
Balloon Valvuloplasty Device Market Report 2026
Global Outlook – By Product Type (Standard Balloon, Cutting Balloon, Scoring Balloon, High-Pressure Balloon, Low-Pressure Balloon), By Age Group (Pediatric, Adult), By End-use (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Other End Uses) – Market Size, Trends, Strategies, and Forecast to 2035
Balloon Valvuloplasty Device Market Overview
• Balloon Valvuloplasty Device market size has reached to $2.57 billion in 2025 • Expected to grow to $3.35 billion in 2030 at a compound annual growth rate (CAGR) of 5.3% • Growth Driver: Minimally Invasive Procedures Boosting Balloon Valvuloplasty Device Market Growth • Market Trend: Advancements In Balloon Valvuloplasty Devices • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Balloon Valvuloplasty Device Market?
A balloon valvuloplasty device is a medical instrument used to treat heart valve stenosis by inserting a balloon into the narrowed valve and inflating it to widen the opening, improving blood flow. This non-surgical procedure is minimally invasive and aims to relieve symptoms such as chest pain and shortness of breath, enhancing cardiac function and patient quality of life. The main product types of balloon valvuloplasty devices are standard balloons, cutting balloons, scoring balloons, high-pressure balloons, and low-pressure balloons. A standard balloon valvuloplasty device is a medical instrument used to treat narrowed heart valves by inflating a balloon at the site of stenosis and widening the valve opening to improve blood flow. It is commonly employed in minimally invasive procedures to alleviate symptoms of valve narrowing and enhance cardiac function. These are used by pediatrics and adults and utilized for various end-uses, such as hospitals, ambulatory surgical centers, specialty clinics, and others.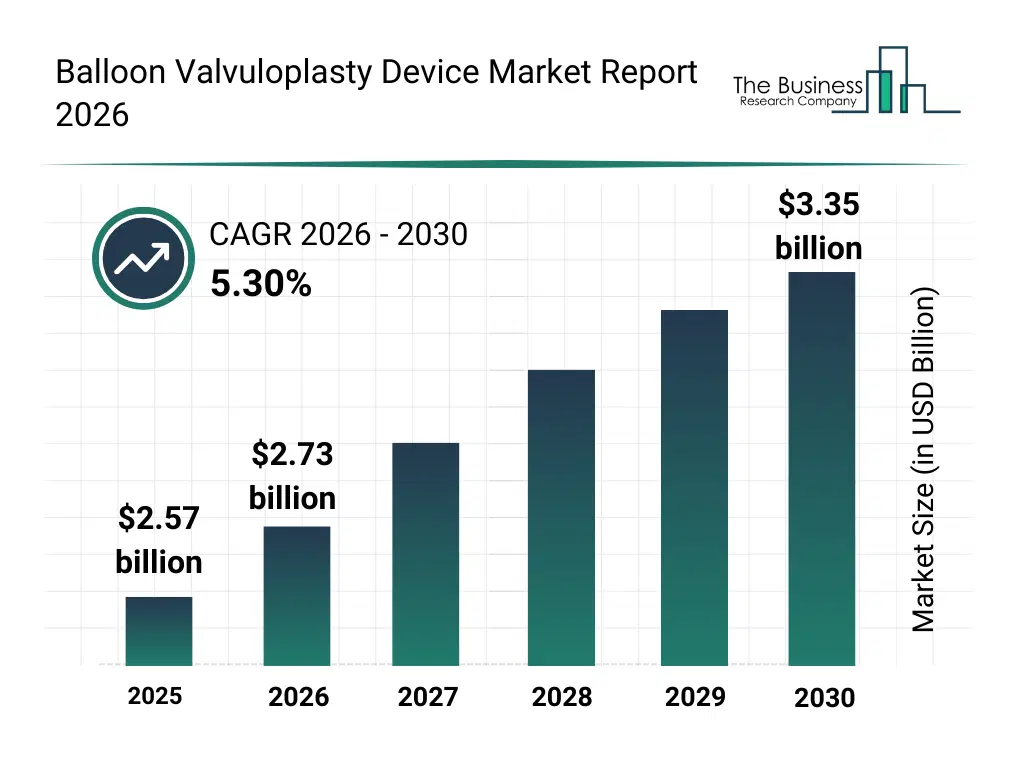
What Is The Balloon Valvuloplasty Device Market Size and Share 2026?
The balloon valvuloplasty device market size has grown strongly in recent years. It will grow from $2.57 billion in 2025 to $2.73 billion in 2026 at a compound annual growth rate (CAGR) of 5.9%. The growth in the historic period can be attributed to rise in prevalence of cardiac valve stenosis, early adoption of minimally invasive interventional cardiology procedures, development of standard and compliant balloon technologies, expansion of hospital-based cardiac catheterization labs, increasing treatment volumes in pediatric congenital heart cases.What Is The Balloon Valvuloplasty Device Market Growth Forecast?
