
Cell And Gene Therapy Clinical Trial Services Market Report 2026
Global Outlook – By Service (Clinical Trial Design And Planning, Supply And Logistic Services, Regulatory Affairs And Compliance, Data Management And Biostatics, Site Management And Monitoring, Other Services), By Therapy Type (Gene Therapy, Cell Therapy, Gene Modified Cell Therapy), By Indication (Oncology, Hematology, Metabolic Disorders, Infectious Diseases, Neurology, Cardiovascular Diseases, Musculoskeletal Disorders, Other Indications), By End-Use (Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Academic And Research Institutes, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Cell And Gene Therapy Clinical Trial Services Market Overview
• Cell And Gene Therapy Clinical Trial Services market size has reached to $4.18 billion in 2025 • Expected to grow to $6.44 billion in 2030 at a compound annual growth rate (CAGR) of 8.7% • Growth Driver: Role Of Cell And Gene Therapy Trials In Tackling Genetic Diseases • Market Trend: Innovative Solutions In Cell And Gene Therapy Clinical Trial Services Market • North America was the largest region in 2025.What Is Covered Under Cell And Gene Therapy Clinical Trial Services Market?
Cell and gene therapy clinical trial services refer to specialized services offered by contract research organizations (CROs) or clinical trial management corporations to facilitate the design, implementation, and management of clinical trials for cell and gene therapies. These services, which range from protocol design and patient recruiting to regulatory compliance and data administration, serve as the foundation for trials to progress smoothly, ensuring precision, efficacy, and adherence to high standards. The main types of cell and gene therapy clinical trial services include clinical trial design and planning, supply and logistic services, regulatory affairs and compliance, data management and biostatics, site management and monitoring, and others. Clinical trial design and planning involve creating and organizing the blueprint for clinical trials, deciding what the trial will test, and how it will be conducted. Several types of therapies are included, such as gene therapy, cell therapy, and gene-modified cell therapy, which are used for various indications, such as oncology, hematology, metabolic disorders, infectious diseases, neurology, cardiovascular diseases, musculoskeletal disorders, and others. These are used by various end-users, including pharmaceutical and biotechnology companies, contract research organizations, academic and research institutes, and others.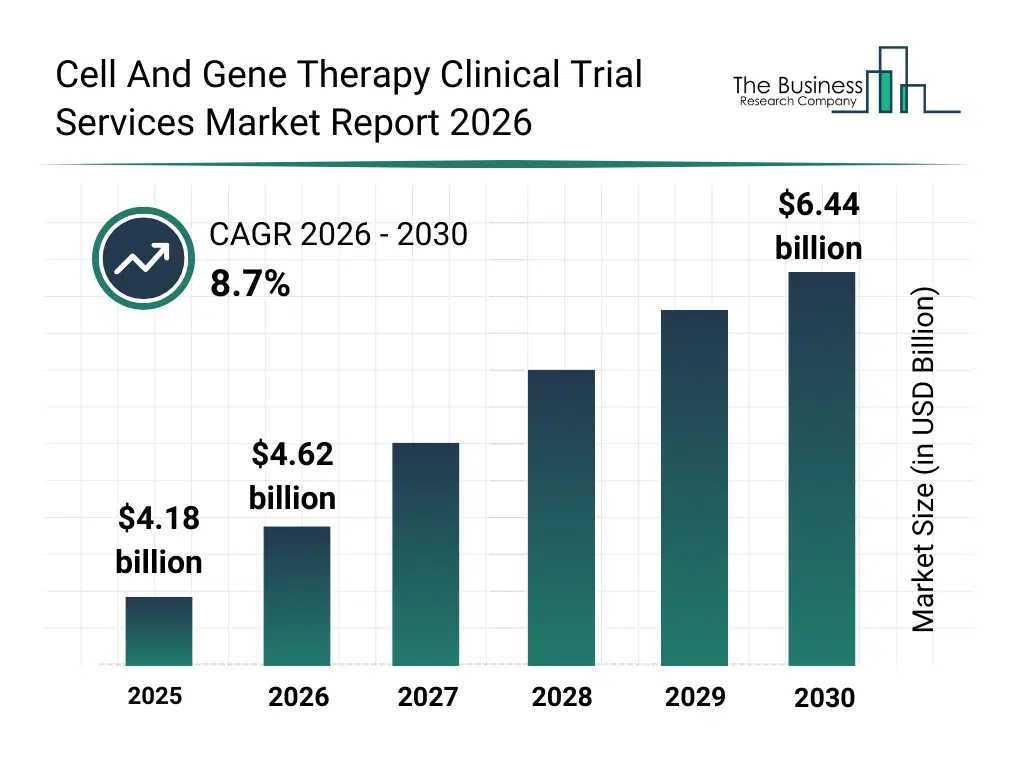
What Is The Cell And Gene Therapy Clinical Trial Services Market Size and Share 2026?
