
Bioprocessing Market 2026
By Type (Upstream Processing, Downstream Processing), By Application (Monoclonal Antibodies, Cell And Gene Therapy, Vaccines, Other Applications), By End User (Biopharmaceutical Companies, Contract Manufacturing Organizations, Academic Research Institutes, Other End Users), By Scale Of Operation (Commercial Scale, Preclinical, Clinical), By Product (Equipment, Consumables, Services), And By Region, Opportunities And Strategies – Global Forecast To 2035
Bioprocessing Market Definition
Bioprocessing refers to the use of living cells or their components (such as enzymes, bacteria, or yeast) to obtain desired products, particularly in the production of pharmaceuticals, biofuels, chemicals, and food ingredients. It is an essential part of biotechnology manufacturing, especially for complex biological therapies that cannot be synthesized chemically. Bioprocessing is primarily used in the biopharmaceutical industry to manufacture biologics such as monoclonal antibodies, vaccines, hormones, enzymes, and cell and gene therapies. These products are used across a wide range of medical treatments, including cancer, autoimmune disorders, infectious diseases, and rare genetic conditions. The bioprocessing market consists of sales, by entities (organizations, sole traders, or partnerships), of processes that use living cells, organisms, or their components (like enzymes) to manufacture desired products such as drugs, vaccines, and biologics. Bioprocessing encompasses methods like fermentation, cell cultivation, and purification to create pharmaceuticals, food products, biofuels, and industrial enzymes.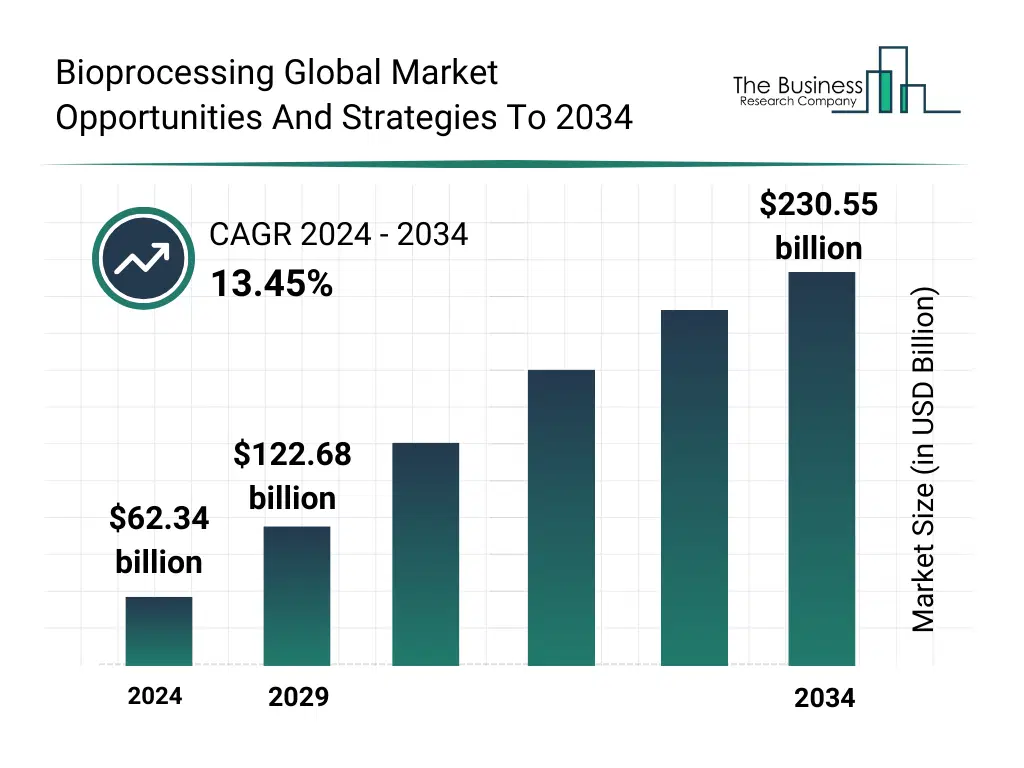
Bioprocessing Market Size
The global bioprocessing market reached a value of nearly $62,344.10 million in 2024, having grown at a compound annual growth rate (CAGR) of 12.93% since 2019. The market is expected to grow from $62,344.10 million in 2024 to $122,677.10 million in 2029 at a rate of 14.50%. The market is then expected to grow at a CAGR of 13.45% from 2029 and reach $230,550.77 million in 2034. Growth in the historic period resulted from government support in biotechnology, expansion of pharmaceutical and biotechnology industries, increasing investments in biotech R&D and growing demand for personalized medicine. Factors that negatively affected growth in the historic period were high energy and water consumption in manufacturing and skilled workforce shortage. Going forward, expansion of contract development and manufacturing organizations (CDMOs), rise in rare disease and orphan drug development, demand for cell and gene therapies and rising prevalence of chronic diseases will drive the growth. Factors that could hinder the growth of the bioprocessing market in the future include impact of trade war and tariff, long development and scale-up timelines and risk of contamination and process failures.Bioprocessing Market Drivers
The key drivers of the bioprocessing market include: Expansion Of Contract Development And Manufacturing Organizations (CDMOs) The growth of Contract Development and Manufacturing Organizations (CDMOs) is expected to be a major driver of the bioprocessing market during the forecast period. As global pharmaceutical companies continue to outsource development and manufacturing to specialized partners, the need for scalable, reliable, and regulatory-compliant bioprocessing solutions such as next-generation bioreactors, downstream purification systems, sterile manufacturing units, and platforms for scaling from clinical to commercial production is set to rise significantly. This trend is fueled by mounting cost pressures, efforts to diversify supply chains away from China, and government incentives aimed at boosting domestic CDMO capabilities. As a result, increased investment in bioprocessing infrastructure is anticipated, positioning CDMO expansion as a central force behind market growth. The expansion of contract development and manufacturing organizations (CDMOs) contribution during the forecast period in 2024 is 1.75%.Bioprocessing Market Restraints