The balloon valvuloplasty device market size is expected to see strong growth in the next few years. It will grow to $3.35 billion in 2030 at a compound annual growth rate (CAGR) of 5.3%. The growth in the forecast period can be attributed to growing aging population with aortic and mitral valve stenosis, rising demand for high-pressure and scoring balloon devices, wider use of balloon valvuloplasty in ambulatory and specialty clinics, increasing shift toward non-surgical cardiac treatment pathways, ongoing innovation in balloon materials and device design. Major trends in the forecast period include increasing preference for minimally invasive valve repair procedures, rising adoption of balloon valvuloplasty as an alternative to open-heart surgery, growing utilization in pediatric and adult congenital heart valve disorders, expansion of valve dilation procedures in specialty cardiology centers, focus on improving patient outcomes and shorter recovery times.Global Balloon Valvuloplasty Device Market Segmentation
1) By Product Type: Standard Balloon, Cutting Balloon, Scoring Balloon, High-Pressure Balloon, Low-Pressure Balloon 2) By Age Group: Pediatric, Adult 3) By End-use: Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Other End Uses Subsegments: 1) By Standard Balloon: Non-Compliant Balloons, Compliant Balloons 2) By Cutting Balloon: Single-Cutting Balloon, Multi-Cutting Balloon 3) By Scoring Balloon: Single-Score Balloons, Dual-Score Balloons 4) By High-Pressure Balloon: Non-Compliant High-Pressure Balloons, Compliant High-Pressure Balloons 5) By Low-Pressure Balloon: Semi-Compliant Low-Pressure Balloons, Non-Compliant Low-Pressure BalloonsWhat Is The Driver Of The Balloon Valvuloplasty Device Market?
The rising inclination towards minimally invasive procedures is expected to propel the growth of the balloon valvuloplasty device market going forward. Minimally invasive procedures refer to medical techniques performed through small incisions or natural body openings, reducing trauma and promoting faster recovery compared to traditional surgeries. The demand for minimally invasive procedures is on the rise due to their reduced recovery times, lower risk of complications, and lower post-operative pain compared to traditional surgeries. Balloon valvuloplasty devices are used in minimally invasive surgical procedures to widen narrowed heart valves, improve blood flow, and reduce symptoms. For instance, in January 2024, Intuitive Surgical Inc., a US-based manufacturer of robotic products, reported that they had placed 415 da Vinci surgical systems (robotic surgical systems that use a minimally invasive surgical approach) in the fourth quarter of 2023, representing a 12% increase compared to 2022. Therefore, the rising inclination towards minimally invasive procedures is driving the growth of the balloon valvuloplasty device industry.Key Players In The Global Balloon Valvuloplasty Device Market
Major companies operating in the balloon valvuloplasty device market are Boston Scientific Corporation, Medtronic plc, Abbott Laboratories, Edwards Lifesciences Corporation, Terumo Corporation, B Braun Melsungen AG, Cook Medical Incorporated, Balton Sp z o o, Meril Life Sciences Pvt Ltd, JenaValve Technology Inc, Osypka Medical GmbH, Scitech Medical, Translumina GmbH, AndraTec GmbH, Palex Medical SA, Biosensors International Group Ltd, Hexacath, MicroPort Scientific Corporation, Lepu Medical Technology Beijing Co Ltd, QT Vascular LtdGlobal Balloon Valvuloplasty Device Market Trends and Insights
Major companies operating in the balloon valvuloplasty device market are focusing on developing advanced products, such as Pacing Guidewires, to gain a competitive edge in the market. A pacing guidewire is a specialized wire used to provide temporary cardiac pacing during the procedure, ensuring the heart maintains a stable rhythm while the balloon catheter is inserted and inflated to dilate the narrowed valve. For instance, in June 2023, Teleflex Incorporated, a US-based medical device company, received clearance from the Food and Drug Administration (FDA) for the Wattson Temporary Pacing Guidewire. This new device is designed specifically for use during transcatheter aortic valve replacement (TAVR) and balloon aortic valvuloplasty (BAV) procedures. The Wattson Temporary Pacing Guidewire offers dual functionality, supporting both valve delivery and ventricular bipolar pacing during these structural heart procedures. Its flexible distal pigtail shape, multiple electrodes, and bipolar design are engineered to help reduce the risk of ventricular perforation while providing reliable electrical capture during rapid pacing.What Are Latest Mergers And Acquisitions In The Balloon Valvuloplasty Device Market?
In November 2023, OrbusNeich Medical Group Holdings Limited, a Hong Kong-based medical device company, acquired Eucatech AG for $2.3 million. The acquisition will allow OrbusNeich to expand its product offerings and provide more comprehensive treatment solutions for patients with cardiovascular diseases. Eucatech AG is a Germany-based medical technology company that provides balloon valvuloplasty devices as part of its product portfolio.Regional Outlook
North America was the largest region in the balloon valvuloplasty device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Balloon Valvuloplasty Device Market?
The balloon valvuloplasty device market consists of sales of scoring balloons, high-pressure balloons, and standard balloons. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Balloon Valvuloplasty Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.73 billion |
| Revenue Forecast In 2035 | $3.35 billion |
| Growth Rate | CAGR of 5.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Age Group, End-use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Boston Scientific Corporation, Medtronic plc, Abbott Laboratories, Edwards Lifesciences Corporation, Terumo Corporation, B Braun Melsungen AG, Cook Medical Incorporated, Balton Sp z o o, Meril Life Sciences Pvt Ltd, JenaValve Technology Inc, Osypka Medical GmbH, Scitech Medical, Translumina GmbH, AndraTec GmbH, Palex Medical SA, Biosensors International Group Ltd, Hexacath, MicroPort Scientific Corporation, Lepu Medical Technology Beijing Co Ltd, QT Vascular Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
