
Cardiovascular Clinical Trials Market Report 2026
Global Outlook – By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design (Interventional, Observational, Expanded Access), By Indication (Acute Coronary Syndrome, Coronary Artery Disease, Ischemic Heart Disease, Pulmonary Arterial Hypertension, Stroke, Cardiac Arrhythmias, Heart Failure, Other Indications) - Market Size, Trends, And Global Forecast 2026-2035
Cardiovascular Clinical Trials Market Overview
• Cardiovascular Clinical Trials market size has reached to $5.72 billion in 2025 • Expected to grow to $7.7 billion in 2030 at a compound annual growth rate (CAGR) of 5.9% • Growth Driver: Rising Incidence Of Cardiovascular Diseases Fuels Growth Of The Cardiovascular Clinical Trials Market • Market Trend: Cereno Scientific Partners With CRO For Phase I Study • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Cardiovascular Clinical Trials Market?
Cardiovascular clinical trials refer to investigations that are aimed at new ways to prevent, diagnose, treat, and manage cardiovascular illnesses (CVDs), such as coronary artery disease, heart failure, arrhythmias, and hypertension. These studies are critical for expanding medical knowledge and improving patient care in the field of cardiology. The clinics focus on testing new medications, procedures, or devices related to heart health and stroke outcomes. The main phases of cardiovascular clinical trials are phase I, phase II, phase III, and phase IV. Phase I clinical trials are the initial stage of testing a new treatment on a small group of people to evaluate its safety and dosage. The various study designs included are interventional, observational, and expanded access, with indications such as acute coronary syndrome, coronary artery disease, ischemic heart disease, pulmonary arterial hypertension, stroke, cardiac arrhythmias, heart failure, and others.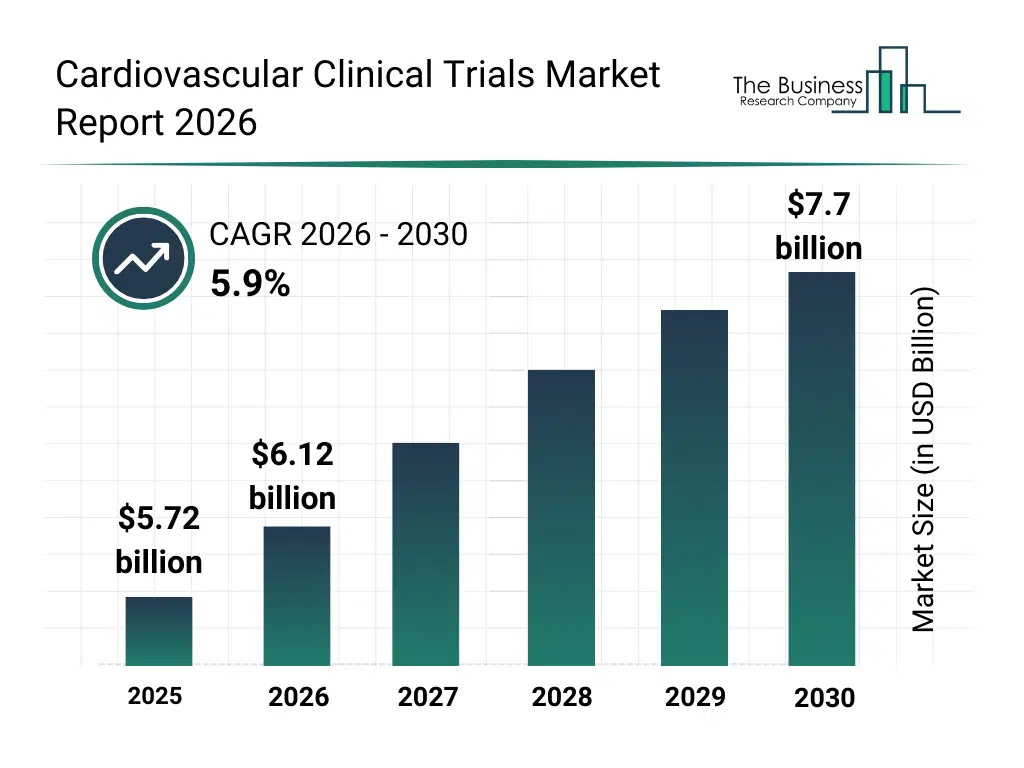
What Is The Cardiovascular Clinical Trials Market Size and Share 2026?
The cardiovascular clinical trials market size has grown strongly in recent years. It will grow from $5.72 billion in 2025 to $6.12 billion in 2026 at a compound annual growth rate (CAGR) of 7.0%. The growth in the historic period can be attributed to growing burden of cardiovascular diseases, expansion of global clinical research activity, increasing regulatory approvals for novel therapies, availability of established trial infrastructure, growth of multicenter studies.What Is The Cardiovascular Clinical Trials Market Growth Forecast?
