
Cell And Gene Therapy Supply Chain/Logistics Market Report 2026
Global Outlook – By Type Of Software Solution (Cell Orchestration Platform, Enterprise Manufacturing System, Inventory Management System, Laboratory Information Management System, Logistics Management System, Patient Management System, Quality Management System, Tracking And Tracing System), By Mode Of Deployment (Cloud-Based Solution, On-Premises Solution), By Component (Cryogenic Shippers, Cryogenic Storage Freezers, Ultra-Low Freezers, Cold Chain Management Systems, Shipment, Storage Medium, Cryogenic Packout Kits, Other Components), By Application Area (Donor Eligibility Assessment, Sample Collection, Manufacturing, Logistics, Patient Verification, Treatment Follow-Up), By End User (Biobank, Cell Therapy Lab, Hospital, Research Institute, Commercial Organization) - Market Size, Trends, And Global Forecast 2026-2035
Cell And Gene Therapy Supply Chain/Logistics Market Overview
• Cell And Gene Therapy Supply Chain/Logistics market size has reached to $1.58 billion in 2025 • Expected to grow to $2.81 billion in 2030 at a compound annual growth rate (CAGR) of 12.1% • Growth Driver: The Surging Demand For Cell And Gene Therapies Reshapes Supply Chain And Logistics • Market Trend: Advancements In Technology Propel Cell And Gene Therapy Logistics • North America was the largest region and fastest growing region.What Is Covered Under Cell And Gene Therapy Supply Chain/Logistics Market?
Cell and gene therapy supply chains and logistics refer to the processes and activities involved in efficiently and effectively managing the flow of materials, information, and resources needed to develop, manufacture, and distribute cell and gene therapies from their initial production to their final administration to patients. These services provide cutting-edge treatments to patients while maintaining the highest safety and efficacy standards throughout the supply chain. The main types of software solutions are cell orchestration platforms, enterprise manufacturing systems, inventory management systems, laboratory information management systems, logistics management systems, patient management systems, quality management systems, and tracking and tracing systems. A cell orchestration platform refers to a software or technological platform that facilitates the coordination, management, and integration of various processes and activities involved in cell therapy development, manufacturing, and delivery and is used to help streamline and automate tasks related to cell sourcing, manufacturing, quality control, logistics, patient tracking, and data management. They are deployed as a cloud-based solution; an on-premises solution in cryogenic shippers; cryogenic storage freezers; ultra-low freezers; cold chain management systems; shipment; storage medium; cryogenic packout kits; and other components. These solutions are used in applications such as donor eligibility assessment, sample collection, manufacturing, logistics, patient verification, and treatment follow-up by end users including biobanks, cell therapy labs, hospitals, research institutes, and commercial organizations.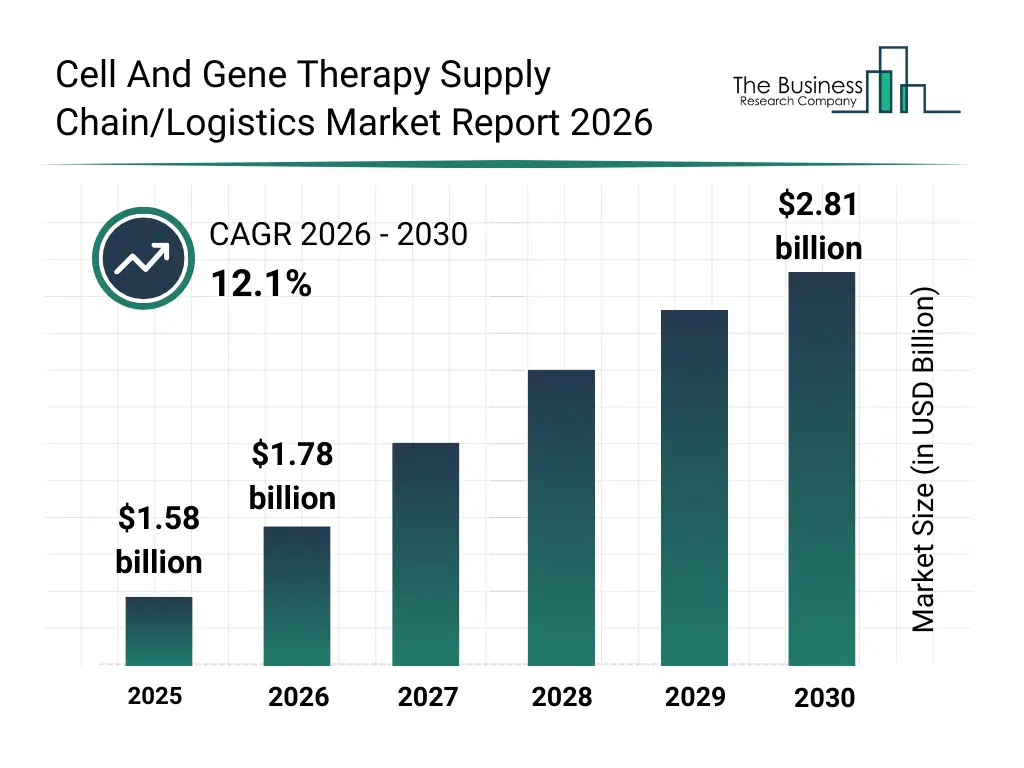
What Is The Cell And Gene Therapy Supply Chain/Logistics Market Size and Share 2026?
