
Cardiopulmonary Bypass Accessory Equipment Market Report 2026
Global Outlook – By Product Type (Single Roller Pump, Double Roller Pump), By Operation (Manually Operated, Electrically Operated, Battery Operated), By Distribution Channel (Direct Tender, Third-Party Distributor), By Application (Lung Transplant Operation, Cardiac Surgery, Other Applications), By End-Use (Hospitals, Surgical Centers, Other End-Uses) – Market Size, Trends, Strategies, and Forecast to 2035
Cardiopulmonary Bypass Accessory Equipment Market Overview
• Cardiopulmonary Bypass Accessory Equipment market size has reached to $16.59 billion in 2025 • Expected to grow to $23.1 billion in 2030 at a compound annual growth rate (CAGR) of 6.8% • Growth Driver: Rising Acute Respiratory Failure Rates Fuel The Cardiopulmonary Bypass Accessory Equipment Market Growth • Market Trend: Advanced Cardiopulmonary Bypass Systems Enhancing Surgical Safety And Efficiency • North America was the largest region in 2025.What Is Covered Under Cardiopulmonary Bypass Accessory Equipment Market?
Cardiopulmonary bypass (CPB) accessory equipment are devices and instruments used during cardiac surgery with CPB to keep blood circulation and oxygenation outside the body after the heart stops. These accessories regulate blood temperature and filter out microemboli, protecting the patient's systemic circulation. The main product types of cardiopulmonary bypass accessory equipment are single-roller pumps and double-roller pumps. A single-roller pump is a peristaltic pump that uses a single revolving roller to compress a flexible tube and move blood through the circuit. This system mimics the heart's natural pumping activity, ensuring a consistent flow of blood. They are operated by various operations, such as manually operated, electrically operated, and battery operated, and distributed by several distribution channels, including direct tender, third-party distributor, and retail sales. They perform various applications, such as lung transplant operations, acute respiratory failure treatment, cardiac surgery, and others, which were used by several end-users, including hospitals, ambulatory care centers, surgical centers, and others.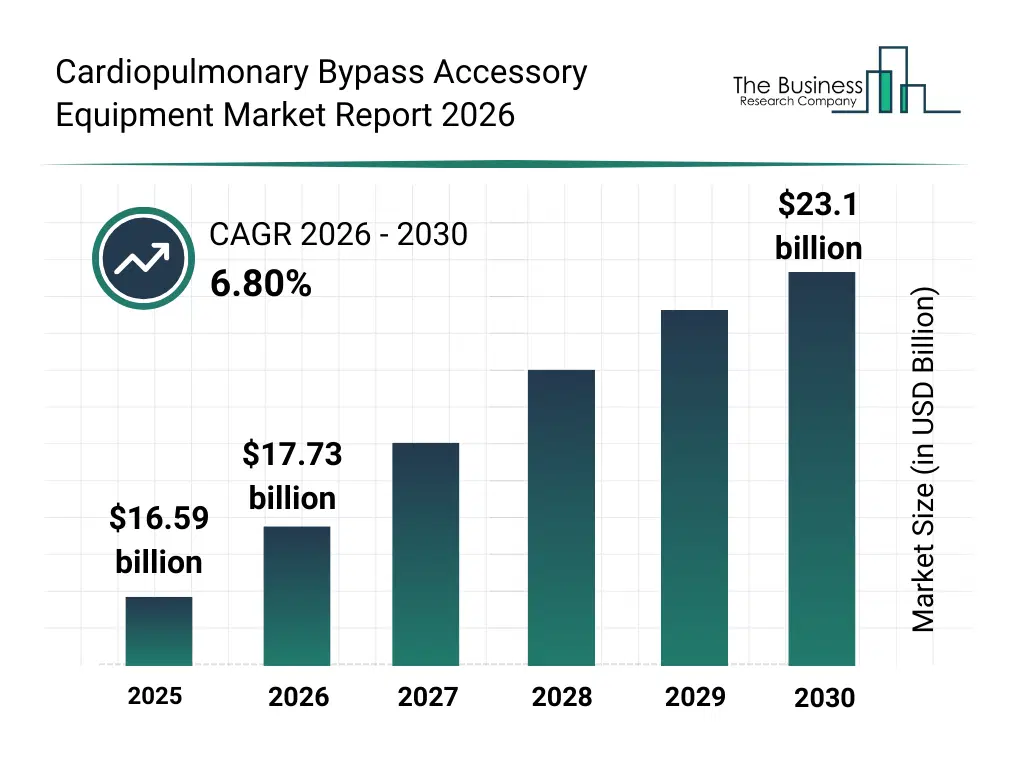
What Is The Cardiopulmonary Bypass Accessory Equipment Market Size and Share 2026?
The cardiopulmonary bypass accessory equipment market size has grown strongly in recent years. It will grow from $16.59 billion in 2025 to $17.73 billion in 2026 at a compound annual growth rate (CAGR) of 6.9%. The growth in the historic period can be attributed to growth in open heart surgical procedures, increasing prevalence of cardiovascular diseases, rising adoption of cardiopulmonary bypass techniques, expansion of hospital cardiac surgery infrastructure, improved survival rates in cardiac surgeries.What Is The Cardiopulmonary Bypass Accessory Equipment Market Growth Forecast?
