
Claudin 18.2 Targeted Therapy Market Report 2026
Global Outlook – By Therapy Type (Monoclonal Antibodies, Bispecific Antibodies, Antibody-Drug Conjugates, Other Therapy Types), By Indication (Gastric Cancer, Pancreatic Cancer, Esophageal Cancer, Other Indications), By Route Of Administration (Intravenous, Oral, Other Route Of Administrations), By End-User (Hospitals, Oncology Centers, Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Claudin 18.2 Targeted Therapy Market Overview
• Claudin 18.2 Targeted Therapy market size has reached to $1.6 billion in 2025 • Expected to grow to $4.61 billion in 2030 at a compound annual growth rate (CAGR) of 23.5% • Growth Driver: Rising Gastrointestinal Cancer Incidence Driving The Market Growth Due To Increasing Prevalence And Unhealthy Diets • Market Trend: Prominent Companies Pioneering Targeted Oncology Solutions • Asia-Pacific was the largest region in 2025.What Is Covered Under Claudin 18.2 Targeted Therapy Market?
Claudin 18.2 targeted therapy is an advanced precision cancer treatment designed to specifically target the Claudin 18.2 protein, abnormally overexpressed on the surface of certain tumor cells. This therapy selectively attacks cancer cells, improving treatment efficacy and reducing damage to healthy tissues. The main therapy types of claudin 18.2 targeted therapy are monoclonal antibodies, bispecific antibodies, antibody-drug conjugates, and others. Monoclonal antibodies are lab-made molecules designed to attach to specific proteins on cancer cells, helping the immune system target and destroy them. These therapies are mainly indicated for gastric cancer, pancreatic cancer, esophageal cancer, and others, and are administered via intravenous, oral, and other routes. The therapies are primarily used by end-users, including hospitals, oncology centers, research institutes, and others.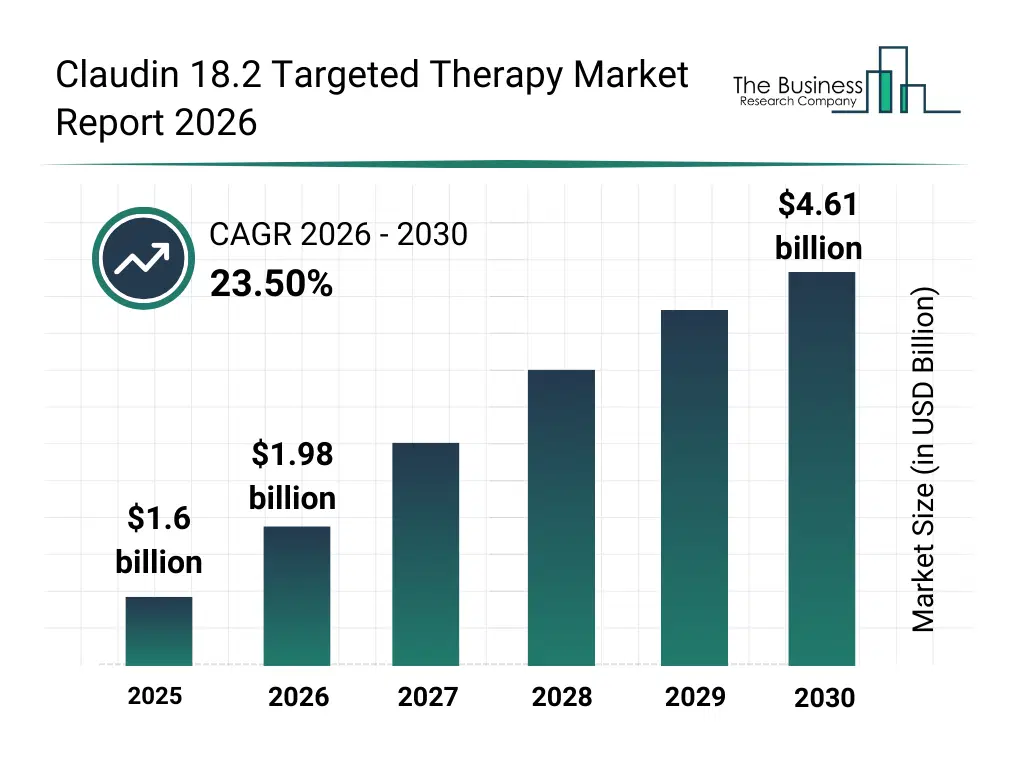
What Is The Claudin 18.2 Targeted Therapy Market Size and Share 2026?
