
Clinical Reference Laboratory Market Report 2026
Global Outlook – By Type (Clinical Chemistry, Genetics Testing, Microbiology And Cytology, Other Types), By Technology (Traditional Laboratory Techniques, Digital Technologies, Automated Testing Platforms, Molecular Diagnostics), By Application (Clinics, Hospitals, Independent Laboratory) – Market Size, Trends, Strategies, and Forecast to 2035
Clinical Reference Laboratory Market Overview
• Clinical Reference Laboratory market size has reached to $47 billion in 2025 • Expected to grow to $64.93 billion in 2030 at a compound annual growth rate (CAGR) of 6.6% • Growth Driver: Rise In Demand For Diagnostic Testing Driving The Growth Of The Market Due To Increasing Prevalence Of Chronic And Infectious Diseases • Market Trend: Advancements In Automated Sample Processing Driving Efficiency And Accuracy In Diagnostic Laboratories • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Reference Laboratory Market?
A clinical reference laboratory is a specialized medical laboratory that performs high-complexity diagnostic testing and analysis on biological specimens such as blood, urine, and tissue samples. These laboratories offer advanced testing services, including molecular diagnostics, toxicology, immunology, and pathology. They are equipped with state-of-the-art technology and adhere to stringent regulatory standards to ensure accuracy, reliability, and compliance with healthcare regulations. The main clinical reference laboratory types include clinical chemistry, genetics testing, microbiology and cytology, and others. Clinical chemistry or clinical biochemistry is a specialized area of clinical pathology that involves the quantitative analysis of bodily fluids such as blood and urine to assess the concentration of various chemical substances. It uses various technologies such as traditional laboratory techniques, digital technologies, automated testing platforms, and molecular diagnostics and is applied by clinics, hospitals, and independent laboratories.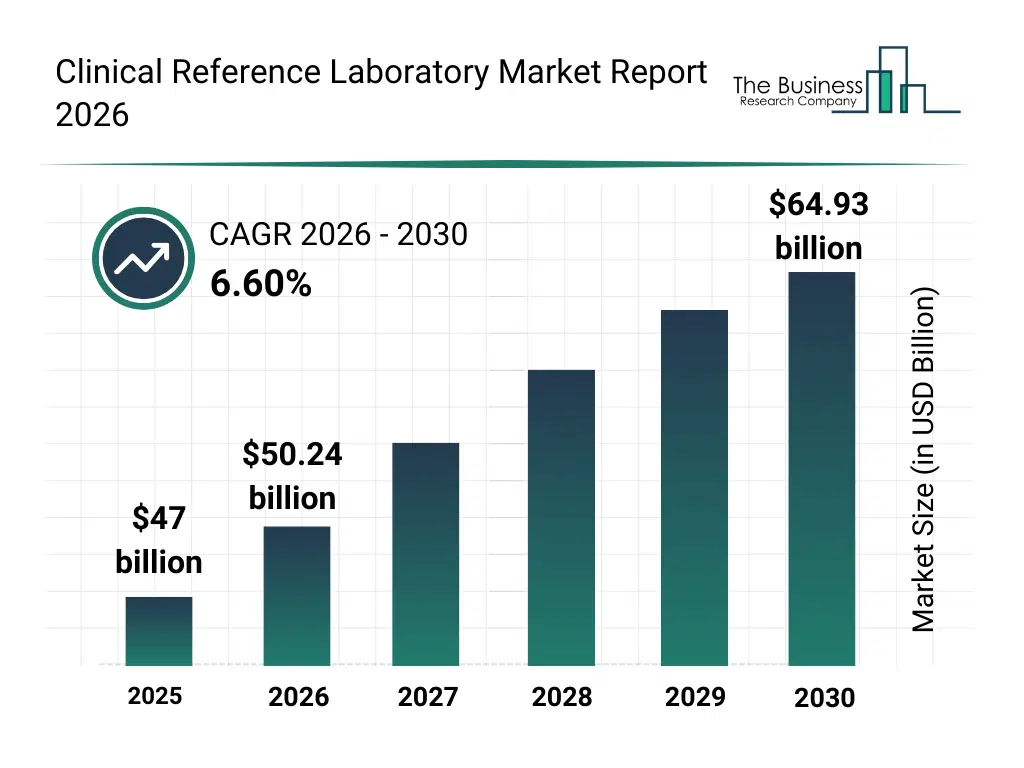
What Is The Clinical Reference Laboratory Market Size and Share 2026?
The clinical reference laboratory market size has grown strongly in recent years. It will grow from $47 billion in 2025 to $50.24 billion in 2026 at a compound annual growth rate (CAGR) of 6.9%. The growth in the historic period can be attributed to chronic disease prevalence, hospital diagnostic outsourcing, infectious disease testing demand, regulatory compliance needs, laboratory consolidation.What Is The Clinical Reference Laboratory Market Growth Forecast?