The cell and gene therapy clinical trial services market size has grown rapidly in recent years. It will grow from $4.18 billion in 2025 to $4.62 billion in 2026 at a compound annual growth rate (CAGR) of 10.3%. The growth in the historic period can be attributed to increasing number of cell and gene therapy clinical trials, rising outsourcing to specialized cros, growing complexity of trial designs, expansion of rare disease research, increasing regulatory oversight of advanced therapies.What Is The Cell And Gene Therapy Clinical Trial Services Market Growth Forecast?
The cell and gene therapy clinical trial services market size is expected to see strong growth in the next few years. It will grow to $6.44 billion in 2030 at a compound annual growth rate (CAGR) of 8.7%. The growth in the forecast period can be attributed to increasing demand for late-stage clinical trial services, rising adoption of digital trial technologies, expansion of global patient recruitment initiatives, growing focus on real-world evidence integration, increasing investments in precision clinical research. Major trends in the forecast period include increasing adoption of decentralized clinical trial models, growing use of digital patient recruitment platforms, rising integration of advanced data management systems, expansion of global trial site networks, enhanced focus on regulatory compliance and trial accuracy.Global Cell And Gene Therapy Clinical Trial Services Market Segmentation
1) By Service: Clinical Trial Design And Planning, Supply And Logistic Services, Regulatory Affairs And Compliance, Data Management And Biostatics, Site Management And Monitoring, Other Services 2) By Therapy Type: Gene Therapy, Cell Therapy, Gene Modified Cell Therapy 3) By Indication: Oncology, Hematology, Metabolic Disorders, Infectious Diseases, Neurology, Cardiovascular Diseases, Musculoskeletal Disorders, Other Indications 4) By End-Use: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Academic And Research Institutes, Other End-Users Subsegments: 1) By Clinical Trial Design And Planning: Protocol Development, Trial Feasibility Studies, Trial Design Consultation, Patient Recruitment And Enrollment Strategy, Risk-Based Monitoring Plans 2) By Supply And Logistic Services: Clinical Trial Supply Chain Management, Cold Chain Logistics, Packaging And Labeling Services, Transportation And Distribution Of Biological Samples, Customs And Import/Export Services 3) By Regulatory Affairs And Compliance: Regulatory Strategy And Consulting, Regulatory Submission And Documentation, Compliance With GMP (Good Manufacturing Practices), Regulatory Pathway Consultation, Clinical Trial Applications (CTAs) 4) By Data Management And Biostatistics: Clinical Data Management (CDM), Statistical Analysis And Reporting, Clinical Data Monitoring, Data Integration And Validation, Electronic Data Capture (EDC) Solutions 5) By Site Management And Monitoring: Site Selection And Initiation, Clinical Monitoring And Site Visits, Patient Recruitment And Retention, Site Training And Support, Site Performance Monitoring 6) By Other Services: Patient Advocacy And Engagement, Real-World Evidence (RWE) Studies, Post-Trial Services, Medical Writing And Documentation, Clinical Trial Auditing And InspectionWhat Is The Driver Of The Cell And Gene Therapy Clinical Trial Services Market?
