
Checkpoint Inhibitor Refractory Cancer Market Report 2026
Global Outlook – By Therapy (Immune Checkpoint Inhibitors, Combination Therapies, Targeted Therapies), By Cancer Type (Non-Small Cell Lung Cancer (NSCLC), Melanoma, Renal Cell Carcinoma (RCC), Head And Neck Squamous Cell Carcinoma (HNSCC), Urothelial Carcinoma, Triple-Negative Breast Cancer (TNBC), Colorectal Cancer, Other Cancer Types), By Mechanism Of Action (Programmed Cell Death Protein (PD)-1 Inhibitors, Programmed Death-Ligand (PD)-L1 Inhibitors, Cytotoxic T-Lymphocyte–Associated Protein (CTLA)-4 Inhibitors, Other Mechanism Of Actions), By Administration Route (Intravenous (IV), Oral), By End-User (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies) – Market Size, Trends, Strategies, and Forecast to 2035
Checkpoint Inhibitor Refractory Cancer Market Overview
• Checkpoint Inhibitor Refractory Cancer market size has reached to $36.69 billion in 2025 • Expected to grow to $63.02 billion in 2030 at a compound annual growth rate (CAGR) of 11.4% • Growth Driver: Rising Incidence Of Cancer Driving The Growth Of The Market Due To An Aging Population • Market Trend: Technological Advancements And Early-Phase Clinical Data In Checkpoint Inhibitor Refractory Cancer Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Checkpoint Inhibitor Refractory Cancer Market?
Checkpoint inhibitor refractory cancer is a type of cancer that fails to respond to immune checkpoint inhibitors such as PD-1, PD-L1, or CTLA-4 blockers. This can occur as primary resistance, where there is no initial response, or as acquired resistance, where the cancer progresses after an initial benefit. Managing these cases often requires alternative treatments, such as combination immunotherapy, targeted agents, or novel experimental strategies. The main therapies for checkpoint inhibitor refractory cancer include immune checkpoint inhibitors, combination therapies, and targeted therapies. Immune checkpoint inhibitors are medicines that help the immune system recognize and attack cancer cells by blocking proteins that stop immune cells from working properly. It is used for various cancer types such as non-small cell lung cancer (NSCLC), melanoma, renal cell carcinoma (RCC), head and neck squamous cell carcinoma (HNSCC), urothelial carcinoma, triple-negative breast cancer (TNBC), colorectal cancer, and others. The various mechanisms of action include programmed cell death protein (PD)-1 inhibitors, programmed death-ligand (PD)-l1 inhibitors, cytotoxic T-lymphocyte–associated protein (CTLA)-4 inhibitors, and others. and the various routes of administration involved are intravenous (IV) and oral. It is used by several end users, including hospital pharmacies, retail pharmacies, and online pharmacies.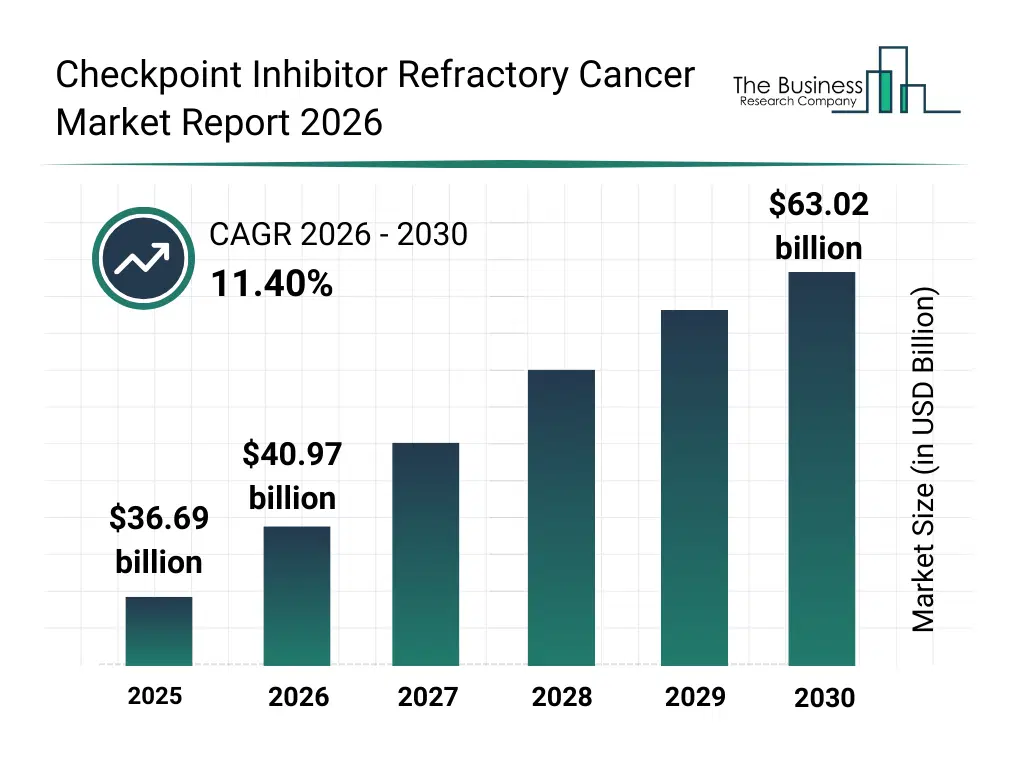
What Is The Checkpoint Inhibitor Refractory Cancer Market Size and Share 2026?