The key restraints on the bioprocessing market include: Long Development And Scale-Up Timelines Lengthy development and scale-up timelines are anticipated to be a major restraint on the growth of the bioprocessing market during the forecast period. The development of advanced biologics—such as cell and gene therapies—requires navigating intricate, time-intensive regulatory pathways and establishing highly specialized manufacturing infrastructure. These therapies typically take 6 to 10 years from initial FDA interaction to full market approval, with prolonged phases involving clinical trials, process validation, and facility qualification. Further delays arise from challenges such as regulatory reviewer turnover, evolving compliance requirements, and administrative inefficiencies, all of which drive up costs and reduce operational agility. These extended timelines hinder manufacturers' ability to respond rapidly to market demands, slowing the adoption and implementation of innovative bioprocessing systems. Consequently, protracted development and scale-up processes are expected to significantly restrain market growth. Growth affected by long development and scale-up timelines during the forecast period in 2024 is -2.24%.Bioprocessing Market Trends
Major trends shaping the bioprocessing market include: Merging Spectral Cytometry With Real-Time Imaging Major companies in the bioprocessing market are increasingly combining spectral detection and imaging technologies in a single platform, pushing the frontier of what can be measured and visualized in live cell assays. These hybrid instruments aim to deliver deeper phenotypic insight, higher throughput, and streamlined workflows without compromising usability. For example, in May 2025, BD (Becton, Dickinson & Company), American medical technology company, launched the BD FACSDiscover A8 Cell Analyzer, touted as the world’s first cell analyzer to integrate BD SpectralFX technology with BD CellView real-time imaging. The SpectralFX module enables measurement of over 50 cellular parameters simultaneously, boosting both sensitivity and multiplexing capabilities. Meanwhile, the CellView imaging component captures spatial and morphological data, from intracellular localization to intercellular interactions, in real time. Alongside these optical advances, BD equipped the A8 with automation (walk-away operation), intuitive software for large dataset handling, and compatibility with its broader FACSDiscover ecosystem. With this breakthrough, BD seeks to lower the barrier for labs to access high-parameter cytometry enriched by imaging. The aim is to empower translational, immuno-oncology, and biomarker discovery research with tools that deliver greater depth and context per experiment, while maintaining throughput, ease-of-use, and integration across its instrument and reagent portfolio. Expansion of Automated Cell and Gene Therapy Manufacturing in Emerging Markets Companies in the market are focusing on expanding automated cell and gene therapy manufacturing capabilities in emerging markets to meet growing global demand and reduce production costs. Automation enhances process consistency, scalability, and regulatory compliance, which are critical for complex therapies. This expansion allows localized manufacturing, improving accessibility and reducing supply chain challenges. As a result, it drives increased adoption of advanced bioprocessing technologies tailored for high-efficiency, large-scale production. For example, in October 2024, Terumo Blood and Cell Technologies, a US-based medical technology company, expanding its offerings in Latin America to help bring automated manufacturing to cell and gene therapy companies in Brazil, Colombia and Mexico. Cell and gene therapies are a rapidly advancing field and hold the potential to treat diseases and transform lives. Affordability and the ability to scale these treatments in the manufacturing process are key factors that make automated technologies promising in helping cell and gene therapies touch more patients' lives. This is especially important in Latin America, where there is a heavy reliance on government healthcare systems.Opportunities And Recommendations In The Bioprocessing Market
