
Bioanalytical Testing Services Market Report 2026
Global Outlook – By Test Type (ADME (Absorption, Distribution, Metabolism, And Excretion), Pharmacokinetic, Pharmacodynamic, Bioequivalence, Bioavailability), By Molecule Type (Small Molecules, Large Molecules), By End User (Pharmaceutical And Biopharmaceutical Companies, Contract Development And Manufacturing Organizations, Contract Research Organizations) – Market Size, Trends, Strategies, and Forecast to 2035
Bioanalytical Testing Services Market Overview
• Bioanalytical Testing Services market size has reached to $3.73 billion in 2025 • Expected to grow to $6.27 billion in 2030 at a compound annual growth rate (CAGR) of 11.1% • Growth Driver: Government-Led Initiatives Propel Bioanalytical Testing Services Market Amidst Pandemic Challenges • Market Trend: Elevating Bioanalytical Testing Services With Advanced Variant Detection Capabilities • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Bioanalytical Testing Services Market?
Bioanalytical testing services refer to services that provide state-of-the-art quantifications of analytes. Bioanalytical testing services help to detect the properties of pharmaceuticals and metabolites in biological fluids such as blood, plasma, serum, urine, or tissue extracts. The main types of tests in bioanalytical testing services are ADME (absorption, distribution, metabolism, and excretion), pharmacokinetic, pharmacodynamic, bioequivalence, and bioavailability. The ADME refers to the absorption, distribution, metabolism, and excretion process which is used to design and find out how a chemical is processed in a living organism. It is used to assess safety and toxicity which informs researchers about the drug viability and gives specific targets for future research and development. The various molecule types tested in bioanalytical testing are small molecules and large molecules, and these testing services are used by pharmaceutical and biopharmaceutical companies, contract development and manufacturing organizations, and contract research organizations.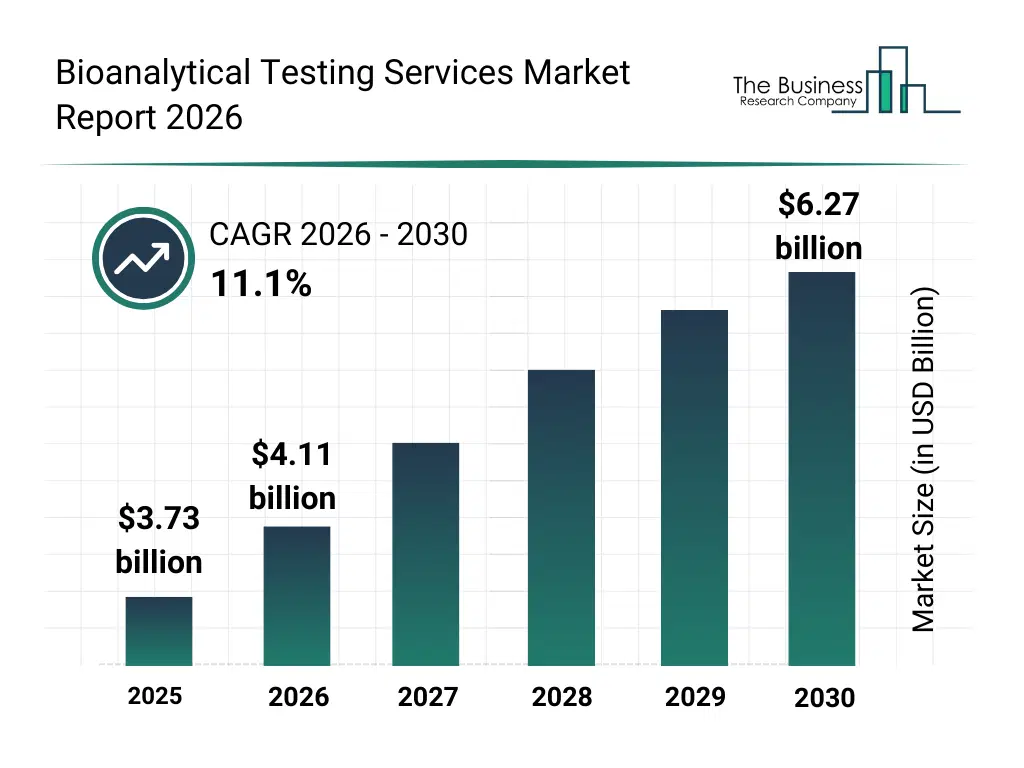
What Is The Bioanalytical Testing Services Market Size and Share 2026?
The bioanalytical testing services market size has grown rapidly in recent years. It will grow from $3.73 billion in 2025 to $4.11 billion in 2026 at a compound annual growth rate (CAGR) of 10.4%. The growth in the historic period can be attributed to growth in pharmaceutical r&d activities, rising demand for clinical drug testing support, increasing reliance on contract testing organizations, expansion of regulatory compliance needs, early development of analytical technology infrastructure.What Is The Bioanalytical Testing Services Market Growth Forecast?