The checkpoint inhibitor refractory cancer market size has grown rapidly in recent years. It will grow from $36.69 billion in 2025 to $40.97 billion in 2026 at a compound annual growth rate (CAGR) of 11.7%. The growth in the historic period can be attributed to increasing use of immune checkpoint inhibitors in oncology, rising incidence of advanced-stage cancers, limited treatment options after immunotherapy failure, growing oncology drug approvals, expansion of cancer treatment centers.What Is The Checkpoint Inhibitor Refractory Cancer Market Growth Forecast?
The checkpoint inhibitor refractory cancer market size is expected to see rapid growth in the next few years. It will grow to $63.02 billion in 2030 at a compound annual growth rate (CAGR) of 11.4%. The growth in the forecast period can be attributed to development of next-generation immunotherapies, increasing investment in combination and targeted therapies, rising precision oncology adoption, expansion of global oncology clinical trials, growing focus on personalized cancer treatment strategies. Major trends in the forecast period include rising focus on overcoming primary and acquired resistance to checkpoint inhibitors, increasing adoption of combination immunotherapy regimens, growth in targeted therapies for immunotherapy-refractory cancers, expansion of clinical trials for novel and experimental therapies, rising demand for biomarker-based patient stratification.Global Checkpoint Inhibitor Refractory Cancer Market Segmentation
1) By Therapy: Immune Checkpoint Inhibitors, Combination Therapies, Targeted Therapies 2) By Cancer Type: Non-Small Cell Lung Cancer (NSCLC), Melanoma, Renal Cell Carcinoma (RCC), Head And Neck Squamous Cell Carcinoma (HNSCC), Urothelial Carcinoma, Triple-Negative Breast Cancer (TNBC), Colorectal Cancer, Other Cancer Types 3) By Mechanism Of Action: Programmed Cell Death Protein (PD)-1 Inhibitors, Programmed Death-Ligand (PD)-L1 Inhibitors, Cytotoxic T-Lymphocyte–Associated Protein (CTLA)-4 Inhibitors, Other Mechanism Of Actions 4) By Administration Route: Intravenous (IV), Oral 5) By End-User: Hospital Pharmacies, Retail Pharmacies, Online Pharmacies Subsegments: 1) By Immune Checkpoint Inhibitors (ICIs): PD-1 Inhibitors, PD-L1 Inhibitors, CTLA-4 Inhibitors 2) By Combination Therapies: Checkpoint Inhibitor + Chemotherapy, Checkpoint Inhibitor + Targeted Therapy, Checkpoint Inhibitor + Other Immunotherapies, Dual Checkpoint Inhibitor Combinations 3) By Targeted Therapies: Tyrosine Kinase Inhibitors (TKIs), Monoclonal Antibodies Targeting Specific Cancer MarkersWhat Is The Driver Of The Checkpoint Inhibitor Refractory Cancer Market?
