
Central Precocious Puberty (CPP) Treatment Market Report 2026
Global Outlook – By Type (Medication, Surgery), By Diagnosis (Blood Tests, Magnetic Resonance Imaging (MRI), Computed Tomography (CT) Scans, X-rays), By Gender (Girls, Boys), By End-User (Hospitals, Specialty Clinics, Homecare, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Central Precocious Puberty (CPP) Treatment Market Overview
• Central Precocious Puberty (CPP) Treatment market size has reached to $1.84 billion in 2025 • Expected to grow to $2.74 billion in 2030 at a compound annual growth rate (CAGR) of 8.2% • Growth Driver: Impact of Obesity on Central Precocious Puberty (CPP) Treatment Market Growth • Market Trend: Advancing Pediatric Endocrinology With Extended-Release Innovation • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Central Precocious Puberty (CPP) Treatment Market?
Central precocious puberty (CPP) treatment refers to medical interventions designed to manage and delay the early onset of puberty caused by premature activation of the hypothalamic-pituitary-gonadal (HPG) axis. Central precocious puberty is characterized by the onset of secondary sexual characteristics before age 8 in girls and before age 9 in boys. The primary treatment involves the use of gonadotropin-releasing hormone (GnRH) agonists, which suppress the premature release of sex hormones and slow down or halt the progression of puberty. The main types in the central precocious puberty (CPP) treatment market are medication and surgery. Medications are drugs prescribed to treat central precocious puberty (CPP) by delaying the onset of puberty. Diagnoses are carried out through blood tests, magnetic resonance imaging (MRI), computed tomography (CT) scans, and X-rays. The treatments are applicable to both girls and boys. The main end-users include hospitals, specialty clinics, homecare settings, and other healthcare facilities.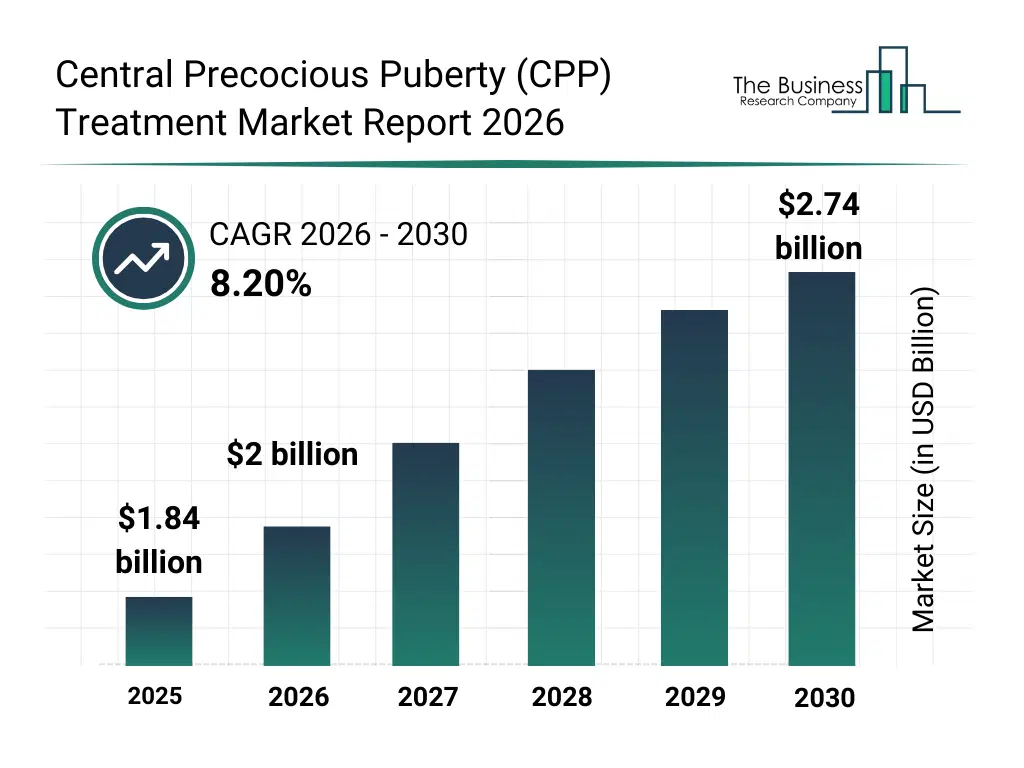
What Is The Central Precocious Puberty (CPP) Treatment Market Size and Share 2026?
