
Cervical Total Disc Replacement Device Market Report 2026
Global Outlook – By Disc Type (Metal On A Biocompatible Material (M-o-B), Metal On Metal (M-o-M)), By Material (Polymeric, Metallic, Biocompatible), By Design (Constrained, Semi-Constrained, Unconstrained), By End User (Hospitals, Ambulatory Surgical Center, Specialty Clinics, Clinics) – Market Size, Trends, Strategies, and Forecast to 2035
Cervical Total Disc Replacement Device Market Overview
• Cervical Total Disc Replacement Device market size has reached to $3.33 billion in 2025 • Expected to grow to $8.09 billion in 2030 at a compound annual growth rate (CAGR) of 19.6% • Growth Driver: Surging Demand For Cervical Total Disc Replacement Devices Amid Rising Spine-Related Injuries • Market Trend: Redefining Spinal Health With Viscoelastic Core For Enhanced Motion • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Cervical Total Disc Replacement Device Market?
A cervical total disc replacement (CTDR) device refers to a medical implant used in a surgical procedure to treat certain spine-related conditions in the cervical spine (neck) by using an artificial disc to replace an intervertebral disc that has been injured or lost its ability to function. The CTDR device is designed to mimic the natural structure and movement of the spine, including backward and forward, side to side, and rotation. The main types of discs in the cervical total disc replacement device are metal on a biocompatible material (M-o-B) and metal on metal (M-o-M). Metal on a biocompatible material (M-o-B) refers to a design concept used in the construction of certain medical implants, including orthopedic and spinal implants. In this context, it involves combining a metal component with another biocompatible material to create a functional and effective medical device. It includes a variety of materials, such as polymeric, metallic, and biocompatible, and comes in various designs, such as constrained, semi-constrained, and unconstrained. It includes end-user hospitals, ambulatory surgical centers, specialty clinics, and clinics.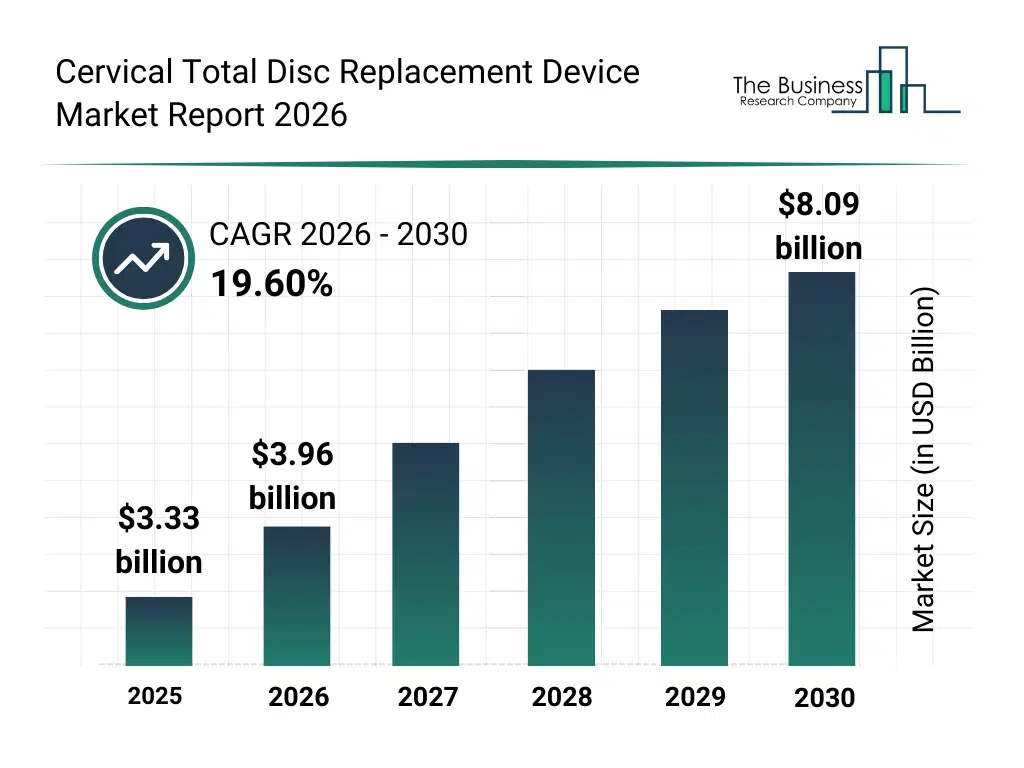
What Is The Cervical Total Disc Replacement Device Market Size and Share 2026?
The cervical total disc replacement device market size has grown rapidly in recent years. It will grow from $3.33 billion in 2025 to $3.96 billion in 2026 at a compound annual growth rate (CAGR) of 18.8%. The growth in the historic period can be attributed to rising incidence of cervical spine disorders, increasing preference for disc replacement over fusion, advancements in spinal surgery techniques, growing surgeon familiarity with ct dr procedures, availability of improved implant materials.What Is The Cervical Total Disc Replacement Device Market Growth Forecast?
