
Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Report 2026
Global Outlook – By Test Type (Laboratory, Point Of Care Testing), By Product (Assays And Kits, Instruments Or Analyzers), By Technology (Isothermal Nucleic Acid Amplification Technology, Polymerase Chain Reaction, Immunodiagnostics, Other Technologies), By End User (Diagnostic Laboratories, Hospitals And Clinics, Other End Users ) - Market Size, Trends, And Global Forecast 2026-2035
Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Overview
• Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing market size has reached to $2.19 billion in 2025 • Expected to grow to $3.47 billion in 2030 at a compound annual growth rate (CAGR) of 9.3% • Growth Driver: Rising Incidence Of STIs Propels The Growth Of The CTNG Testing Market. • Market Trend: Advancements In Rapid Molecular Point-Of-Care Testing For Stis • North America was the largest region in 2025.What Is Covered Under Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market?
Chlamydia trachomatis (CT) and Neisseria gonorrhea (NG) testing refers to the procedure for diagnosing sexually transmitted infections (STIs) brought on by neisseria gonorrhea (NG) and chlamydia trachomatis (CT) within sexually active individuals, especially if they have multiple partners or engage in unprotected sexual activity. These tests are conducted to detect the presence of these bacteria in individuals, typically through methods such as urine samples, swabs of genital areas, or blood tests. The main testing types for chlamydia trachomatis (CT) and Neisseria gonorrhoeae (NG) testing are laboratory and point-of-care testing. Laboratories are facilities equipped with specialized equipment and trained personnel to perform Chlamydia trachomatis (CT) and Neisseria gonorrhoeae (NG) testing on patient samples. It includes various products, including assays and kits, and instruments or analyzers and it includes different technologies, such as isothermal nucleic acid amplification technology, polymerase chain reaction, immunodiagnostics, and others, also end users are diagnostic laboratories, hospitals, clinics, and others.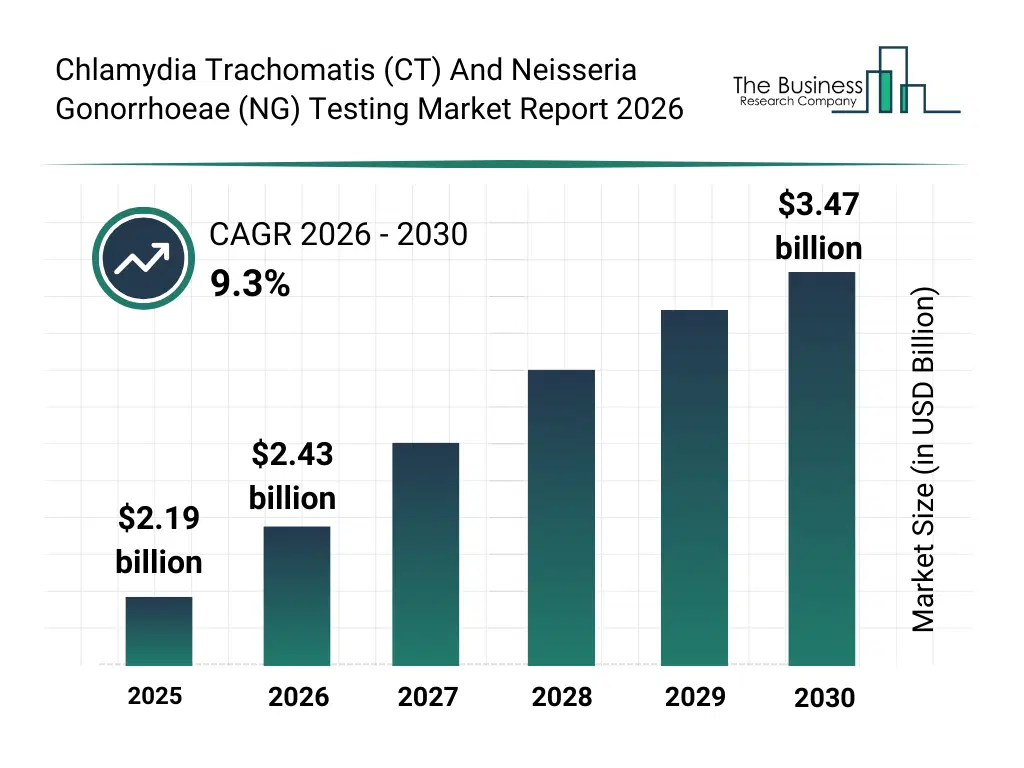
What Is The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Size and Share 2026?
The chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market size has grown rapidly in recent years. It will grow from $2.19 billion in 2025 to $2.43 billion in 2026 at a compound annual growth rate (CAGR) of 10.9%. The growth in the historic period can be attributed to increasing screening of sexually active populations, expansion of laboratory-based testing infrastructure, adoption of NAAT and PCR technologies, rising awareness of co-infection risks, growth of diagnostic laboratories.What Is The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Growth Forecast?
The chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market size is expected to see strong growth in the next few years. It will grow to $3.47 billion in 2030 at a compound annual growth rate (CAGR) of 9.3%. The growth in the forecast period can be attributed to increasing investments in decentralized testing, rising adoption of home-based diagnostics, expansion of automated laboratory platforms, growing focus on early STI detection, increasing demand for integrated CT/NG testing solutions. Major trends in the forecast period include increasing adoption of multiplex sti testing panels, rising demand for rapid point-of-care testing, growing use of self-testing kits, expansion of high-sensitivity molecular assays, enhanced integration of laboratory automation.Global Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Segmentation
1) By Test Type: Laboratory, Point Of Care Testing 2) By Product: Assays And Kits, Instruments Or Analyzers 3) By Technology: Isothermal Nucleic Acid Amplification Technology, Polymerase Chain Reaction, Immunodiagnostics, Other Technologies 4) By End User: Diagnostic Laboratories, Hospitals And Clinics, Other End Users Subsegments: 1) By Laboratory: Nucleic Acid Amplification Tests (NAAT), Polymerase Chain Reaction (PCR) Tests, Enzyme-Linked Immunosorbent Assay (ELISA), Immunofluorescence Assay (IFA), Culture Tests, Direct Fluorescent Antibody (DFA) Tests 2) By Point Of Care Testing: Rapid Diagnostic Tests (RDT), Lateral Flow Assays, Immunochromatographic Tests, Self-Testing Kits, Loop-Mediated Isothermal Amplification (LAMP), Microfluidics-Based TestsWhat Is The Driver Of The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market?
The increasing number of sexually transmitted infections (STIs) is expected to propel the growth of the chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing market going forward. Sexually transmitted infections (STIs) refer to diseases caused by infection with bacteria, viruses, or parasites that can be transmitted through sexual contact. STI rates are rising due to a lack of comprehensive sex education, increased sexual activity, stigma, and barriers. Chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing is used for detecting and managing sexually transmitted infections (STIs) by enabling early diagnosis, treatment, and prevention of transmission. For instance, in October 2025, according to the Minority HIV/AIDS Fund (MHAF), an US-based government agency, an estimated in 2023, approximately 39.9 million individuals worldwide were living with HIV, consisting of 38.6 million adults and 1.4 million children, compared to previous years. Therefore, there is an increasing number of sexually transmitted infections, which is driving the growth of the CTNG testing market.Key Players In The Global Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market
Major companies operating in the chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market are F. Hoffmann-La Roche AG, Thermo Fisher Scientific, Abbott Laboratories, Danaher Corporation, Merck KGaA, Siemens Healthineers, Becton Dickinson and Company, Grifols SA, Hologic Inc, PerkinElmer, bioMérieux, Sysmex Corporation, Bio-Rad Laboratories, QIAGEN, Quidel Corporation, DiaSorin, Cepheid, Seegene Inc, Meridian Bioscience Inc, Sekisui Diagnostics, J. Mitra & Co. Pvt. Ltd, Atlas Medical, Hardy Diagnostics, Molbio Diagnostics Pvt. Ltd, NG BiotechGlobal Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Trends and Insights
Major companies operating in the chlamydia market are focusing on developing innovative solutions such as Point-of-Care Molecular Diagnostics Expansion to enable faster, more accurate onsite detection and improve timely treatment outcomes. Point-of-care molecular diagnostics expansion refers to the broader adoption of rapid, onsite molecular tests that allow immediate detection of infections such as chlamydia without relying on centralized laboratories. For instance, in January 2025, F. Hoffmann-La Roche AG, a Switzerland-based healthcare company, received FDA 510(k) clearance along with a CLIA waiver for its cobas® liat molecular tests, which are designed to diagnose sexually transmitted infections (STIs) at the point of care. These tests include multiplex assay panels that can detect and differentiate multiple STIs, such as chlamydia, gonorrhea, and Mycoplasma genitalium, from a single patient sample with results available in 20 minutes or less.What Are Latest Mergers And Acquisitions In The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market?
In December 2023, Imaware, a US-based health testing company, acquired consumer testing business from Binx Health for an undisclosed amount. This acquisition allows Imaware to expand its portfolio in consumer diagnostics, broadening access to STI screening and other health tests. Binx Health is a US-based company that specializes in at-home and point-of-care diagnostic testing solutions, particularly in sexual health.Regional Outlook
North America was the largest region in the chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market?
The chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing market includes revenues earned by entities by providing services such as diagnostic testing, sample collection, laboratory analysis, and confidential counseling. The market value includes the value of related goods sold by the service provider or included within the service offering. The chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing market also includes the sale of sample collection kits, control materials, laboratory equipment, and software solutions. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Report 2026?
The chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.43 billion |
| Revenue Forecast In 2035 | $3.47 billion |
| Growth Rate | CAGR of 10.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Product, Technology, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Thermo Fisher Scientific, Abbott Laboratories, Danaher Corporation, Merck KGaA, Siemens Healthineers, Becton Dickinson and Company, Grifols SA, Hologic Inc, PerkinElmer, bioMérieux, Sysmex Corporation, Bio-Rad Laboratories, QIAGEN, Quidel Corporation, DiaSorin, Cepheid, Seegene Inc, Meridian Bioscience Inc, Sekisui Diagnostics, J. Mitra & Co. Pvt. Ltd, Atlas Medical, Hardy Diagnostics, Molbio Diagnostics Pvt. Ltd, NG Biotech |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