The claudin 18.2 targeted therapy market size has grown exponentially in recent years. It will grow from $1.6 billion in 2025 to $1.98 billion in 2026 at a compound annual growth rate (CAGR) of 23.7%. The growth in the historic period can be attributed to increasing prevalence of gastric and gastrointestinal cancers, advancements in antibody engineering technologies, expansion of oncology-focused clinical trials, rising investments in targeted cancer therapies, improved understanding of claudin protein biology.What Is The Claudin 18.2 Targeted Therapy Market Growth Forecast?
The claudin 18.2 targeted therapy market size is expected to see exponential growth in the next few years. It will grow to $4.61 billion in 2030 at a compound annual growth rate (CAGR) of 23.5%. The growth in the forecast period can be attributed to increasing regulatory approvals for targeted oncology drugs, rising demand for personalized cancer treatment, expansion of combination therapy approaches, growing oncology pipeline investments, increasing adoption of precision diagnostics. Major trends in the forecast period include increasing focus on biomarker-driven oncology therapies, rising development of claudin 18.2 monoclonal antibodies, growing expansion of indication-specific targeted treatments, enhanced integration of companion diagnostics, increasing strategic collaborations in precision oncology.Global Claudin 18.2 Targeted Therapy Market Segmentation
1) By Therapy Type: Monoclonal Antibodies, Bispecific Antibodies, Antibody-Drug Conjugates, Other Therapy Types 2) By Indication: Gastric Cancer, Pancreatic Cancer, Esophageal Cancer, Other Indications 3) By Route Of Administration: Intravenous, Oral, Other Route Of Administrations 4) By End-User: Hospitals, Oncology Centers, Research Institutes, Other End-Users Subsegments: 1) By Monoclonal Antibodies: Chimeric Monoclonal Antibodies, Humanized Monoclonal Antibodies, Fully Human Monoclonal Antibodies 2) By Bispecific Antibodies: Igg-Like Bispecific Antibodies, Non-Igg-Like Bispecific Antibodies 3) By Antibody-Drug Conjugates: Microtubule Inhibitor-Based Antibody-Drug Conjugates, Dna Damaging Agent-Based Antibody-Drug Conjugates, Topoisomerase Inhibitor-Based Antibody-Drug Conjugates 4) By Other Therapy Types: Car-T Cell Therapy, Vaccines, Small Molecule InhibitorsWhat Is The Driver Of The Claudin 18.2 Targeted Therapy Market?