The cervical total disc replacement device market size is expected to see rapid growth in the next few years. It will grow to $8.09 billion in 2030 at a compound annual growth rate (CAGR) of 19.6%. The growth in the forecast period can be attributed to increasing demand for motion-preserving spinal treatments, rising adoption of outpatient spine surgeries, expansion of aging population with degenerative disc disease, growing investments in spine implant innovation, increasing regulatory approvals for next-generation implants. Major trends in the forecast period include increasing adoption of motion-preserving spine implants, rising demand for minimally invasive cervical procedures, growing use of advanced biocompatible materials, expansion of customized disc implant designs, enhanced focus on long-term implant durability.Global Cervical Total Disc Replacement Device Market Segmentation
1) By Disc Type: Metal On A Biocompatible Material (M-o-B), Metal On Metal (M-o-M) 2) By Material: Polymeric, Metallic, Biocompatible 3) By Design: Constrained, Semi-Constrained, Unconstrained 4) By End User: Hospitals, Ambulatory Surgical Center, Specialty Clinics, Clinics Subsegments: 1) By Metal On A Biocompatible Material (M-O-B): Titanium On Polyethylene, Cobalt-Chromium On Bioceramic, PEEK With Metal Coating 2) By Metal On Metal (M-O-M): Cobalt-Chromium On Cobalt-Chromium, Titanium On Titanium, Stainless Steel On Stainless SteelWhat Is The Driver Of The Cervical Total Disc Replacement Device Market?
The increase in spine-related injuries is expected to propel the growth of the cervical total disc replacement device market. Spine-related injuries are injuries that affect the spinal cord, the nerves that run through the spine, or the bones that make up the spine. Cervical total disc replacement devices are useful in spinal cord injuries and treat spinal instability, which is a condition in which the bones in the neck are not aligned properly. For instance, in May 2023, according to Spinalcord, a US-based online community and resource for people affected by spinal cord injuries, each year, around 18,000 new spinal cord injuries (SCIs) occur in the U.S., maintaining a rate of about 54 cases per million people. Currently, 302,000 individuals live with traumatic SCIs, with 79% of new cases affecting males, primarily among those aged 16 to 30. Therefore, an increase in spine-related injuries is driving the growth of the cervical total disc replacement device industry.Key Players In The Global Cervical Total Disc Replacement Device Market
Major companies operating in the cervical total disc replacement device market are Medtronic plc, Johnson and Johnson Pvt Ltd., Stryker Corporation, Zimmer Biomet Holdings Inc., DePuy Synthes Inc., NuVasive Inc., Globus Medical Inc., ZimVie Inc., Orthofix Medical Inc., Aesculap Implant Systems Inc., Alphatec Holdings Inc., LDR Holding Corporation, Centinel Spine Inc., Spineart SA, CoreLink LLC, Innovative Spinal Technologies Inc., Precision Spine Inc., Spinal Elements Inc., Applied Spine Technologies Inc., Spinal Kinetics LLC, Paradigm Spine LLC, Implanet SA, Osimplant, AxioMed LLCGlobal Cervical Total Disc Replacement Device Market Trends and Insights
Major companies operating in the Cervical Total Disc Replacement Device Market are focusing on innovative designs, such as articulating viscoelastic core and advanced motion technology, to offer patients enhanced mobility and long-term spinal stability, aiming to replicate natural cervical motion while minimizing wear and tear. An articulating viscoelastic core is designed to mimic natural movement by allowing flexible and controlled motion, with the viscoelastic material providing a combination of shock absorption and progressive resistance, similar to how the body's own tissues behave under stress. For instance, in May 2024, NGMedical GmbH, a Germany-based company which manufactures medical technology, launched the MOVE-C Cervical Artificial Disc Prosthesis, an innovative spinal implant designed to mimic natural cervical motion using an articulating surface and viscoelastic core. This new device aims to provide enhanced physiological motion with progressive resistance, offering a unique solution for cervical spine surgeries.What Are Latest Mergers And Acquisitions In The Cervical Total Disc Replacement Device Market?
In May 2023, Synergy Spine Solutions, a US-based medical device company, partnered with LifeHealthcare Group Limited to expand access to the company’s marquis technology. With this partnership, Synergy Spine Solutions and LifeHealthcare aim to expand access to the innovative Synergy Disc® technology in Australia and New Zealand, enhancing the availability of advanced cervical disc replacement solutions for surgeons and their patients, ultimately improving patient care and outcomes in spine surgery. LifeHealthcare Group Limited is an Australia-based diversified healthcare company that provides implantable, non-implantable, and cervical total disc replacement devices.Regional Outlook
North America was the largest region in the cervical total disc replacement device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Cervical Total Disc Replacement Device Market?
The cervical total disc replacement (CTDR) device market consists of sales of artificial discs, endplates, articulating cores, cervicalufix systems, inlays and bearings, and anchors. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Cervical Total Disc Replacement Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.96 billion |
| Revenue Forecast In 2035 | $8.09 billion |
| Growth Rate | CAGR of 18.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Disc Type, Material, Design, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic plc, Johnson and Johnson Pvt Ltd., Stryker Corporation, Zimmer Biomet Holdings Inc., DePuy Synthes Inc., NuVasive Inc., Globus Medical Inc., ZimVie Inc., Orthofix Medical Inc., Aesculap Implant Systems Inc., Alphatec Holdings Inc., LDR Holding Corporation, Centinel Spine Inc., Spineart SA, CoreLink LLC, Innovative Spinal Technologies Inc., Precision Spine Inc., Spinal Elements Inc., Applied Spine Technologies Inc., Spinal Kinetics LLC, Paradigm Spine LLC, Implanet SA, Osimplant, AxioMed LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