The cardiopulmonary bypass accessory equipment market size is expected to see strong growth in the next few years. It will grow to $23.1 billion in 2030 at a compound annual growth rate (CAGR) of 6.8%. The growth in the forecast period can be attributed to rising volume of complex cardiac and lung transplant surgeries, increasing demand for improved intraoperative patient safety, growth in specialized cardiac surgical centers, expanding access to advanced cardiac care in emerging markets, rising investment in surgical quality improvement. Major trends in the forecast period include rising demand for advanced blood temperature management accessories, increasing focus on microemboli filtration and patient safety, growing adoption of disposable CPB accessory equipment, expansion of CPB usage in complex cardiac surgeries, increasing standardization of CPB accessories in surgical protocols.Global Cardiopulmonary Bypass Accessory Equipment Market Segmentation
1) By Product Type: Single Roller Pump, Double Roller Pump 2) By Operation: Manually Operated, Electrically Operated, Battery Operated 3) By Distribution Channel: Direct Tender, Third-Party Distributor 4) By Application: Lung Transplant Operation, Cardiac Surgery, Other Applications 5) By End-Use: Hospitals, Surgical Centers, Other End-Uses Subsegments: 1) By Single Roller Pump: Disposable Single Roller Pump, Reusable Single Roller Pump 2) By Double Roller Pump: Disposable Double Roller Pump, Reusable Double Roller PumpWhat Is The Driver Of The Cardiopulmonary Bypass Accessory Equipment Market?
The growing prevalence of acute respiratory failure is expected to propel the growth of the cardiopulmonary bypass accessory equipment market. Acute respiratory failure occurs when the respiratory system fails in one or both of its gas exchange activities, such as oxygenation and carbon dioxide removal. Acute respiratory failure is rising due to several factors, including the aging population, infectious diseases, and medical procedures. Cardiopulmonary bypass equipment responds to the demand for advanced respiratory support by providing comprehensive oxygenation and ventilation management in ICUs and hospitals, effectively maintaining complex situations of respiratory distress. For instance, in April 2024, according to the Australian Bureau of Statistics, an Australian-based government agency, there were 361 deaths due to respiratory syncytial virus (RSV) in 2023, up from 241 in 2022. Therefore, the growing prevalence of acute respiratory failure is driving the growth of the cardiopulmonary bypass accessory equipment industry.Key Players In The Global Cardiopulmonary Bypass Accessory Equipment Market
Major companies operating in the cardiopulmonary bypass accessory equipment market are Medtronic plc, Getinge AB, LivaNova PLC, Terumo Corporation, Edwards Lifesciences Corporation, Braile Biomédica, EUROSETS S.r.l., MicroPort Scientific Corporation, Nipro Corporation, Xenios AG, Tianjin Medical, CalMed Technology Inc., Avalon Laboratories LLC, Boston Scientific Corporation, ALung Technologies Inc., Kewei (Guangzhou) Medical Instrument Co Ltd, SynCardia Systems LLC, Hemovent GmbH, Elite LifeCare, Technowood International Pte LtdGlobal Cardiopulmonary Bypass Accessory Equipment Market Trends and Insights
Major companies operating in the cardiopulmonary bypass accessory equipment market are developing advanced perfusion systems to ensure safe and effective cardiopulmonary bypass procedures through advanced features and better clinical workflows. The perfusion system comprises a next-generation heart-lung machine (HLM) and precise sensing technology for data-driven decision-making for safe cardiopulmonary bypass (CPB) management. For instance, in March 2023, LiveNova PLC, a UK-based company that develops devices used for cardiac surgery, launched the Essenz Heart-Lung Machine (HLM) and Essenz Patient Monitor, which together form the Essenz Perfusion System. Essenz's heart-lung machine system, based on LivaNova's proven S5 HLM, enhances clinical workflows and patient care quality. The device has several elements that prevent air bubbles and other possible emboli from entering the patient's bloodstream, lowering the risk of stroke and other problems. It features intra-operative quality indicator thresholds and goal-directed perfusion (GDP) modules to provide personalized patient care strategies.What Are Latest Mergers And Acquisitions In The Cardiopulmonary Bypass Accessory Equipment Market?
In June 2024, Becton, Dickinson, and Company, a US-based medical technology company acquired Edwards Lifesciences' Critical Care product group (Critical Care) for $4.2 billion. This strategic acquisition aims to improve BD's smart connected care solutions by integrating advanced monitoring technologies and AI-driven clinical decision support tools. This acquisition will expand BD's monitoring capabilities, accelerate revenue growth, and allow Edwards Lifesciences to focus on structural heart innovations. Edwards Lifesciences is a US-based medical technology company involved in heart valve technologies and hemodynamic monitoring solutions.Regional Outlook
North America was the largest region in the cardiopulmonary bypass accessory equipment market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Cardiopulmonary Bypass Accessory Equipment Market?
The cardiopulmonary bypass accessory equipment market consists of sales of heat exchangers, oxygenators, perfusion monitors, flow meters, and pressure regulators. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Cardiopulmonary Bypass Accessory Equipment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $17.73 billion |
| Revenue Forecast In 2035 | $23.1 billion |
| Growth Rate | CAGR of 6.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Operation, Distribution Channel, Application, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic plc, Getinge AB, LivaNova PLC, Terumo Corporation, Edwards Lifesciences Corporation, Braile Biomédica, EUROSETS S.r.l., MicroPort Scientific Corporation, Nipro Corporation, Xenios AG, Tianjin Medical, CalMed Technology Inc., Avalon Laboratories LLC, Boston Scientific Corporation, ALung Technologies Inc., Kewei (Guangzhou) Medical Instrument Co Ltd, SynCardia Systems LLC, Hemovent GmbH, Elite LifeCare, Technowood International Pte Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