The rising incidence of genetic disease is expected to propel the growth of the cell and gene therapy clinical trial services market going forward. Genetic diseases are ailments caused by variations in a person's DNA or genetic composition. The incidence of genetic diseases is on the rise due to numerous factors, including the increased availability of genetic testing, the occurrence of genetic mutations, and the prevalence of consanguineous marriage. Cell and gene therapy clinical trial services address the rising incidence of genetic diseases by encouraging the development of novel treatments, tailoring therapies to individual patients, accelerating the translation of research into clinical practice, and expanding the options available to patients suffering from genetic disorders. For instance, in 2023, according to the National Health Service, a UK-based government department, in England, sickle cell disorder (SCD) is the most widespread genetic condition, with around 17,000 people living with it (NHSE, 2023). Therefore, the rising incidence of genetic disease is driving the growth of the cell and gene therapy clinical trial services industry.Key Players In The Global Cell And Gene Therapy Clinical Trial Services Market
Major companies operating in the cell and gene therapy clinical trial services market are Thermo Fisher Scientific Inc., Sharp Services LLC, IQVIA, Laboratory Corporation of America Holdings, ICON plc, Syneos Health Inc., Catalent Pharma Solutions Inc., Charles River Laboratories International Inc., Parexel International Corporation, PRA Health Sciences Inc., Covance Inc., Medpace Holdings Inc., BioClinica Inc., Precision Medicine Group LLC, Worldwide Clinical Trials LLC, Clinigen Group plc, Evidera Inc., Advarra LLC, Veristat LLC, Clinipace Inc., Celonic AG, Cromsource Inc., Novotech Pty Ltd., MedSource Holdings Inc., Frontage Laboratories Inc.Global Cell And Gene Therapy Clinical Trial Services Market Trends and Insights
Major companies operating in the cell and gene therapy clinical trial services market are focusing their efforts on introducing advanced technology with customer relationship management system technology, to improve efficiency, and gain a competitive edge in the market. The customer relationship management system utilized by cell and gene therapy clinical trial services streamlines and coordinates interactions with trial participants, healthcare providers, and other stakeholders throughout the clinical trial process. For instance, in April 2023, AmerisourceBergen Corporation, a US-based healthcare company, launched the Cell and Gene Therapy (CGT) Integration Hub, a platform that aims to increase connectivity and streamline processes throughout the cell and gene therapy treatment pathway. This platform is backed by AmerisourceBergen's customer relationship management (CRM) system and attempts to streamline the path-to-care process, in part by giving physicians and patient services teams better visibility across the therapy development and delivery process. The CGT Integration Hub, which includes features such as expedited benefits investigation, real-time status tracking, and proactive alerts, aims to simplify care coordination, minimize barriers, and improve the entire patient and provider experience when delivering cell and gene therapies.What Are Latest Mergers And Acquisitions In The Cell And Gene Therapy Clinical Trial Services Market?
In January 2024, Kyowa Kirin Co. Ltd., a Japan-based pharmaceutical company, acquired Orchard Therapeutics Limited for an undisclosed amount. Through this acquisition, Kyowa Kirin aims to enhance its focus on addressing the unmet medical needs of individuals with genetic and severe diseases by leveraging Orchard's expertise in developing and commercializing gene therapies for rare diseases. Orchard Therapeutics Limited is a UK-based biotechnology company primarily dedicated to the research, development, and commercialization of gene therapies for rare diseases.Regional Outlook
North America was the largest region in the cell and gene therapy clinical trial services market in 2025. It is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Cell And Gene Therapy Clinical Trial Services Market?
The cell and gene therapy clinical trial services market includes revenues earned by providing services, such as regulatory pathways navigation, logistics management, patient retention strategy management, and biomarker analysis services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Cell And Gene Therapy Clinical Trial Services Market Report 2026?
The cell and gene therapy clinical trial services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the cell and gene therapy clinical trial services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Cell And Gene Therapy Clinical Trial Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.62 billion |
| Revenue Forecast In 2035 | $6.44 billion |
| Growth Rate | CAGR of 10.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service, Therapy Type, Indication, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Sharp Services LLC, IQVIA, Laboratory Corporation of America Holdings, ICON plc, Syneos Health Inc., Catalent Pharma Solutions Inc., Charles River Laboratories International Inc., Parexel International Corporation, PRA Health Sciences Inc., Covance Inc., Medpace Holdings Inc., BioClinica Inc., Precision Medicine Group LLC, Worldwide Clinical Trials LLC, Clinigen Group plc, Evidera Inc., Advarra LLC, Veristat LLC, Clinipace Inc., Celonic AG, Cromsource Inc., Novotech Pty Ltd., MedSource Holdings Inc., Frontage Laboratories Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
