
Biologic Hernia Repair Mesh Market Report 2026
Global Outlook – By Absorption (Partially Absorbable Hernia Repair Mesh, Fully Absorbable Hernia Repair Mesh), By Product Type (Porcine, Bovine, Human Dermis, Other Product Types), By Distribution Channel (Direct Sales, Distributors, Online Sales, Pharmacies), By Application (Inguinal Hernia Repair, Ventral Hernia Repair, Umbilical Hernia Repair, Other Hernia Repairs), By End-User (Hospitals, Ambulatory Surgical Centers, Clinics, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Biologic Hernia Repair Mesh Market Overview
• Biologic Hernia Repair Mesh market size has reached to $1.85 billion in 2025 • Expected to grow to $2.63 billion in 2030 at a compound annual growth rate (CAGR) of 7.2% • Growth Driver: Growing Hernia Prevalence Fuels The Market Expansion Due To Rising Obesity And Advancements In Biologic Repair Solutions • Market Trend: Advancing Hernia Repair With Reinforced Biologic Mesh For Improved Outcomes • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Biologic Hernia Repair Mesh Market?
Biologic hernia repair mesh is a type of implant made from processed human or animal tissues. It is used to strengthen and support weakened areas during hernia repair surgeries. It is gradually absorbed by the body and promotes natural tissue regeneration. This makes it suitable for patients with a higher risk of infection or complex repairs. The main types of biologic hernia repair mesh are partially absorbable hernia repair mesh and fully absorbable hernia repair mesh. Partially absorbable mesh is made from a mix of materials that are both absorbable and non-absorbable. The product types commonly used include porcine, bovine, human dermis, and others. These meshes are distributed through various channels such as direct sales, distributors, online platforms, and pharmacies. They are applied in procedures such as inguinal, ventral, umbilical, and other hernia repairs, and catering to end users are hospitals, ambulatory surgical centers, clinics, and others.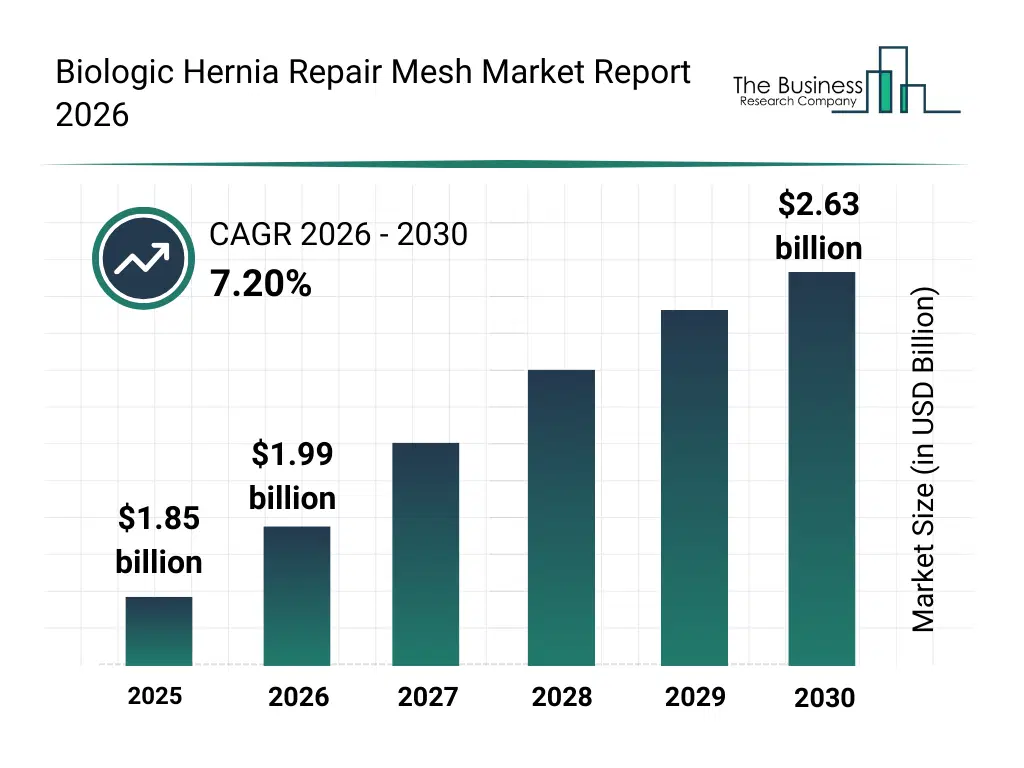
What Is The Biologic Hernia Repair Mesh Market Size and Share 2026?
The biologic hernia repair mesh market size has grown strongly in recent years. It will grow from $1.85 billion in 2025 to $1.99 billion in 2026 at a compound annual growth rate (CAGR) of 7.6%. The growth in the historic period can be attributed to increasing incidence of hernia disorders, limitations of synthetic mesh in infected cases, growing surgical procedure volumes, rising hospital-based hernia repairs, early adoption of tissue-based implants.What Is The Biologic Hernia Repair Mesh Market Growth Forecast?
