
Balloon Introducer System Market Report 2026
Global Outlook – By Product Type (Normal Balloon, Cutting And Scoring Balloon, Drug-Coated Balloon, Stent Graft Balloon Introducer), By Technology (Rapid Exchange, Over The Wire, Fixed Wire Balloon Catheter), By Application (Coronary Artery Diseases, Peripheral Artery Diseases, Neurovascular Diseases), By End User (Hospitals, Ambulatory Surgical Centers, Catheterization Laboratories) – Market Size, Trends, Strategies, and Forecast to 2035
Balloon Introducer System Market Overview
• Balloon Introducer System market size has reached to $3.27 billion in 2025 • Expected to grow to $5.23 billion in 2030 at a compound annual growth rate (CAGR) of 9.8% • Growth Driver: Growing Prevalence Of Cardiovascular Diseases To Drive Balloon Introducer System Market Expansion • Market Trend: Cordis Launches Global Coronary Registry To Advance Evidence-Based Innovation In Drug-Eluting Balloon Therapy • North America was the largest region in 2025.What Is Covered Under Balloon Introducer System Market?
A balloon introducer system refers to a medical device used in interventional procedures to facilitate the insertion of a balloon catheter into a patient's vascular system. This system is typically employed during procedures like angioplasty, where a balloon catheter is inserted into a blood vessel to widen narrowed or blocked arteries. The main types of products in the balloon introducer system are normal balloons, cutting and scoring balloons, drug-coated balloons, and stencil balloon introducers. Normal balloons are standard devices used in angioplasty procedures to dilate narrowed or blocked arteries. These products utilize different technologies such as rapid exchange, over-the-Wire, and fixed-wire balloon catheters, making them suitable for various applications such as coronary artery diseases, peripheral artery diseases, and neurological diseases. They cater to a range of end users, including hospitals, ambulatory surgical centers, and catheterization laboratories.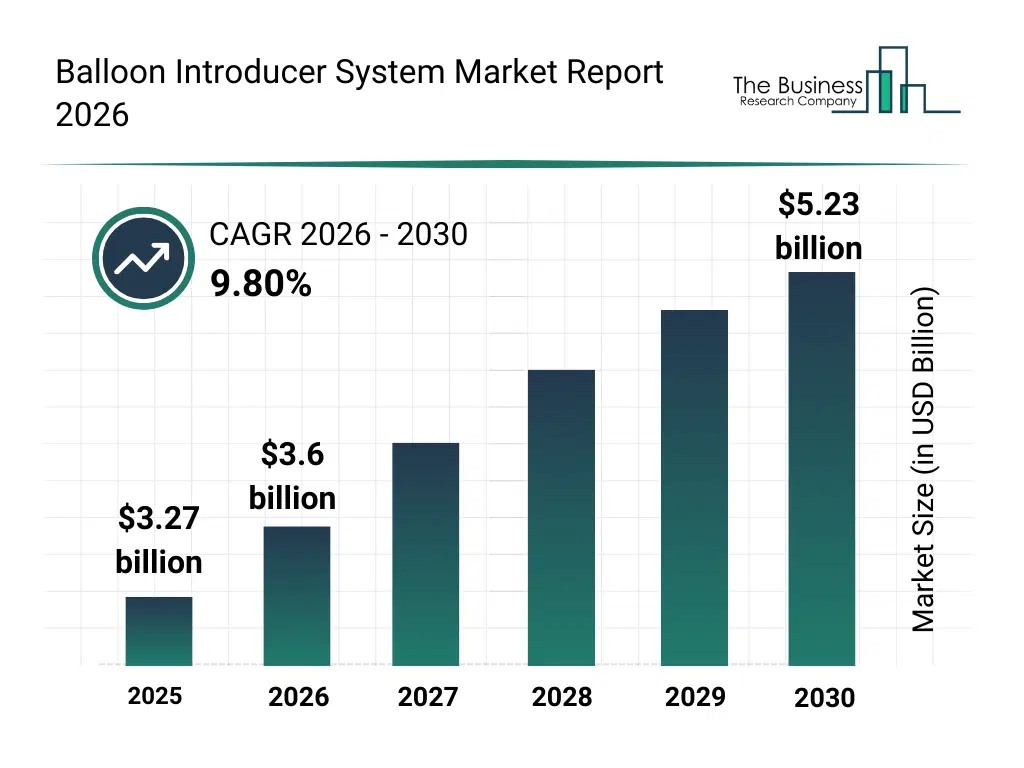
What Is The Balloon Introducer System Market Size and Share 2026?
The balloon introducer system market size has grown rapidly in recent years. It will grow from $3.27 billion in 2025 to $3.6 billion in 2026 at a compound annual growth rate (CAGR) of 10.1%. The growth in the historic period can be attributed to limited availability of advanced balloon introducers, reliance on conventional normal balloons, increasing prevalence of coronary artery diseases, growing hospital interventional capabilities, adoption of stent graft procedures.What Is The Balloon Introducer System Market Growth Forecast?
