
Biological Safety Testing Products And Services Market Report 2026
Global Outlook – By Product (Reagents And Kits, Instruments, Services), By Test Type (Endotoxin Tests, Sterility Tests, Cell Line Authentication And Characterization Tests, Bioburden Tests, Adventitious Agent Detection Tests, Residual Host Contamination Detection Tests, Other Test Types), By Application (Vaccines And Therapeutics, Blood And Blood-Based Products, Gene Therapy, Tissue And Tissue-based Products, Stem Cell) - Market Size, Trends, And Global Forecast 2026-2035
Biological Safety Testing Products And Services Market Overview
• Biological Safety Testing Products And Services market size has reached to $6.12 billion in 2025 • Expected to grow to $11.34 billion in 2030 at a compound annual growth rate (CAGR) of 13.1% • Growth Driver: Impact Of Growing Gene Therapy Demand On Biological Safety Testing Market • Market Trend: Innovative Advancements In Biological Safety Testing Products And Services • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Biological Safety Testing Products And Services Market?
Biological safety testing products and services refer to equipment and methods used to confirm that biological products are safe and uncontaminated by harmful agents, ensuring their safety and effectiveness for medical use. Its primary aim is to detect and prevent contamination by bacteria, viruses, fungi, and other pathogens. The main products and services in biological safety testing products and services are reagents and kits, instruments, and services. Reagents and kits are various chemicals, enzymes, antibodies, and other materials used to detect and measure the presence of contaminants, pathogens, or impurities in biological products. The various test types include endotoxin tests, sterility tests, cell line authentication and characterization tests, bioburden tests, adventitious agent detection tests, residual host contamination detection tests, and others used for different applications such as vaccines and therapeutics, blood and blood-based products, gene therapy, tissue and tissue-based products, and stem cells.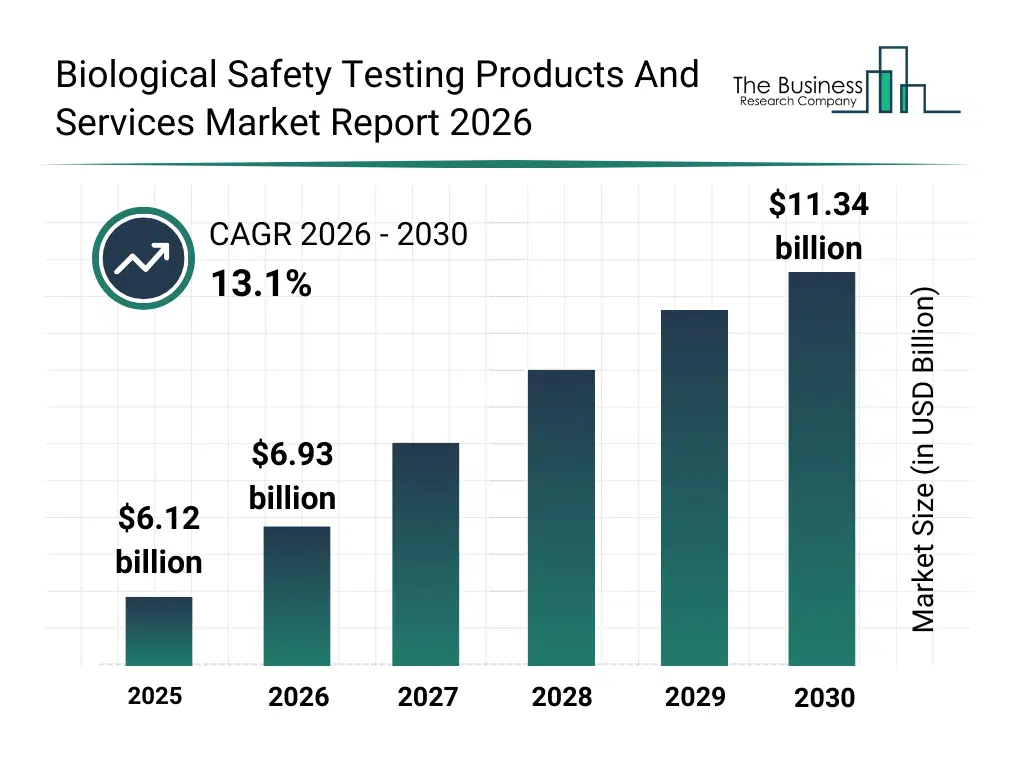
What Is The Biological Safety Testing Products And Services Market Size and Share 2026?
The biological safety testing products and services market size has grown rapidly in recent years. It will grow from $6.12 billion in 2025 to $6.93 billion in 2026 at a compound annual growth rate (CAGR) of 13.3%. The growth in the historic period can be attributed to rising production of vaccines and biologics, expansion of pharmaceutical manufacturing capacity, increasing regulatory scrutiny on product safety, wider adoption of cell and gene therapies, growth of clinical research activities.What Is The Biological Safety Testing Products And Services Market Growth Forecast?
