
Biomarker Clinical Phase Outsourcing Services Market Report 2026
Global Outlook – By Type (Surrogate Endpoints, Predictive Biomarker, Prognostic Biomarker, Safety Biomarker, Other Biomarker Types), By Service Type (Biomarker Validation, Biomarker Testing Services, Biomarker Sample Analysis), By Therapeutic Area (Oncology, Neurology, Cardiology, Autoimmune Diseases, Other Therapeutic Areas), By End-User (Pharmaceutical Companies, Biotechnology Companies) - Market Size, Trends, And Global Forecast 2026-2035
Biomarker Clinical Phase Outsourcing Services Market Overview
• Biomarker Clinical Phase Outsourcing Services market size has reached to $11.91 billion in 2025 • Expected to grow to $30.4 billion in 2030 at a compound annual growth rate (CAGR) of 20.1% • Growth Driver: Expanding Clinical Trial Landscape Fuels Growth Of Biomarker Clinical Phase Outsourcing Services Market • Market Trend: Advancements Drive Competition In Biomarker Clinical Phase Outsourcing Services • North America was the largest region in 2025.What Is Covered Under Biomarker Clinical Phase Outsourcing Services Market?
Biomarker clinical phase outsourcing services refer to the practice of contracting specialized firms or organizations to conduct various aspects of clinical trials related to biomarkers. Biomarkers are measurable indicators of biological states or conditions, often used in medical research and clinical trials to assess the effectiveness or safety of a drug or treatment. The main types of biomarker clinical phase outsourcing services are surrogate endpoints, predictive biomarkers, prognostic biomarkers, safety biomarkers, and others. Surrogate endpoints are biomarkers intended to substitute for a clinical endpoint and Outsourcing services utilize surrogate endpoints to streamline clinical trials by providing early indications of efficacy, reducing trial duration, and potentially accelerating regulatory approvals. Service types such as biomarker validation, biomarker testing services, and biomarker sample analysis are used for therapeutic areas that include oncology, neurology, cardiology, autoimmune diseases, and others. The end users are pharmaceutical companies, biotechnology companies, and academic and research institutions.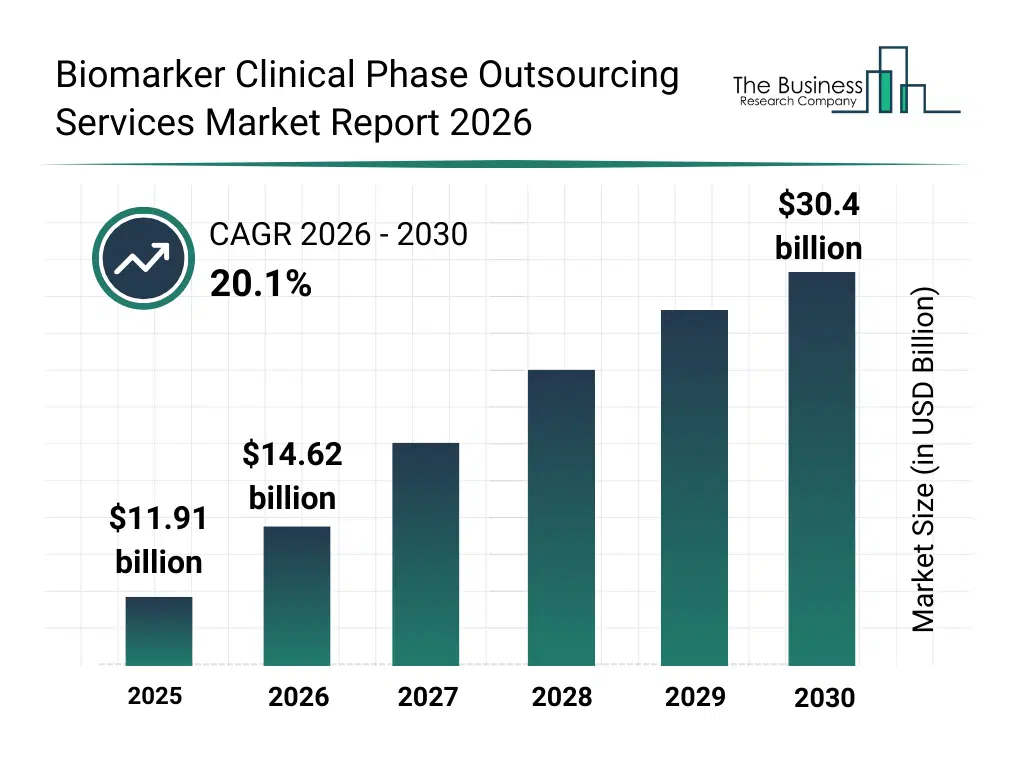
What Is The Biomarker Clinical Phase Outsourcing Services Market Size and Share 2026?