The bioanalytical testing services market size is expected to see rapid growth in the next few years. It will grow to $6.27 billion in 2030 at a compound annual growth rate (CAGR) of 11.1%. The growth in the forecast period can be attributed to rising biologics and biosimilar development, increasing clinical trial intensity worldwide, growing focus on precise pharmacokinetic assessment, expansion of personalized medicine initiatives, stronger emphasis on safe therapeutic validation. Major trends in the forecast period include increasing demand for drug assessment accuracy, rising focus on reliable quantification of analytes, growing expansion of outsourced bioanalytical testing, strengthening emphasis on regulatory standard compliance, increasing adoption of specialized biological sample analysis.Global Bioanalytical Testing Services Market Segmentation
1) By Test Type: ADME (Absorption, Distribution, Metabolism, And Excretion), Pharmacokinetic, Pharmacodynamic, Bioequivalence, Bioavailability 2) By Molecule Type: Small Molecules, Large Molecules 3) By End User: Pharmaceutical And Biopharmaceutical Companies, Contract Development And Manufacturing Organizations, Contract Research Organizations Subsegments: 1) By ADME (Absorption, Distribution, Metabolism, and Excretion): Absorption Studies, Distribution Studies, Metabolism Studies, Excretion Studies 2) By Pharmacokinetic: Cmax (Maximum Concentration), Tmax (Time To Reach Cmax), AUC (Area Under the Curve), Half-Life Studies 3) By Pharmacodynamic: Efficacy Testing, Safety Testing, Dose-Response Relationship Studies 4) By Bioequivalence: In Vivo Bioequivalence Studies, In Vitro Bioequivalence Studies 5) By Bioavailability: Absolute Bioavailability Studies, Relative Bioavailability StudiesWhat Is The Driver Of The Bioanalytical Testing Services Market?
The rising government initiative to control infectious disease outbreaks is significantly contributing to the growth of the bioanalytical testing services market. The outbreak of the COVID-19 pandemic has aided in the growth in demand for bioanalytical testing services. Countries worldwide have been taking initiatives and increasing spending to prevent the spread and impact of the diseases. For instance, in March 2023, according to a report published by the Congressional Research Service, a US-based public policy research institute. The funding from National Institutes of Health (NIH) for Allergy/Infectious Diseases (NIAID) has increased from $6, 323 million to $6, 562 in 2023. Therefore, the rising government initiatives to control infectious disease outbreaks is driving the bioanalytical testing services industry.Key Players In The Global Bioanalytical Testing Services Market
Major companies operating in the bioanalytical testing services market report are CD BioSciences, Thermo Fisher Scientific Inc., Merck KGaA, Laboratory Corporation of America Holdings (LabCorp), Eurofins Scientific SE, ICON plc, PPD Inc., SGS SA, WuXi AppTec, Syneos Health Inc., Charles River Laboratories Inc., Intertek Group, Covance Inc., PAREXEL International Corporation, Medpace Holdings Inc., Almac Group, Pace Analytical Services LLC, Altasciences, BioAgilytix Labs, Frontage Laboratories Inc., Toxikon Inc., Absorption Systems LLC, Lotus Labs Pvt. Ltd., Bioneeds India Private Limited, Vipragen Biosciences Pvt. Ltd., Source BioScienceGlobal Bioanalytical Testing Services Market Trends and Insights
Major companies operating in the bioanalytical testing services market are focused on introducing advanced solutions, such as reverse transcription PCR (RT-PCR) tests, to sustain their position in the market. The reverse transcription PCR (RT-PCR) test is a laboratory technique that involves the conversion of RNA into complementary DNA (cDNA) using an enzyme called reverse transcriptase. For instance, in June 2023, Eurofins Scientific SE, a France-based testing laboratory company, launched the NovaType SARS-CoV-2 RT-PCR assay. The NovaType SARS-CoV-2 RT-PCR assay is clinically validated and has a short turnaround time, which is suitable for re-testing millions of positive samples to detect if the virus is a specific variant. It is a real-time reverse transcription PCR (RT-PCR) test designed for the qualitative detection of SARS-CoV-2, the virus that causes COVID-19, and the subsequent identification of specific variants, such as B.1.1.7 and B.1.351.What Are Latest Mergers And Acquisitions In The Bioanalytical Testing Services Market?
In August 2023, Scantox AB, a Denmark-based provider of bioanalytical and toxicology services, acquired Q&Q Labs AB for an undisclosed amount. With this acquisition, Scantox AB aims to strengthen its bioanalytical services offering and expand its capabilities in supporting drug development and safety assessment. Q&Q Labs AB is a Sweden-based provider of bioanalytical testing services, including pharmacokinetic, immunogenicity, and biomarker analysis for pharmaceutical and biotech companies.Regional Outlook
North America was the largest region in the bioanalytical testing services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Bioanalytical Testing Services Market?
The bioanalytical testing services market includes revenues earned by entities by providing services such as method development, high throughput sample analysis, method validation, and pharmacokinetic and toxicokinetic support. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Bioanalytical Testing Services Market Report 2026?
The bioanalytical testing services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the bioanalytical testing services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Bioanalytical Testing Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.11 billion |
| Revenue Forecast In 2035 | $6.27 billion |
| Growth Rate | CAGR of 10.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Molecule Type, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | CD BioSciences, Thermo Fisher Scientific Inc., Merck KGaA, Laboratory Corporation of America Holdings (LabCorp), Eurofins Scientific SE, ICON plc, PPD Inc., SGS SA, WuXi AppTec, Syneos Health Inc., Charles River Laboratories Inc., Intertek Group, Covance Inc., PAREXEL International Corporation, Medpace Holdings Inc., Almac Group, Pace Analytical Services LLC, Altasciences, BioAgilytix Labs, Frontage Laboratories Inc., Toxikon Inc., Absorption Systems LLC, Lotus Labs Pvt. Ltd., Bioneeds India Private Limited, Vipragen Biosciences Pvt. Ltd., Source BioScience |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