Opportunities – The top opportunities in the bioprocessing market segmented by type will arise in the downstream processing segment, which will gain $36,199.80 million of global annual sales by 2029. The top opportunities in the bioprocessing market segmented by application will arise in the cell and gene therapy segment, which will gain $25,716.57 million of global annual sales by 2029. The top opportunities in the bioprocessing market segmented by end user will arise in the biopharmaceutical companies segment, which will gain $36,373.04 million of global annual sales by 2029. The top opportunities in the bioprocessing market segmented by scale of operation will arise in the commercial-scale segment, which will gain $45,485.25 million of global annual sales by 2029. The top opportunities in the bioprocessing market segmented by product will arise in the consumables segment, which will gain $39,833.89 million of global annual sales by 2029. The bioprocessing market size will gain the most in the USA at $23,492.72 million. Recommendations- To take advantage of the opportunities, The Business Research Company recommends the bioprocessing companies to focus on integrated spectral cytometry and real-time imaging to drive high-value research adoption, focus on automated cell and gene therapy expansion in emerging markets, focus on advancing TFF consumables for usability and sustainability gains, focus on enabling animal-free and in vitro pyrogen testing adoption, focus on advancing high-parameter spectral cytometry for drug discovery and cell analysis, focus on accelerating CHO cell line engineering with integrated high-throughput systems, focus on scaling animal-free endotoxin testing to strengthen quality and sustainability, focus on downstream processing to capture highest growth and quality demand, expand in emerging markets, continue to focus on developed markets, provide competitively priced offerings, continue to use B2B promotions, participate in trade shows and events, focus on cell and gene therapies to capture strongest growth and innovation demand and focus on contract manufacturing organizations to maximize growth and outsourcing demand.Bioprocessing Market Segmentation
The bioprocessing market is segmented by type, by application, by end user, by scale of operation and by product.By Type –
The bioprocessing market is segmented by type into:
- a) Upstream Processing
- b) Downstream Processing
By Application –
The bioprocessing market is segmented by application into:
- a) Monoclonal Antibodies
- b) Cell And Gene Therapy
- c) Vaccines
- d) Other Applications
By End User –
The bioprocessing market is segmented by end user into:
- a) Biopharmaceutical Companies
- b) Contract Manufacturing Organizations
- c) Academic Research Institutes
- d) Other End Users
By Scale Of Operation –
The bioprocessing market is segmented by scale of operation into:
- a) Commercial Scale
- b) Preclinical
- c) clinical
By Product –
The bioprocessing market is segmented by product into:
- a) Equipment
- b) Consumables
- c) Services
By Geography - The bioprocessing market is segmented by geography into:
- • China
- • India
- • Japan
- • Australia
- • Indonesia
- • South Korea
- • USA
- • Canada
- • Brazil
- • France
- • Germany
- • UK
- • Italy
- • Spain
- • Russia
-
o Asia Pacific
o Africa