The cardiovascular clinical trials market size is expected to see strong growth in the next few years. It will grow to $7.7 billion in 2030 at a compound annual growth rate (CAGR) of 5.9%. The growth in the forecast period can be attributed to increasing demand for faster trial execution, rising use of ai-driven trial analytics, expansion of remote patient engagement, growing focus on patient-centric trial designs, increasing investment in cardiovascular drug pipelines. Major trends in the forecast period include increasing adoption of decentralized trial models, rising use of digital patient monitoring tools, growing integration of biomarkers in study design, expansion of real-world evidence generation, enhanced focus on adaptive trial designs.Global Cardiovascular Clinical Trials Market Segmentation
1) By Phase: Phase I, Phase II, Phase III, Phase IV 2) By Study Design: Interventional, Observational, Expanded Access 3) By Indication: Acute Coronary Syndrome, Coronary Artery Disease, Ischemic Heart Disease, Pulmonary Arterial Hypertension, Stroke, Cardiac Arrhythmias, Heart Failure, Other Indications Subsegments: 1) By Phase I: First-in-Human Trials, Dose Escalation Studies, Safety and Tolerability Studies, Pharmacokinetics And Pharmacodynamics Studies 2) By Phase II: Efficacy Studies, Optimal Dosage And Administration Route Studies, Early Safety And Efficacy Trials, Biomarker Development Trials 3) By Phase III: Large-Scale Efficacy Trials, Randomized Controlled Trials (RCTs), Long-Term Safety And Efficacy Studies, Multicenter Trials 4) By Phase IV: Post-Marketing Surveillance, Long-Term Safety Studies, Real-World Evidence (RWE) Studies, Comparative Effectiveness ResearchWhat Is The Driver Of The Cardiovascular Clinical Trials Market?
The growing incidence of cardiovascular diseases is expected to drive the growth of the cardiovascular clinical trials market going forward. Cardiovascular disease refers to a group of conditions affecting the heart and blood vessels, including coronary artery disease, heart failure, and stroke. There is an increased incidence of cardiovascular diseases due to various factors, such as sedentary lifestyles, unhealthy diets, smoking, obesity, high blood pressure, diabetes, and genetic predispositions. Cardiovascular clinical trials help in discovering and validating new drugs that can more effectively manage risk factors such as hypertension, high cholesterol, and diabetes, which are significant contributors to CVDs. For instance, in September 2024, according to the data published by the Minnesota Department of Health, a US-based state health agency, approximately 30% of adults in Minnesota reported having high blood pressure in 2023, totaling nearly 1.4 million people, and in 2022, hypertensive diseases were the underlying or contributing cause of death for 14,225 residents, representing almost 28% of all deaths in the state. Therefore, the growing incidence of cardiovascular diseases is driving the growth of the cardiovascular clinical trials industry.Key Players In The Global Cardiovascular Clinical Trials Market
Major companies operating in the cardiovascular clinical trials market are Pfizer Inc., Johnson & Johnson, AstraZeneca PLC, Novartis AG, Eli Lilly and Company, Amgen Inc., Boehringer Ingelheim International GmbH, Merck & Co Inc., Sanofi, Baxter International Inc., IQVIA Holdings Inc., ICON plc, Syneos Health Inc., PPD Inc., Medpace Holdings Inc., Worldwide Clinical Trials, SGS SA, Charles River Laboratories International Inc., WuXi AppTec Co Ltd, Gilead Sciences Inc.Global Cardiovascular Clinical Trials Market Trends and Insights
Major companies operating in the cardiovascular clinical trials market are focused on adopting strategic partnerships, such as contract research organization (CRO) partnerships, to expand their distribution networks and reach a wider customer base. A CRO partnership plays a crucial role in the clinical research industry and involves collaborations between different types of companies to provide specialized services and support for various aspects of clinical trials and research. For instance, in September 2023, Cereno Scientific AB, a Sweden-based biopharmaceutical company, collaborated with Clinical Trial Consultants (CTC), a Sweden-based full-service CRO with a focus on clinical conduct, to conduct a Phase I study for CS014, a histone deacetylase inhibitor aimed at preventing arterial and venous thrombosis. CTC will also assist with Phase I preparation processes such as study protocol development and the clinical trial application process for the study, which will be conducted in Sweden. This first-in-human Phase I trial is scheduled to begin in the first half of 2024. This collaboration signifies a significant step in advancing research on cardiovascular health.What Are Latest Mergers And Acquisitions In The Cardiovascular Clinical Trials Market?
In February 2023, AstraZeneca, a US-based pharmaceutical company, acquired CinCor Pharma for $1.8 billion. The acquisition strengthens AstraZeneca's cardiorenal pipeline by adding baxdrostat (CIN-107), an aldosterone synthase inhibitor (ASI) for blood pressure reduction in treatment-resistant hypertension, to its portfolio. CinCor Pharma is a US-based clinical-stage biopharmaceutical company providing cardiovascular clinical trials.Regional Outlook
North America was the largest region in the cardiovascular clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Cardiovascular Clinical Trials Market?
The cardiovascular clinical trials market includes revenues earned by entities by providing services, such as protocol development, regulatory affairs, and patient recruitment and retention. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Cardiovascular Clinical Trials Market Report 2026?
The cardiovascular clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the cardiovascular clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Cardiovascular Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.12 billion |
| Revenue Forecast In 2035 | $7.7 billion |
| Growth Rate | CAGR of 7.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Phase, Study Design, Indication |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Johnson & Johnson, AstraZeneca PLC, Novartis AG, Eli Lilly and Company, Amgen Inc., Boehringer Ingelheim International GmbH, Merck & Co Inc., Sanofi, Baxter International Inc., IQVIA Holdings Inc., ICON plc, Syneos Health Inc., PPD Inc., Medpace Holdings Inc., Worldwide Clinical Trials, SGS SA, Charles River Laboratories International Inc., WuXi AppTec Co Ltd, Gilead Sciences Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