The biologic hernia repair mesh market size is expected to see strong growth in the next few years. It will grow to $2.63 billion in 2030 at a compound annual growth rate (CAGR) of 7.2%. The growth in the forecast period can be attributed to advancements in biologic tissue processing technologies, growing demand for patient-specific repair solutions, rising awareness of post-surgical complications, expansion of ambulatory surgical centers, increasing surgeon preference for regenerative materials. Major trends in the forecast period include rising preference for biologic mesh in high-risk hernia repairs, increasing adoption of absorbable and regenerative mesh solutions, growing use in complex and contaminated surgical cases, expansion of minimally invasive and laparoscopic hernia procedures, higher clinical focus on reduced infection and recurrence rates.Global Biologic Hernia Repair Mesh Market Segmentation
1) By Absorption: Partially Absorbable Hernia Repair Mesh, Fully Absorbable Hernia Repair Mesh 2) By Product Type: Porcine, Bovine, Human Dermis, Other Product Types 3) By Distribution Channel: Direct Sales, Distributors, Online Sales, Pharmacies 4) By Application: Inguinal Hernia Repair, Ventral Hernia Repair, Umbilical Hernia Repair, Other Hernia Repairs 5) By End-User: Hospitals, Ambulatory Surgical Centers, Clinics, Other End Users Subsegments: 1) By Partially Absorbable Hernia Repair Mesh: Composite Mesh, Reinforced Biologic Mesh 2) By Fully Absorbable Hernia Repair Mesh: Extracellular Matrix (ECM) Mesh, Acellular Dermal Matrix (ADM) MeshWhat Is The Driver Of The Biologic Hernia Repair Mesh Market?
The increasing incidence of hernia cases is expected to propel the growth of the biologic hernia repair mesh market going forward. Hernia cases refer to the number of instances involving the abnormal protrusion of an internal organ or tissue through a weakened area in the muscle or connective tissue. The rise in hernia cases is attributed to growing obesity rates, as excess body weight exerts increasing pressure on abdominal muscles, thereby elevating the risk of hernia formation. Biologic hernia repair mesh aids in treating hernia cases by offering a scaffold composed of natural tissue that facilitates healing, integrates with the patient’s body, and minimizes the likelihood of rejection or persistent inflammation. For instance, in November 2024, according to the British Hernia Society, a UK-based national professional organization representing hernia surgeons in the United Kingdom and Ireland, the newly launched British Hernia Society Registry recorded registrations from over 100 surgeons at its debut conference. By January 2025, the registry had logged 400 cases contributed by 30 surgeons across 31 hospitals, reflecting rapid early uptake and highlighting the continued clinical burden of hernia cases. Therefore, the increasing incidence of hernia cases is driving the growth of the biologic hernia repair mesh industry.Key Players In The Global Biologic Hernia Repair Mesh Market
Major companies operating in the biologic hernia repair mesh market are Johnson And Johnson, Medtronic plc, DSM Biomedical, W L Gore And Associates Inc, Meril Life Sciences, Cook Medical LLC, Integra LifeSciences Corporation, MTF Biologics, LifeCell Corporation, AlloSource, TELA Bio Inc, Kerecis, Assut Europe S p A, Serag Wiessner GmbH And Co KG, Aroa Biosurgery Limited, ACell Inc, Tissue Regenix Group plc, RTI Surgical Inc, Beijing Datsing Bio Tech Co Ltd, Shanghai Pine And Power Biotech Co Ltd, Axogen Inc, XenoSure BiologicsGlobal Biologic Hernia Repair Mesh Market Trends and Insights
Major companies operating in the biologic hernia repair mesh market are focusing on developing innovative solutions, such as reinforced biologic mesh, to enhance strength, flexibility, and integration with host tissue. Reinforced biologic mesh is a type of surgical mesh that combines biologic tissue with synthetic fibers to enhance durability, support tissue regeneration, and minimize hernia recurrence. For instance, in April 2024, TELA Bio Inc., a US-based medical technology company, introduced OviTex inguinal hernia repair (IHR), a reinforced biologic mesh option for inguinal hernia repair. It is a reinforced tissue matrix specifically designed for laparoscopic and robotic inguinal hernia procedures, leveraging a combination of biologic ovine extracellular matrix (ECM) and minimal synthetic polymer to deliver strength, facilitate rapid tissue integration, and reduce the risk of complications. Its advantages include lower recurrence rates, accelerated healing, and higher patient satisfaction, effectively overcoming the drawbacks associated with conventional synthetic meshes.What Are Latest Mergers And Acquisitions In The Biologic Hernia Repair Mesh Market?
In February 2024, RTI Surgical Inc., a US-based contract development and manufacturing organization (CDMO), acquired Cook Biotech Inc. for an undisclosed amount. Through this acquisition, RTI Surgical Inc. seeks to establish itself as the only regenerative medicine CDMO capable of providing both allograft and xenograft biomaterials at scale, enhancing its leadership in soft tissue and broadening its market presence. Cook Biotech Inc. is a US-based regenerative medicine company that manufactures a biologic hernia repair mesh.Regional Outlook
North America was the largest region in the biologic hernia repair mesh market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Biologic Hernia Repair Mesh Market?
The biologic hernia repair mesh market consists of sales of strattice reconstructive tissue matrix, surgimend, and ovitex. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Biologic Hernia Repair Mesh Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.99 billion |
| Revenue Forecast In 2035 | $2.63 billion |
| Growth Rate | CAGR of 7.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Absorption, Product Type, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson And Johnson, Medtronic plc, DSM Biomedical, W L Gore And Associates Inc, Meril Life Sciences, Cook Medical LLC, Integra LifeSciences Corporation, MTF Biologics, LifeCell Corporation, AlloSource, TELA Bio Inc, Kerecis, Assut Europe S p A, Serag Wiessner GmbH And Co KG, Aroa Biosurgery Limited, ACell Inc, Tissue Regenix Group plc, RTI Surgical Inc, Beijing Datsing Bio Tech Co Ltd, Shanghai Pine And Power Biotech Co Ltd, Axogen Inc, XenoSure Biologics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