The central precocious puberty (cpp) treatment market size has grown strongly in recent years. It will grow from $1.84 billion in 2025 to $2 billion in 2026 at a compound annual growth rate (CAGR) of 8.5%. The growth in the historic period can be attributed to limited treatment options for cpp, lack of awareness about early puberty management, growing pediatric population, increasing hospital and clinic infrastructure, reliance on conventional hormonal therapies.What Is The Central Precocious Puberty (CPP) Treatment Market Growth Forecast?
The central precocious puberty (cpp) treatment market size is expected to see strong growth in the next few years. It will grow to $2.74 billion in 2030 at a compound annual growth rate (CAGR) of 8.2%. The growth in the forecast period can be attributed to development of novel gnrh-based therapies, integration of digital monitoring tools, rising pediatric endocrinology specialization, increasing government healthcare initiatives, expansion of homecare and telemedicine programs. Major trends in the forecast period include rising awareness of early diagnosis of cpp, increasing adoption of gnrh agonists and antagonists, growth in personalized hormonal therapy approaches, expansion of pediatric endocrinology clinics and specialty centers, integration of homecare and remote monitoring solutions.Global Central Precocious Puberty (CPP) Treatment Market Segmentation
1) By Type: Medication, Surgery 2) By Diagnosis: Blood Tests, Magnetic Resonance Imaging (MRI), Computed Tomography (CT) Scans, X-rays 3) By Gender: Girls, Boys 4) By End-User: Hospitals, Specialty Clinics, Homecare, Other End-Users Subsegments: 1) By Medication: Gonadotropin-Releasing Hormone (GnRH) Agonists, GnRH Antagonists, Aromatase Inhibitors 2) By Surgery: Ovarian Tissue Removal, Surgical Removal Of Hormone-Producing TumorsWhat Is The Driver Of The Central Precocious Puberty (CPP) Treatment Market?
The increasing prevalence of hormonal disorders is expected to propel the growth of the central precocious puberty (CPP) treatment market going forward. Hormonal disorders refer to a wide range of medical conditions caused by imbalances in the body's hormone levels. The rise in prevalence of hormonal disorders is primarily due to obesity, which can disrupt the balance of hormones such as insulin, estrogen, and leptin, leading to conditions like diabetes, thyroid dysfunction, and reproductive disorders. Central precocious puberty (CPP) treatment helps hormonal disorders by regulating early hormone overproduction, slowing premature sexual development, and restoring normal hormonal balance. For instance, in February 2025, according to the World Health Organization, a Switzerland-based intergovernmental organization, the given report states that polycystic ovary syndrome is a major public health issue and one of the most common hormonal disorders in women of reproductive age, affecting an estimated 6–13%. Up to 70% of cases remain undiagnosed. Therefore, the increasing prevalence of hormonal disorders is driving the growth of the central precocious puberty (CPP) treatment industry.Key Players In The Global Central Precocious Puberty (CPP) Treatment Market
Major companies operating in the central precocious puberty (cpp) treatment market are Pfizer Inc., Merck & Co. Inc., AbbVie Inc., AstraZeneca plc, Takeda Pharmaceutical Company Limited, Teva Pharmaceutical Industries Ltd., Astellas Pharma Inc., Sun Pharmaceutical Industries Ltd., Ipsen S.A., Dr Reddy's Laboratories Limited, Endo Pharmaceuticals Inc., Livzon Pharmaceutical Group Co. Ltd., Daewoong Pharmaceutical Co.Ltd, Bachem Holding AG, Ferring Pharmaceuticals, Tolmar Pharmaceuticals Inc., Arbor Pharmaceuticals, Debiopharm Group, Beijing Biote Pharmaceutical Ltd, GP Pharm S.A.Global Central Precocious Puberty (CPP) Treatment Market Trends and Insights