The clinical reference laboratory market size is expected to see strong growth in the next few years. It will grow to $64.93 billion in 2030 at a compound annual growth rate (CAGR) of 6.6%. The growth in the forecast period can be attributed to genomic testing expansion, AI-based diagnostics adoption, personalized medicine growth, healthcare digitization, population health screening demand. Major trends in the forecast period include expansion of molecular diagnostics, automation of laboratory testing, growth of high-complexity testing, demand for rapid diagnostic turnaround, centralization of diagnostic services.Global Clinical Reference Laboratory Market Segmentation
1) By Type: Clinical Chemistry, Genetics Testing, Microbiology And Cytology, Other Types 2) By Technology: Traditional Laboratory Techniques, Digital Technologies, Automated Testing Platforms, Molecular Diagnostics 3) By Application: Clinics, Hospitals, Independent Laboratory Subsegments: 1) By Clinical Chemistry: Blood Gas Analysis, Enzyme Testing, Electrolyte Testing, Lipid Panel, Liver Function Testing, Renal Function Testing 2) By Genetics Testing: Carrier Screening, Diagnostic Testing, Predictive And Presymptomatic Testing, Pharmacogenomics, Prenatal And Newborn Testing 3) By Microbiology And Cytology: Bacterial Culture Testing, Fungal Culture Testing, Viral Testing, Parasitology Testing, Cytopathology 4) By Other Types: Immunology Testing, Hematology Testing, Toxicology Testing, Endocrinology TestingWhat Is The Driver Of The Clinical Reference Laboratory Market?
The increasing demand for diagnostic testing is expected to propel the growth of the clinical reference laboratory market going forward. Diagnostic testing analyzes biological samples to detect, diagnose, and monitor diseases, medical conditions, or infections. The rise in diagnostic testing is due to advancements in medical technology, which have made tests more accessible, accurate, and efficient. Clinical reference laboratories are essential for diagnostic testing to provide highly specialized and precise analysis using advanced technology and standardized protocols, ensuring reliable test results that aid in early disease detection, treatment planning, and patient monitoring across various medical fields. For instance, in April 2024, according to a report published by the National Health Service (NHS), a UK-based publicly funded healthcare system, in April 2024, 376,200 patients were waiting over six weeks for a key diagnostic test, reflecting an increase of 21,200 from the previous month. Therefore, the increasing demand for diagnostic testing will fuel the growth of the clinical reference laboratory industry.Key Players In The Global Clinical Reference Laboratory Market
Major companies operating in the clinical reference laboratory market are Mayo Clinic Laboratories, Eurofins Scientific, Quest Diagnostics, Sonic Healthcare, Unilabs, ARUP Laboratories, Lancet Laboratories, NeoGenomics Laboratories, Ampath Laboratories, Clinical Reference Laboratory, Al Borg Medical Laboratories, Medsol Diagnostics, Medlabs Group, Cerba International, Lifebrain Group, CRL Diagnostics, Dr. Lal PathLabs, SRL Diagnostics, Synlab International, ACM Global Laboratories, Laboratory Corporation of America (Labcorp), KingMed DiagnosticsGlobal Clinical Reference Laboratory Market Trends and Insights
Major companies operating in the clinical reference laboratory market are focusing on developing technologically advanced products, such as automated sample processing platforms, to enhance efficiency, accuracy, and scalability. Automated sample processing platforms are high-throughput laboratory systems that use robotics and advanced software to handle, prepare, and analyze biological samples with minimal human intervention, ensuring efficiency, accuracy, and standardization in diagnostic and research workflows. For instance, in January 2023, QIAGEN NV, a Germany-based provider of sample and assay technologies for molecular diagnostics, launched the EZ2 Connect MDx platform for diagnostic laboratories. The EZ2 Connect MDx is an IVD platform designed for automated sample processing, enabling laboratories to efficiently purify DNA and RNA from 24 samples in parallel in as little as 30 minutes. It features magnetic-bead technology for nucleic acid extraction from various sample types, including blood plasma, serum, and stool. The platform utilizes prefilled and sealed reagent cartridges, integrated cameras for load checking, and UV decontamination to ensure high process safety and minimize contamination risks. Additionally, it is equipped with onboard pipetting and heating, along with automated piercing of sealed cartridges for seamless operation. The resulting analyte is compatible with multiple downstream technologies, such as real-time PCR, digital PCR, and next-generation sequencing.What Are Latest Mergers And Acquisitions In The Clinical Reference Laboratory Market?
In April 2024, Eurofins Scientific, a Luxembourg-based testing laboratory company, acquired Ascend Clinical LLC for an undisclosed amount. Through this acquisition, Eurofins Scientific aims to expand its footprint in renal and transplant testing by integrating Ascend Clinical's expertise, advanced laboratory services and established client base with Eurofins' extensive network, technological capabilities, and industry relationships to enhance diagnostic offerings, improve patient care, and drive growth in the clinical diagnostics sector across the United States. Ascend Clinical LLC is a US-based clinical reference laboratory testing and services provider.Regional Outlook
North America was the largest region in the clinical reference laboratory market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Clinical Reference Laboratory Market?
The clinical reference laboratory market consists of revenues earned by entities by providing services such as pathology services, biomarker testing and pharmacogenomics. The market value includes the value of related goods sold by the service provider or included within the service offering. The clinical reference laboratory market also includes sales of diagnostic test kits, blood collection tubes, microbiology culture media and immunoassay reagents. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Clinical Reference Laboratory Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $50.24 billion |
| Revenue Forecast In 2035 | $64.93 billion |
| Growth Rate | CAGR of 6.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Technology, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Mayo Clinic Laboratories, Eurofins Scientific, Quest Diagnostics, Sonic Healthcare, Unilabs, ARUP Laboratories, Lancet Laboratories, NeoGenomics Laboratories, Ampath Laboratories, Clinical Reference Laboratory, Al Borg Medical Laboratories, Medsol Diagnostics, Medlabs Group, Cerba International, Lifebrain Group, CRL Diagnostics, Dr. Lal PathLabs, SRL Diagnostics, Synlab International, ACM Global Laboratories, Laboratory Corporation of America (Labcorp), KingMed Diagnostics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