The rising incidence of gastrointestinal cancers is expected to propel the growth of the claudin 18.2 targeted therapy market going forward. Gastrointestinal (GI) cancers refer to malignant tumors that develop in any part of the digestive tract, from the esophagus to the rectum. The rise in gastrointestinal cancers is largely due to unhealthy diets rich in processed foods and low in fiber, promoting gut inflammation and increasing cancer risk. Claudin 18.2 targeted therapy helps treat gastrointestinal cancers by selectively targeting the Claudin 18.2 protein, overexpressed on cancer cells, enabling drugs or antibodies to kill cancer cells while sparing healthy tissue and improving treatment effectiveness. For instance, in June 2025, according to the Centers for Disease Control and Prevention (CDC), a US-based public health agency, 147,931 new colorectal cancer cases were reported in 2022, and 53,779 deaths from colorectal cancer were recorded in 2023. Therefore, the rising incidence of gastrointestinal cancers is driving the growth of the Claudin 18.2 targeted therapy industry.Key Players In The Global Claudin 18.2 Targeted Therapy Market
Major companies operating in the claudin 18.2 targeted therapy market are Bristol Myers Squibb Company, Amgen Inc., Astellas Pharma Inc., Jiangsu Hengrui Medicine Co. Ltd., Shanghai Henlius Biotech Inc., Legend Biotech Corporation, Zai Lab Limited, Harbour Biomed Ltd., CARsgen Therapeutics Holdings Limited, Genor Biopharma Co. Ltd., Phanes Therapeutics Inc., Suzhou Zelgen Biopharmaceuticals Co. Ltd., Triumvira Immunologics Inc., SOTIO Biotech a.s., Leap Therapeutics, Allogene Therapeutics, AskGene Pharma Inc., Elpiscience Biopharmaceutical Co. Ltd., I-Mab Biopharma Co. Ltd., Antegene Biopharma Co. Ltd.Global Claudin 18.2 Targeted Therapy Market Trends and Insights
Major companies operating in the claudin 18.2 targeted therapy market are focusing on developing innovative solutions, such as monoclonal antibodies, to specifically target claudin 18.2-expressing cancer cells and enhance treatment outcomes. A monoclonal antibody is a laboratory-engineered protein designed to bind precisely to a single target, such as a protein on the surface of cancer cells, aiding the immune system in recognizing and attacking these cells. For instance, in March 2024, Astellas Pharma Inc., a Japan-based pharmaceutical company, obtained approval from Japan's Ministry of Health, Labour and Welfare for VYLOY (zolbetuximab) for the treatment of patients with CLDN18.2-positive cancer. This targeted therapy for advanced gastric and gastroesophageal junction cancer selectively binds to the CLDN18.2 protein on tumor cells, activating the immune system to destroy cancer cells. Clinical trials showed it significantly improves progression-free and overall survival when combined with chemotherapy compared to chemotherapy alone. This innovative therapy provides a new, effective option for patients with HER2-negative, CLDN18.2-positive tumors, fulfilling an unmet need in aggressive gastric cancer.What Are Latest Mergers And Acquisitions In The Claudin 18.2 Targeted Therapy Market?
In February 2024, AstraZeneca plc, a UK-based pharmaceutical industry company, acquired Gracell Biotechnologies Inc. for an undisclosed amount. With this acquisition, AstraZeneca aims to accelerate its cell therapy strategy by expanding its pipeline with Gracell's innovative CAR-T therapies targeting blood cancers and autoimmune diseases, leveraging Gracell’s FasTCAR platform to improve treatment efficacy and manufacturing speed. Gracell Biotechnologies Inc. is a China-based clinical-stage biopharmaceutical company that offers claudin 18.2 targeted therapy.Regional Insights
Asia-Pacific was the largest region in the claudin 18.2 targeted therapy market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Claudin 18.2 Targeted Therapy Market?
The claudin 18.2 targeted therapy market consists of sales of zolbetuximab, CMG901, and bispecific antibodies. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Claudin 18.2 Targeted Therapy Market Report 2026?
The claudin 18.2 targeted therapy market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the claudin 18.2 targeted therapy industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Claudin 18.2 Targeted Therapy Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.98 billion |
| Revenue Forecast In 2035 | $4.61 billion |
| Growth Rate | CAGR of 23.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Therapy Type, Indication, Route Of Administration, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Bristol Myers Squibb Company, Amgen Inc., Astellas Pharma Inc., Jiangsu Hengrui Medicine Co. Ltd., Shanghai Henlius Biotech Inc., Legend Biotech Corporation, Zai Lab Limited, Harbour Biomed Ltd., CARsgen Therapeutics Holdings Limited, Genor Biopharma Co. Ltd., Phanes Therapeutics Inc., Suzhou Zelgen Biopharmaceuticals Co. Ltd., Triumvira Immunologics Inc., SOTIO Biotech a.s., Leap Therapeutics, Allogene Therapeutics, AskGene Pharma Inc., Elpiscience Biopharmaceutical Co. Ltd., I-Mab Biopharma Co. Ltd., Antegene Biopharma Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
