
Catheter-Associated Urinary Tract Infections Treatment Market Report 2026
Global Outlook – By Treatment Type (Antibiotics, Non-Antibiotic Therapies), By Drug Type (Penicillin And Combinations, Quinolones, Cephalosporin, Aminoglycoside Antibiotics, Sulphonamides, Azoles And Amphotericin B, Tetracycline, Nitrofurans, Other Drug Types), By Indication Type (Asymptomatic Catheter-Associated Urinary Tract Infection, Symptomatic Catheter-Associated Urinary Tract Infection), By Distribution Channel (Hospital Pharmacies, Gynaecology And Urology Clinics, Drug Stores, Retail Pharmacies, Online Drug Stores), By End-Users (Clinic, Hospital, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Catheter-Associated Urinary Tract Infections Treatment Market Overview
• Catheter-Associated Urinary Tract Infections Treatment market size has reached to $1.26 billion in 2025 • Expected to grow to $1.67 billion in 2030 at a compound annual growth rate (CAGR) of 5.9% • Growth Driver: The Rise In Hospital-Acquired Infections Driving The Growth Of The Market Due To Increasing Antimicrobial Resistance And Patient Risk • Market Trend: Next-Generation Automated Bladder Irrigation Enhances Safety And Infection Prevention • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Catheter-Associated Urinary Tract Infections Treatment Market?
Catheter-associated urinary tract infection (CAUTI) treatment refers to the clinical management of urinary tract infections that develop in patients with an indwelling urinary catheter, focusing on eliminating the infection, relieving symptoms, and preventing complications. Treatment typically involves timely removal or replacement of the catheter when possible, appropriate antimicrobial therapy based on clinical assessment and urine culture results, and supportive care, while also addressing underlying risk factors to reduce recurrence. The main treatment types of catheter-associated urinary tract infections treatment include antibiotics and non-antibiotic therapies. Antibiotics refer to drugs used to prevent or treat infections caused by bacteria associated with catheter use. These treatments are available as penicillin and combinations, quinolones, cephalosporin, aminoglycoside antibiotics, sulphonamides, azoles and amphotericin B, tetracycline, nitrofurans, and other drug types and are indicated for asymptomatic catheter-associated urinary tract infection and symptomatic catheter-associated urinary tract infection. They are distributed through hospital pharmacies, gynaecology and urology clinics, drug stores, retail pharmacies, and online drug stores, and are used by several end-users such as clinics, hospitals, and other end-users.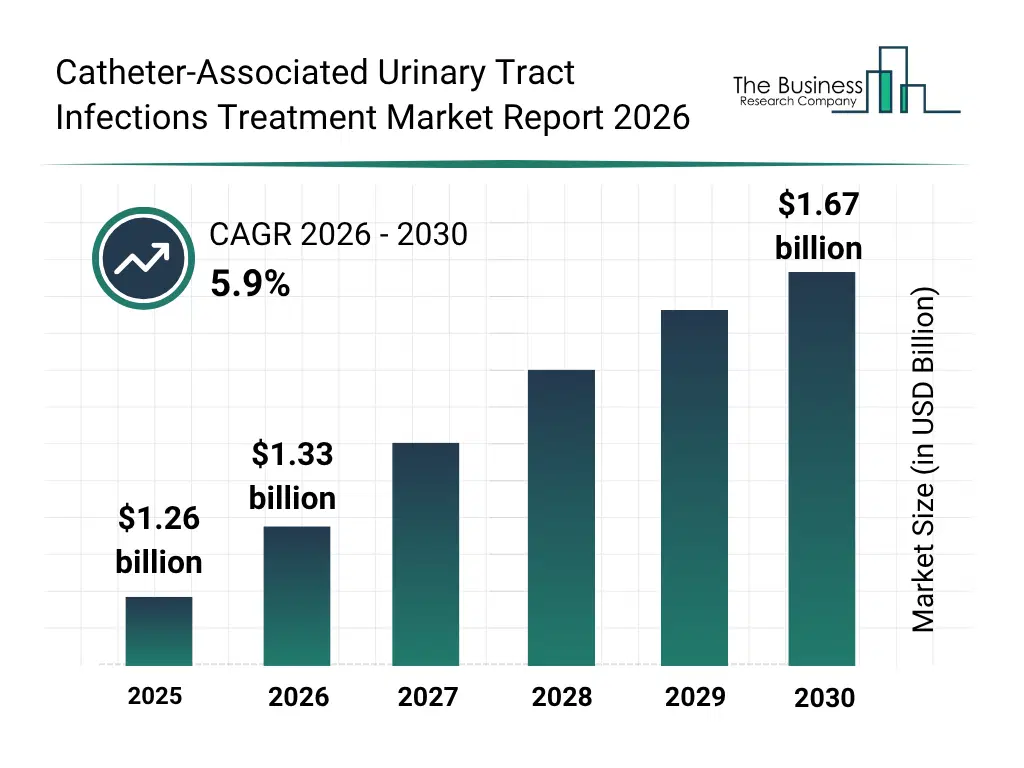
What Is The Catheter-Associated Urinary Tract Infections Treatment Market Size and Share 2026?