The biomarker clinical phase outsourcing services market size has grown exponentially in recent years. It will grow from $11.91 billion in 2025 to $14.62 billion in 2026 at a compound annual growth rate (CAGR) of 22.7%. The growth in the historic period can be attributed to increasing complexity of clinical trial designs, rising oncology clinical research activity, growth of biomarker-driven drug development, expansion of translational research programs, increasing collaboration with specialized service providers.What Is The Biomarker Clinical Phase Outsourcing Services Market Growth Forecast?
The biomarker clinical phase outsourcing services market size is expected to see exponential growth in the next few years. It will grow to $30.4 billion in 2030 at a compound annual growth rate (CAGR) of 20.1%. The growth in the forecast period can be attributed to increasing demand for personalized medicine trials, rising adoption of ai-driven biomarker analytics, expansion of multi-biomarker clinical studies, growing investments in precision oncology, increasing regulatory emphasis on biomarker validation. Major trends in the forecast period include increasing outsourcing of biomarker clinical trial activities, rising use of predictive and prognostic biomarkers, growing integration of omics-based biomarker analysis, expansion of companion diagnostics development, enhanced focus on precision medicine trials.Global Biomarker Clinical Phase Outsourcing Services Market Segmentation
1) By Type: Surrogate Endpoints, Predictive Biomarker, Prognostic Biomarker, Safety Biomarker, Other Biomarker Types 2) By Service Type: Biomarker Validation, Biomarker Testing Services, Biomarker Sample Analysis 3) By Therapeutic Area: Oncology, Neurology, Cardiology, Autoimmune Diseases, Other Therapeutic Areas 4) By End-User: Pharmaceutical Companies, Biotechnology Companies Subsegments: 1) By Surrogate Endpoints: Tumor Marker-Based Endpoints, Imaging-Based Endpoints, Genetic or Genomic Markers 2) By Predictive Biomarker: Genetic Predictive Biomarkers, Protein Predictive Biomarkers, Metabolomic Predictive Biomarkers 3) By Prognostic Biomarker: Cancer Prognostic Biomarkers, Cardiovascular Prognostic Biomarkers, Neurological Prognostic Biomarkers 4) By Safety Biomarker: Toxicity Biomarkers, Adverse Drug Reaction Biomarkers, Drug Interaction Biomarkers 5) By Other Biomarker Types: Inflammation Biomarkers, Infectious Disease Biomarkers, Autoimmune Disease BiomarkersWhat Is The Driver Of The Biomarker Clinical Phase Outsourcing Services Market?