The biological safety testing products and services market size is expected to see rapid growth in the next few years. It will grow to $11.34 billion in 2030 at a compound annual growth rate (CAGR) of 13.1%. The growth in the forecast period can be attributed to increasing demand for advanced biologics safety assays, rising investments in automated testing platforms, expansion of gene and cell therapy pipelines, growing need for faster release testing, increasing emphasis on contamination risk mitigation. Major trends in the forecast period include increasing adoption of rapid microbial detection methods, rising demand for automated sterility testing systems, growing use of pcr-based safety testing, expansion of outsourced biological testing services, enhanced focus on regulatory-compliant testing solutions.Global Biological Safety Testing Products And Services Market Segmentation
1) By Product: Reagents And Kits, Instruments, Services 2) By Test Type: Endotoxin Tests, Sterility Tests, Cell Line Authentication And Characterization Tests, Bioburden Tests, Adventitious Agent Detection Tests, Residual Host Contamination Detection Tests, Other Test Types 3) By Application: Vaccines And Therapeutics, Blood And Blood-Based Products, Gene Therapy, Tissue And Tissue-based Products, Stem Cell Subsegments: 1) By Reagents And Kits: Endotoxin Testing Kits, Mycoplasma Detection Kits, Sterility Testing Kits, Microbial Detection Reagents, Cell Culture Media Reagents 2) By Instruments: PCR Instruments, Microplate Readers, Bioluminescence Detection Systems, Automated Cell Culture Systems, ELISA Readers 3) By Services: Microbial Testing Services, Biocompatibility Testing Services, Endotoxin Testing Services, Environmental Monitoring Services, Consulting And Validation ServicesWhat Is The Driver Of The Biological Safety Testing Products And Services Market?
The growing demand for gene therapies is expected to propel the growth of the biological safety testing products and services market going forward. Gene therapy is a medical approach that uses genes to treat or prevent disease by replacing, modifying, or supplementing faulty genes. The increased investment and research related to gene mutation and supportive regulatory environments further accelerate innovation and expand gene therapeutic applications. Biological safety testing products and services in gene therapy ensure viral vectors and genetic materials are contaminant-free and safe to use. For instance, in January 2024, according to a published document by the American Society of Gene & Cell Therapy, a US-based non-profit medical and scientific organization, in quarter four, 2023, the number of gene therapies in Phase III clinical trials grew by 10%, the first quarterly increase of that type since quarter three, 2022. Therefore, the growing demand for gene therapies drives the market of biological safety testing products and services.Key Players In The Global Biological Safety Testing Products And Services Market
Major companies operating in the biological safety testing products and services market are Merck KGaA, Thermo Fisher Scientific Inc., Quest Diagnostics Incorporated, SGS Société Générale de Surveillance SA, Eurofins Scientific, Agilent Technologies Inc., Lonza Group AG, WuXi AppTec Co. Ltd., Charles River Laboratories Inc., BioMérieux SA, Almac Group, Samsung Biologics Co. Ltd., Maravai LifeSciences Holdings Inc., Genscript Biotech Corp, Wako Pure Chemical Industries Ltd., Associates of Cape Cod Inc., InvivoGen S.A.S., Microcoat Biotechnologie GmbH, BSL BIOSERVICE Scientific Laboratories Munich GmbH, Redberry, PathoQuestGlobal Biological Safety Testing Products And Services Market Trends and Insights
Major companies operating in the biological safety testing products and services market are developing rapid animal-free bacterial endotoxin testing to enhance patient safety and expedite drug development. This innovative approach aims to reduce testing time and improve reliability without the use of animal-based products. For instance, in January 2024, Charles River Laboratories International, a US-based pharmaceutical and biotechnology company, introduced the Endosafe Trillium cartridge, which employs recombinant technology to detect and quantify endotoxins without using animal-derived materials. It is the first rapid animal-free bacterial endotoxin test that features a faster turnaround time and eliminates the need for animal-derived reagents, thus enhancing sustainability and ethical testing practices. This product aims to provide rapid, accurate results while reducing reliance on the traditional Limulus amebocyte lysate (LAL) derived from horseshoe crabs.What Are Latest Mergers And Acquisitions In The Biological Safety Testing Products And Services Market?
In September 2023, Technical Safety Services, a US-based biotechnology company, acquired Controlled Environment Management LLC for an undisclosed amount. This acquisition aims to enhance TSS's support for pharmaceutical, biotechnology, and healthcare clients by expanding its geographic presence and service capabilities. Controlled Environment Management LLC is a US-based company that provides services essential for biological safety testing, such as testing, validation, certification, and calibration of controlled environments.Regional Outlook
North America was the largest region in the biological safety testing products and services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Biological Safety Testing Products And Services Market?
The biological safety testing product and services market consists of revenues earned by entities by providing services such as viral clearance studies, environmental monitoring, toxicology testing, and batch release testing. The market value includes the value of related goods sold by the service provider or included within the service offering. The biological safety testing market also includes sales of polymerase chain reaction (PCR) machines, biosafety cabinets, autoclaves, and centrifuges. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Biological Safety Testing Products And Services Market Report 2026?
The biological safety testing products and services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the biological safety testing products and services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Biological Safety Testing Products And Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.93 billion |
| Revenue Forecast In 2035 | $11.34 billion |
| Growth Rate | CAGR of 13.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Test Type, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Merck KGaA, Thermo Fisher Scientific Inc., Quest Diagnostics Incorporated, SGS Société Générale de Surveillance SA, Eurofins Scientific, Agilent Technologies Inc., Lonza Group AG, WuXi AppTec Co. Ltd., Charles River Laboratories Inc., BioMérieux SA, Almac Group, Samsung Biologics Co. Ltd., Maravai LifeSciences Holdings Inc., Genscript Biotech Corp, Wako Pure Chemical Industries Ltd., Associates of Cape Cod Inc., InvivoGen S.A.S., Microcoat Biotechnologie GmbH, BSL BIOSERVICE Scientific Laboratories Munich GmbH, Redberry, PathoQuest |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