The catheter-associated urinary tract infections treatment market size has grown strongly in recent years. It will grow from $1.26 billion in 2025 to $1.33 billion in 2026 at a compound annual growth rate (CAGR) of 5.7%. The growth in the historic period can be attributed to increasing prevalence of catheter-associated urinary tract infections, rising hospital and clinic patient volume, adoption of antibiotics for treatment, growing healthcare infrastructure, increasing awareness of infection control measures.What Is The Catheter-Associated Urinary Tract Infections Treatment Market Growth Forecast?
The catheter-associated urinary tract infections treatment market size is expected to see strong growth in the next few years. It will grow to $1.67 billion in 2030 at a compound annual growth rate (CAGR) of 5.9%. The growth in the forecast period can be attributed to growing adoption of urinary catheter coatings, increasing use of probiotics and immunotherapy, rising demand for personalized treatment solutions, expansion of online drug delivery and telehealth services, increasing focus on preventive care and early intervention. Major trends in the forecast period include increasing adoption of non-antibiotic therapies, rising demand for catheter coatings and antimicrobial devices, growing focus on personalized cauti treatment plans, expansion of online and retail drug distribution channels, rising awareness on early detection and prevention of cauti.Global Catheter-Associated Urinary Tract Infections Treatment Market Segmentation
1) By Treatment Type: Antibiotics, Non-Antibiotic Therapies 2) By Drug Type: Penicillin And Combinations, Quinolones, Cephalosporin, Aminoglycoside Antibiotics, Sulphonamides, Azoles And Amphotericin B, Tetracycline, Nitrofurans, Other Drug Types 3) By Indication Type: Asymptomatic Catheter-Associated Urinary Tract Infection, Symptomatic Catheter-Associated Urinary Tract Infection 4) By Distribution Channel: Hospital Pharmacies, Gynaecology And Urology Clinics, Drug Stores, Retail Pharmacies, Online Drug Stores 5) By End-Users: Clinic, Hospital, Other End-Users Subsegments: 1) By Antibiotics: Penicillins, Cephalosporins, Fluoroquinolones, Aminoglycosides, Sulfonamides 2) By Non-Antibiotic Therapies: Urinary Catheter Coatings, Probiotics, Cranberry Extracts, Immunotherapy, Bladder Irrigation SolutionsWhat Are The Drivers Of The Catheter-Associated Urinary Tract Infections Treatment Market?