The balloon introducer system market size is expected to see strong growth in the next few years. It will grow to $5.23 billion in 2030 at a compound annual growth rate (CAGR) of 9.8%. The growth in the forecast period can be attributed to development of cutting and scoring balloon introducers, expansion of drug-coated balloon usage, rising neurovascular intervention procedures, increasing ambulatory surgical centers adoption, integration of smart and automated delivery systems. Major trends in the forecast period include rising adoption of drug-coated balloon technologies, expansion of minimally invasive cardiovascular procedures, growing preference for rapid exchange and over-the-wire systems, increasing awareness of peripheral and neurovascular disease treatments, focus on catheterization laboratory efficiency and workflow optimization.Global Balloon Introducer System Market Segmentation
1) By Product Type: Normal Balloon, Cutting And Scoring Balloon, Drug-Coated Balloon, Stent Graft Balloon Introducer 2) By Technology: Rapid Exchange, Over The Wire, Fixed Wire Balloon Catheter 3) By Application: Coronary Artery Diseases, Peripheral Artery Diseases, Neurovascular Diseases 4) By End User: Hospitals, Ambulatory Surgical Centers, Catheterization Laboratories Subsegments: 1) By Normal Balloon: Standard Balloon Introducers, Specialty Balloon Introducers 2) By Cutting And Scoring Balloon: Cutting Balloon Introducers, Scoring Balloon Introducers 3) By Drug-Coated Balloon: Paclitaxel-Coated Balloons, Sirolimus-Coated Balloons 4) By Stent Graft Balloon Introducer: Stent Graft Delivery Systems, Balloon Expandable Stent Graft IntroducersWhat Is The Driver Of The Balloon Introducer System Market?
The increase in cardiovascular diseases is expected to propel the growth of the balloon introducer system market going forward. Cardiovascular diseases refer to disorders affecting the heart and blood vessels, including heart attacks, strokes, and hypertension. The prevalence of cardiovascular diseases is increasing due to the global rise in obesity and diabetes, coupled with the widespread adoption of sedentary lifestyles and poor dietary habits. A balloon introducer system aids in minimally invasive procedures to open blocked blood vessels, improving blood flow in cardiovascular disease treatment. For instance, in October 2025, according to the Australian Institute Of Health And Welfare, an Australia-based government agency reported that in 2023, an estimated 57,100 adults aged 25 and above experienced acute coronary events, averaging roughly 156 incidents per day. Therefore, the increase in cardiovascular diseases is driving the growth of the balloon introducer system industry.Key Players In The Global Balloon Introducer System Market
Major companies operating in the balloon introducer system market are Cardinal Health Inc., Johnson & Johnson, Abbott Laboratories, Medtronic plc, Boston Scientific Corporation, B. Braun Melsungen AG, Terumo Corporation, Nipro Corporation, Cook Medical Inc., Teleflex Incorporated, BIOTRONIK SE & Co. KG, Merit Medical Systems Inc., MicroPort Scientific Corporation, Blue Sail Medical Co. Ltd., Asahi Intecc Co. Ltd., Japan Lifeline Co. Ltd., AngioDynamics Inc., OrbusNeich Medical, Biosensors International Group Ltd., Alvimedica Medical Technologies, Hexacath, Cardionovum GmbH, Concept Medical Inc, Endocor GmbHGlobal Balloon Introducer System Market Trends and Insights
Major companies in the balloon introducer system market are focusing on advancing evidence-based product innovation and launching large-scale clinical programs to validate performance in real-world settings, as demonstrated by Cordis’ initiation of the SELUTION Global Coronary Registry. For instance, in May 2025, Cordis Corporation, a US-based medical technology company, has announced the launch of its SELUTION Global Coronary Registry, a major initiative designed to evaluate real-world outcomes of the SELUTION SLR Drug-Eluting Balloon in up to 10,000 patients worldwide. This large-scale, five-year registry will strengthen the evidence already generated through pre-clinical studies, proof-of-concept trials, and five ongoing Level-1 randomized studies. Supported by an esteemed global steering committee of leading interventional cardiologists, the program aims to deepen understanding of DEB use in contemporary PCI practice. With strong early data and growing clinical interest, Cordis reinforces its commitment to evidence-based innovation and expects to share significant findings later this year.What Are Latest Mergers And Acquisitions In The Balloon Introducer System Market?
In October 2023, Cordis Corporation, a US-based medical company, acquired M.A. Med Alliance SA for $1.135 billion. Through this acquisition, Cordis aims to bolster its market leadership in drug-eluting balloon technology by integrating the SELUTION SLR technology, enhancing coronary and peripheral artery disease treatment, and reaching up to two million patients globally by 2027. MedAlliance is a Switzerland-based medical technology company specializing in advanced drug-eluting balloons and balloon introducer systems for interventional cardiology and endovascular procedures.Regional Outlook
North America was the largest region in the balloon introducer system market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Balloon Introducer System Market?
The balloon introducer system market consists of sales of balloon introducer sheaths, balloon dilators, hemostatic valves, and guiding catheters. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Balloon Introducer System Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.6 billion |
| Revenue Forecast In 2035 | $5.23 billion |
| Growth Rate | CAGR of 10.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health Inc., Johnson & Johnson, Abbott Laboratories, Medtronic plc, Boston Scientific Corporation, B. Braun Melsungen AG, Terumo Corporation, Nipro Corporation, Cook Medical Inc., Teleflex Incorporated, BIOTRONIK SE & Co. KG, Merit Medical Systems Inc., MicroPort Scientific Corporation, Blue Sail Medical Co. Ltd., Asahi Intecc Co. Ltd., Japan Lifeline Co. Ltd., AngioDynamics Inc., OrbusNeich Medical, Biosensors International Group Ltd., Alvimedica Medical Technologies, Hexacath, Cardionovum GmbH, Concept Medical Inc, Endocor GmbH |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