Major companies operating in the central precocious puberty (CPP) treatment market are focusing on conducting clinical trial to develop innovative therapies, such as long-acting GnRH analog therapies, to improve treatment adherence, efficacy, and convenience for pediatric patients experiencing early onset puberty, compared with traditional shorter-acting injections that require more frequent administration and monitoring. Long-acting GnRH analogs act by suppressing the pituitary-gonadal axis, reducing premature sex hormone production, and slowing progression of secondary sexual characteristics until the appropriate age for puberty. For instance, in September 2024, Debiopharm Group, a Switzerland-based biopharmaceutical company, announced positive results from Phase 3 trials of Debio 4326, a novel long-acting treatment for CPP. Debio 4326 is composed of a sustained-release GnRH analog formulated for monthly subcutaneous injection; it functions by continuously downregulating gonadotropin secretion to stabilize hormone levels and delay premature pubertal development; unique features include extended dosing interval, pediatric-friendly administration, and predictable pharmacokinetics; applications include management of children diagnosed with CPP, monitoring of pubertal progression, and support of normal growth and psychosocial development; benefits include improved patient compliance, reduced injection frequency, and effective management of hormone levels to restore age-appropriate development.What Are Latest Mergers And Acquisitions In The Central Precocious Puberty (CPP) Treatment Market?
In December 2023, Zydus Lifesciences Ltd, an India-based pharmaceutical company, partnered with Daewoong Pharmaceutical. The partnership aims to leverage Zydus' clinical development and commercialization capabilities and Daewoong's proprietary technology and manufacturing expertise to ensure the successful development and availability of leuprolide acetate for depot suspension in the U.S. market, thereby enhancing patient access to this essential treatment. Daewoong Pharmaceutical Co. Ltd. is a South Korea-based pharmaceutical company specializing in the research, development, and manufacturing of pharmaceutical products, including leuprolide acetate, for treating central precocious puberty (CPP).Regional Insights
North America was the largest region in the central precocious puberty (CPP) treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Central Precocious Puberty (CPP) Treatment Market?
The central precocious puberty (CPP) treatment market consists of revenues earned by entities by providing services such as endocrinology consultations, regular hormone assessments, and growth monitoring. The market value includes the value of related goods sold by the service provider or included within the service offering. The central precocious puberty (CPP) treatment market also includes sales of leuprolide acetate, triptorelin, histrelin, and goserelin. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Central Precocious Puberty (CPP) Treatment Market Report 2026?
The central precocious puberty (cpp) treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the central precocious puberty (cpp) treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Central Precocious Puberty (CPP) Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2 billion |
| Revenue Forecast In 2035 | $2.74 billion |
| Growth Rate | CAGR of 8.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Diagnosis, Gender, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Merck & Co. Inc., AbbVie Inc., AstraZeneca plc, Takeda Pharmaceutical Company Limited, Teva Pharmaceutical Industries Ltd., Astellas Pharma Inc., Sun Pharmaceutical Industries Ltd., Ipsen S.A., Dr Reddy's Laboratories Limited, Endo Pharmaceuticals Inc., Livzon Pharmaceutical Group Co. Ltd., Daewoong Pharmaceutical Co.Ltd, Bachem Holding AG, Ferring Pharmaceuticals, Tolmar Pharmaceuticals Inc., Arbor Pharmaceuticals, Debiopharm Group, Beijing Biote Pharmaceutical Ltd, GP Pharm S.A. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