The rising hospital-acquired infection rates are expected to propel the growth of the catheter-associated urinary tract infections treatment market going forward. Hospital-acquired infection rates refer to the frequency of infections patients develop during hospital stays that were not present or incubating at the time of admission. The rising hospital-acquired infection rates are primarily due to antimicrobial resistance, which allows pathogens to survive standard treatments and spread more easily within healthcare settings. Catheter-associated urinary tract infections treatment helps reduce hospital-acquired infections by effectively preventing and managing infections related to indwelling catheters, thereby improving patient safety and lowering healthcare-associated complications. For instance, in May 2024, according to the Center for Infectious Disease Research and Policy (CIDRAP), a US-based disease research organization, the number of patients acquiring at least one healthcare-acquired infection in hospitals across the European Union and European Economic Area was nearly 4.3 million per year in 2022 and 2023. Therefore, rising hospital-acquired infection rates are driving the growth of the catheter-associated urinary tract infections treatment industry. The growing geriatric population is expected to propel the growth of the catheter-associated urinary tract infection treatment market going forward. The geriatric population refers to older adults, typically aged 65 years and above, who may require specialized healthcare and social support due to age-related physical, mental, and functional changes. The growing geriatric population is driven by longer life expectancy, as improvements in healthcare, nutrition, and living conditions enable people to live longer, resulting in a higher proportion of older individuals. Catheter-associated urinary tract infection treatment helps the geriatric population by reducing infection-related complications, improving urinary function, and enhancing overall quality of life. For instance, in July 2024, according to the UK Parliament's House of Commons Library, a UK-based government administration, the number of people aged 65 or older in the UK was 12.7 million in 2022, representing 19% of the total population, and is expected to increase to 22.1 million by 2072, accounting for 27% of the population. Therefore, a growing geriatric population is driving the growth of the catheter-associated urinary tract infection treatment market.Key Players In The Global Catheter-Associated Urinary Tract Infections Treatment Market
Major companies operating in the catheter-associated urinary tract infections treatment market are Cardinal Health Inc., Pfizer Incorporated, Bayer Aktiengesellschaft, AstraZeneca Public Limited Company, Medline Industries, Limited Partnership, Becton, Dickinson Company, B. Braun Societät mit beschränkter Haftung, Merck Company Inc, Coloplast A/S, Teleflex Incorporated, ConvaTec Group Public Limited Company, Hollister Incorporated, Vygon S.A., Wellspect Healthcare AB, Urocare Products Inc, Paul Hartmann Aktiengesellschaft, Bactiguard AB, NanoVibronix Inc, WellLead Medical Co Limited, Poiesis Medical LLC.Global Catheter-Associated Urinary Tract Infections Treatment Market Trends and Insights
Major companies operating in the catheter-associated urinary tract infection treatment market are focusing on developing advanced solutions such as automated bladder irrigation systems to enhance infection prevention and standardization of catheter care. Automated bladder irrigation systems are a technology that controls the inflow and outflow of irrigation fluid using sensors to deliver safe and consistent cleansing cycles while reducing contamination risks through a closed system design. For instance, in January 2026, UMED Inc., a South Korea-based healthcare company, showcased UroRinse Light globally at CES 2026, presenting the world’s first fully automated and standardized bladder irrigation system designed for long-term urinary catheter users. The system uses sensor-based automated control to manage irrigation flow and ensure reproducible treatment cycles while minimizing contamination through a closed structure. It enables use across hospitals, care facilities, and home settings, allowing patients and caregivers to perform safe and repeatable bladder irrigation outside traditional clinical environments. The device integrates with a mobile application and cloud database to record irrigation history and monitor patient status, with planned integration into electronic medical record (EMR) systems and remote care platforms to support continuous monitoring and proactive infection prevention.Regional Insights
North America was the largest region in the catheter-associated urinary tract infections treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Catheter-Associated Urinary Tract Infections Treatment Market?
The catheter-associated urinary tract infection treatment market consists of revenues earned by entities by providing services such as catheter maintenance and monitoring services, diagnostic testing and laboratory services, clinical consultation and treatment management, telehealth monitoring, and wound and urinary tract care management. The market value includes the value of related goods sold by the service provider or included within the service offering. The catheter-associated urinary tract infection treatment market also includes sales of catheter maintenance and closed drainage systems, catheter care kits and auxiliary devices, catheter care kits and auxiliary devices, hydrophilic‑coated catheters, and catheter securing devices and stabilizers. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Catheter-Associated Urinary Tract Infections Treatment Market Report 2026?
The catheter-associated urinary tract infections treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the catheter-associated urinary tract infections treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Catheter-Associated Urinary Tract Infections Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.33 billion |
| Revenue Forecast In 2035 | $1.67 billion |
| Growth Rate | CAGR of 5.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Type, Drug Type, Indication Type, Distribution Channel, End-Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Cardinal Health Inc., Pfizer Incorporated, Bayer Aktiengesellschaft, AstraZeneca Public Limited Company, Medline Industries, Limited Partnership, Becton, Dickinson Company, B. Braun Societät mit beschränkter Haftung, Merck Company Inc, Coloplast A/S, Teleflex Incorporated, ConvaTec Group Public Limited Company, Hollister Incorporated, Vygon S.A., Wellspect Healthcare AB, Urocare Products Inc, Paul Hartmann Aktiengesellschaft, Bactiguard AB, NanoVibronix Inc, WellLead Medical Co Limited, Poiesis Medical LLC. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