The cell and gene therapy supply chain/logistics market size has grown rapidly in recent years. It will grow from $1.58 billion in 2025 to $1.78 billion in 2026 at a compound annual growth rate (CAGR) of 12.2%. The growth in the historic period can be attributed to increasing complexity of cell and gene therapy logistics, early adoption of cryogenic shipping solutions, expansion of centralized manufacturing models, growing regulatory focus on traceability, rising use of specialized logistics providers.What Is The Cell And Gene Therapy Supply Chain/Logistics Market Growth Forecast?
The cell and gene therapy supply chain/logistics market size is expected to see rapid growth in the next few years. It will grow to $2.81 billion in 2030 at a compound annual growth rate (CAGR) of 12.1%. The growth in the forecast period can be attributed to increasing commercialization of personalized therapies, rising demand for end-to-end supply chain visibility, expansion of global therapy distribution networks, growing adoption of cloud-based logistics platforms, increasing investments in smart cold chain infrastructure. Major trends in the forecast period include expansion of integrated cold chain management platforms, growing adoption of real-time tracking and traceability systems, rising use of cryogenic logistics solutions, increasing integration of patient-centric supply chain models, enhanced focus on chain of custody compliance.Global Cell And Gene Therapy Supply Chain/Logistics Market Segmentation
1) By Type Of Software Solution: Cell Orchestration Platform, Enterprise Manufacturing System, Inventory Management System, Laboratory Information Management System, Logistics Management System, Patient Management System, Quality Management System, Tracking And Tracing System 2) By Mode Of Deployment: Cloud-Based Solution, On-Premises Solution 3) By Component: Cryogenic Shippers, Cryogenic Storage Freezers, Ultra-Low Freezers, Cold Chain Management Systems, Shipment, Storage Medium, Cryogenic Packout Kits, Other Components 4) By Application Area: Donor Eligibility Assessment, Sample Collection, Manufacturing, Logistics, Patient Verification, Treatment Follow-Up 5) By End User: Biobank, Cell Therapy Lab, Hospital, Research Institute, Commercial Organization Subsegments: 1) By Cell Orchestration Platform: Workflow Management, Scheduling And Coordination Tools 2) By Enterprise Manufacturing System: Production Planning And Control, Manufacturing Execution Systems (MES) 3) By Inventory Management System: Stock Tracking And Management, Inventory Forecasting And Optimization 4) By Laboratory Information Management System (LIMS): Sample Tracking And Management, Data Management And Reporting 5) By Logistics Management System: Route Optimization And Planning, Transportation Management 6) By Patient Management System: Patient Scheduling And Coordination, Data Management And Tracking Of Patient Outcomes 7) By Quality Management System: Compliance Tracking, Quality Assurance And Control 8) By Tracking And Tracing System: Real-Time Tracking Of Shipments, Chain Of Custody DocumentationWhat Is The Driver Of The Cell And Gene Therapy Supply Chain/Logistics Market?