The rise in clinical trials is expected to propel the growth of the biomarker clinical phase outsourcing services market going forward. Clinical trials are research studies conducted with human participants to evaluate the safety and effectiveness of new medical treatments, interventions, or diagnostic procedures. The surge in clinical trials is facilitated by advancements in medical research, rising prevalence of chronic diseases, increased funding from both governmental and private sources, and the growing demand for innovative treatments and therapies. Outsourcing providers often have specialized knowledge and experience in biomarker discovery, validation, and analysis. This expertise can enhance the quality and reliability of the biomarker data collected during the trial. For instance, in November 2025, according to the National Library of Medicine, a US-based biomedical library reported that ClinicalTrials.gov currently lists 558,242 studies with locations in all 51 states and in 224 countries and territories. Therefore, the rise in clinical trials will drive the growth in the biomarker clinical phase outsourcing services industry forward.Key Players In The Global Biomarker Clinical Phase Outsourcing Services Market
Major companies operating in the biomarker clinical phase outsourcing services market are Thermo Fisher Scientific Inc., Labcorp Drug Development Inc., ICON plc, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Syneos Health Inc., Charles River Laboratories International Inc., Parexel International Corporation, QIAGEN N.V., Medpace Inc., Evotec SE, Precision for Medicine Inc., BioAgilytix Labs LLC, Celerion Inc., Frontage Laboratories Inc., SomaLogic Inc., Metabolon Inc., Caprion Proteomics Inc., Intertek Group plc, Bioclinica Inc., GenScript Biotech Corporation, Selvita S.A.Global Biomarker Clinical Phase Outsourcing Services Market Trends and Insights
Major companies in the biomarker clinical phase outsourcing services are focusing on advancements with high-throughput flow cytometry and ELISpot testing to gain a competitive edge in the market. High-throughput flow cytometry and ELISpot testing are utilized for biomarker analysis, offering rapid and precise assessment of immune responses and cellular markers. For instance, in May 2024, KCAS Bio, a U.S.-based bioanalytical CRO offering GLP/GCP-compliant biomarker and immune-monitoring services, formed a global spectral flow cytometry alliance with Crux Biolabs, an Australian CRO specializing in ELISpot and flow cytometry assays. Through this partnership, KCAS Bio placed harmonized spectral cytometers at Crux Biolabs’ facilities, allowing clients to access high-throughput, multiparametric flow cytometry across the U.S., Europe, and Australia. This alliance strengthens KCAS’s biomarker development and immune-monitoring capabilities by providing rapid, precise assessment of cellular biomarkers in clinical trials, enabling clients to generate high-quality data that supports regulatory submissions and accelerates drug development.What Are Latest Mergers And Acquisitions In The Biomarker Clinical Phase Outsourcing Services Market?
In March 2023, SSI Strategy, a US-based a life sciences consulting services, partnered with Koneksa to enhance patient monitoring in clinical research. This collaboration aims to improve patient monitoring and streamline research processes, enabling pharmaceutical and biotech companies to gain deeper insights and accelerate the development of new therapies. Together, they focus on advancing the use of real-world and remote monitoring technologies to support more efficient and patient-centered clinical studies. Koneksa is a US-based health technology company specializing in digital biomarkers and clinical research.Regional Outlook
North America was the largest region in the biomarker clinical phase outsourcing services market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Biomarker Clinical Phase Outsourcing Services Market?
The biomarker clinical phase outsourcing services market includes revenues earned by entities by consulting services, diagnostic tests, clinical trial design, and biomarker sample analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Biomarker Clinical Phase Outsourcing Services Market Report 2026?
The biomarker clinical phase outsourcing services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the biomarker clinical phase outsourcing services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Biomarker Clinical Phase Outsourcing Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $14.62 billion |
| Revenue Forecast In 2035 | $30.4 billion |
| Growth Rate | CAGR of 22.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Service Type, Therapeutic Area, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Labcorp Drug Development Inc., ICON plc, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Syneos Health Inc., Charles River Laboratories International Inc., Parexel International Corporation, QIAGEN N.V., Medpace Inc., Evotec SE, Precision for Medicine Inc., BioAgilytix Labs LLC, Celerion Inc., Frontage Laboratories Inc., SomaLogic Inc., Metabolon Inc., Caprion Proteomics Inc., Intertek Group plc, Bioclinica Inc., GenScript Biotech Corporation, Selvita S.A. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
