patient Recruitment Support Services, Data Management And Analytics Services, Protocol Optimization Services, Regulatory Compliance Support Services, Consulting And Advisory ServicesWhat Is The Driver Of The Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market?
The growing clinical trials are expected to propel the growth of the artificial intelligence (AI)-powered clinical trial site feasibility market going forward. Clinical trials refer to the clinical research studies being conducted globally to test the safety and efficacy of new medical treatments, drugs, and devices. The rising number of clinical trials is largely driven by increasing investment in biomedical research and development, as both government and private sectors recognize the critical need for innovative treatments to address growing healthcare challenges. The expansion of clinical trials creates demand for AI-powered site feasibility tools as research organizations need efficient systems to identify optimal locations, assess site capabilities, and predict enrollment success rates for their studies. For instance, in April 2025, according to the Multidisciplinary Digital Publishing Institute, a Switzerland-based publisher of open-access scientific journals, the total number of interventional clinical trials, hereafter referred to as clinical trials, available on the clinicaltrials.gov webpage was 404,637. Therefore, growing clinical trials are driving the growth of the artificial intelligence (AI)-powered clinical trial site feasibility industry.Key Players In The Global Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market
Major companies operating in the artificial intelligence (AI)-powered clinical trial site feasibility market are Oracle Corporation, Thermo Fisher Scientific Inc., NVIDIA Corporation, IQVIA Holdings Inc., Labcorp Holdings Inc., Dassault Systèmes SE, Parexel International Corporation, ZS Associates Inc., Veeva Systems Inc., Verily Life Sciences LLC, Signant Health Holding Corp., Saama Technologies LLC, Elligo Health Research Inc., ConcertAI Inc., Medable Inc., Science 37 Inc., Castor EDC Inc., Unlearn.AI Inc., Antidote Technologies Inc., Deep 6 AIGlobal Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Trends and Insights
Major companies operating in the artificial intelligence-powered clinical trial site feasibility market are focusing on developing advanced solutions, such as an artificial intelligence-powered feasibility and recruitment platform to optimize site selection and improve overall operational efficiency in clinical trials. An artificial intelligence-powered feasibility and recruitment platform is a software solution that leverages AI and machine learning to assess trial protocols and site data to identify optimal clinical sites and enhance patient recruitment by efficiently matching eligible participants to accelerate trial timelines and boost study success. For instance, in June 2023, ObjectiveHealth, a US-based integrated research and technology platform company, launched ObjectiveScreen, an artificial intelligence-powered feasibility and recruitment platform designed to optimize clinical trial performance by enhancing patient identification, prequalification, and retention through real-time integration with electronic health records (EHRs) and the use of AI and machine learning algorithms. The platform enables clinical trial sites to connect directly with patient data, stratify health risks, and identify the most qualified candidates to accelerate enrollment and improve trial outcomes.What Are Latest Mergers And Acquisitions In The Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market?
In February 2023, ZS Associates, a US-based technology and management consulting firm, acquired Trials.ai for an undisclosed amount. Through this acquisition, ZS Associates aims to enhance its presence in the medical sector by improving clinical trial efficiency, expediting therapy delivery to the market, and providing a better participant experience. Trials.ai is a US-based, healthcare-focused, pure-play AI and analytics company that supports and enhances the process of clinical trial site feasibility.Regional Outlook
North America was the largest region in the artificial intelligence (AI)-powered clinical trial site feasibility market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market?
The artificial intelligence (AI)-powered clinical trial site feasibility market includes revenues earned by providing services such as site identification and selection, predictive enrollment modeling, investigator performance analysis, risk assessment and mitigation, and patient population analytics. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Report 2026?
The artificial intelligence (ai)-powered clinical trial site feasibility market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the artificial intelligence (ai)-powered clinical trial site feasibility industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Artificial Intelligence (AI)-Powered Clinical Trial Site Feasibility Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.89 billion |
| Revenue Forecast In 2035 | $4.38 billion |
| Growth Rate | CAGR of 23.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Component, Deployment Mode, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Oracle Corporation, Thermo Fisher Scientific Inc., NVIDIA Corporation, IQVIA Holdings Inc., Labcorp Holdings Inc., Dassault Systèmes SE, Parexel International Corporation, ZS Associates Inc., Veeva Systems Inc., Verily Life Sciences LLC, Signant Health Holding Corp., Saama Technologies LLC, Elligo Health Research Inc., ConcertAI Inc., Medable Inc., Science 37 Inc., Castor EDC Inc., Unlearn.AI Inc., Antidote Technologies Inc., Deep 6 AI |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