The increasing need for cell and gene therapies is driving the cell and gene therapy supply chain or logistics market. Cell and gene therapies are advanced medical treatments that modify cells or genes to treat diseases, offering potential breakthroughs for untreatable conditions. These cell and gene therapies need to be stored at the proper temperature and storage conditions, which can be achieved using supply chain and logistics services, which help from sourcing to distributing and storing these cell and gene therapies. For instance, in 2023, according to the American Society of Gene & Cell Therapy, a US-based non-profit medical and scientific organization, the number of gene therapies in Phase III clinical trials grew by 10% in Q3 2023, the first quarterly increase since Q3 2022. Therefore, the increasing need for cell and gene therapies drives the cell and gene therapy supply chain or logistics market forward.Key Players In The Global Cell And Gene Therapy Supply Chain/Logistics Market
Major companies operating in the cell and gene therapy supply chain/logistics market are AmerisourceBergen Corporation, United Parcel Service, Thermo Fisher Scientific Inc., Avantor Sciences, Lonza Group AG, Arvato Group, Catalent Inc., Charles River Laboratories International, Parexel International Corporation, Cytiva, Almac Group, World Courier, Sharp Packaging Services, PCI Pharma Services, Cryoport Inc., Brooks Life Sciences, BioLife Solutions Inc., Biocair Inc., QuickSTAT, Clarkston Consulting Inc., Central Pharma, TrakCel limited, MAK-SYSTEM International Group, Hypertrust Patient Data Care GmbH, Atelerix Ltd., Polar Express Transportation, Be The Match BioTherapies Inc., Yourway Biopharma Services Company, Azenta Life Sciences Inc., Biostor Ltd.Global Cell And Gene Therapy Supply Chain/Logistics Market Trends and Insights
Modern technology platforms are a key trend gaining popularity in the cell and gene therapy supply chain/logistics markets. Major companies operating in the market are developing modern technology platforms for cell and gene therapies to sustain their position in the market. For instance, in April 2023, AmerisourceBergen Corporation., a US-based healthcare company specializing in cell and gene therapy supply chain logistics, launched the Cell and Gene Therapy (CGT) Integration Hub. The Cell and Gene Therapy (CGT) Integration Hub is powered by AmerisourceBergen’s innovative customer relationship management (CRM) system. It can be integrated into the biopharma platform to facilitate better monitoring of the development and delivery of cell and gene therapies.What Are Latest Mergers And Acquisitions In The Cell And Gene Therapy Supply Chain/Logistics Market?
In October 2025, Peli BioThermal, a US-based provider of temperature-controlled packaging solutions, acquired EVO, for an undisclosed amount. With this acquisition, Peli BioThermal aims to expand its cold chain capabilities, enhance service offerings for temperature-sensitive shipments, strengthen its global logistics network, and support growth in life sciences and healthcare markets. EVO is a US-based cold chain company, that offers cell and gene therapy supply chain or logistics.Regional Outlook
North America was the largest region in the cell and gene therapy supply chain/logistics market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Cell And Gene Therapy Supply Chain/Logistics Market?
The cell and gene therapy supply chain and logistics market includes revenues earned by entities by providing sourcing and collection, quality testing, distribution, and transportation services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Cell And Gene Therapy Supply Chain/Logistics Market Report 2026?
The cell and gene therapy supply chain/logistics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the cell and gene therapy supply chain/logistics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Cell And Gene Therapy Supply Chain/Logistics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.78 billion |
| Revenue Forecast In 2035 | $2.81 billion |
| Growth Rate | CAGR of 12.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type Of Software Solution, Mode Of Deployment, Component, Application Area, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | AmerisourceBergen Corporation, United Parcel Service, Thermo Fisher Scientific Inc., Avantor Sciences, Lonza Group AG, Arvato Group, Catalent Inc., Charles River Laboratories International, Parexel International Corporation, Cytiva, Almac Group, World Courier, Sharp Packaging Services, PCI Pharma Services, Cryoport Inc., Brooks Life Sciences, BioLife Solutions Inc., Biocair Inc., QuickSTAT, Clarkston Consulting Inc., Central Pharma, TrakCel limited, MAK-SYSTEM International Group, Hypertrust Patient Data Care GmbH, Atelerix Ltd., Polar Express Transportation, Be The Match BioTherapies Inc., Yourway Biopharma Services Company, Azenta Life Sciences Inc., Biostor Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