The rising incidence of cancer is expected to propel the growth of the checkpoint inhibitor refractory cancer market going forward. Cancer refers to a disease characterized by the uncontrolled division and growth of abnormal cells in the body. The rising incidence of cancer is mainly due to an aging population, as longer life expectancy increases the likelihood of developing cancer over time. Recognizing checkpoint inhibitor refractory cancer is valuable in cancer management, as it enables early detection of treatment resistance, guiding clinicians to promptly modify therapy and pursue alternative approaches that may enhance patient outcomes. For instance, in February 2025, according to the United Nations, a US-based intergovernmental organization, an estimated 2.3 million new breast cancer cases were diagnosed in 2022, resulting in 670,000 deaths, and global projections indicate a 38% increase in cases and a 68% rise in annual deaths by 2050. Therefore, the rising incidence of cancer is driving the growth of the checkpoint inhibitor refractory cancer industry.Key Players In The Global Checkpoint Inhibitor Refractory Cancer Market
Major companies operating in the checkpoint inhibitor refractory cancer market are Pfizer Inc., Johnson & Johnson Private Limited, Merck & Co. Inc., AbbVie Inc., Novartis International AG, Sanofi S.A., Bristol-Myers Squibb Company, AstraZeneca PLC, GlaxoSmithKline Plc, Eli Lilly and Company, Gilead Sciences Inc., Amgen Inc., Regeneron Pharmaceuticals Inc., Eisai Co. Ltd., Exelixis Inc., Exicure Inc., ImmunityBio Inc., ENB Therapeutics Inc., Kartos Therapeutics Inc., Pliant Therapeutics Inc.Global Checkpoint Inhibitor Refractory Cancer Market Trends and Insights
Major companies operating in the checkpoint inhibitor refractory cancer market are focusing on developing innovative products, such as dual selective inhibitors, to reduce the likelihood of drug resistance by targeting two critical pathways involved in disease progression. Dual selective inhibitors are compounds that block two distinct biological targets or pathways at the same time while limiting impact on other unrelated pathways. For instance, in March 2025, Pliant Therapeutics Inc., a US-based clinical-stage biopharmaceutical company, announced Interim Phase 1 Data for PLN-101095 in Patients with Immune Checkpoint Inhibitor-Refractory Advanced Solid Tumors. Pliant Therapeutics’ interim Phase 1 data for PLN-101095 in patients with immune checkpoint inhibitor–refractory advanced solid tumors demonstrated promising early activity and a manageable safety profile. At the highest tested dose of 1,000 mg twice daily combined with pembrolizumab, the trial reported a 50% objective response rate, including confirmed partial responses in patients with non-small cell lung cancer, cholangiocarcinoma, and melanoma, showing significant tumor reductions. PLN-101095 was generally well tolerated, with most treatment-emergent adverse events being mild to moderate. The study is ongoing, with higher-dose cohorts currently enrolling and additional data expected by the end of 2025.What Are Latest Mergers And Acquisitions In The Checkpoint Inhibitor Refractory Cancer Market?
In October 2023, Bristol-Myers Squibb Company, a US-based pharmaceutical industry company, acquired Mirati Therapeutics Inc. for an undisclosed amount. Through this acquisition, Bristol Myers Squibb aims to strengthen and diversify its oncology portfolio by integrating Mirati’s innovative cancer therapies and clinical-stage assets, particularly targeting KRAS-mutated cancers and checkpoint inhibitor refractory cancers. Mirati Therapeutics Inc. is a US-based provider of therapies specifically designed for checkpoint inhibitor refractory (resistant) cancers.Regional Insights
North America was the largest region in the checkpoint inhibitor refractory cancer market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Checkpoint Inhibitor Refractory Cancer Market?
The checkpoint inhibitor refractory cancer market includes revenues earned by entities by providing services such as diagnostic and biomarker services, clinical trials and research services, specialized oncology treatment services, and patient support and consulting services. The market value includes the value of related goods sold by the service provider or included within the service offering. The checkpoint inhibitor refractory cancer market consists of sales of tumor-infiltrating lymphocyte (TIL) therapy kits, genomic sequencing panels, tumor organoids, drug discovery tools, and bispecific antibodies. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Checkpoint Inhibitor Refractory Cancer Market Report 2026?
The checkpoint inhibitor refractory cancer market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the checkpoint inhibitor refractory cancer industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Checkpoint Inhibitor Refractory Cancer Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $40.97 billion |
| Revenue Forecast In 2035 | $63.02 billion |
| Growth Rate | CAGR of 11.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Therapy, Cancer Type, Mechanism Of Action, Administration Route, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Johnson & Johnson Private Limited, Merck & Co. Inc., AbbVie Inc., Novartis International AG, Sanofi S.A., Bristol-Myers Squibb Company, AstraZeneca PLC, GlaxoSmithKline Plc, Eli Lilly and Company, Gilead Sciences Inc., Amgen Inc., Regeneron Pharmaceuticals Inc., Eisai Co. Ltd., Exelixis Inc., Exicure Inc., ImmunityBio Inc., ENB Therapeutics Inc., Kartos Therapeutics Inc., Pliant Therapeutics Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
