Clinical Reference Laboratory Service Market Report 2026

Clinical Reference Laboratory Service Market Report 2026
Global Outlook – By Service Type (Clinical Chemistry, Hematology, Microbiology, Cytology, Genetic Testing, Other Service Types), By Sample Type (Blood Samples, Urine Samples, Tissue Samples, Saliva Samples, Other Sample Types), By Technology (Laboratory Information Systems (LIS), Automated Laboratory Equipment, High-Throughput Centralized Testing Platforms, Other Technologies), By Application (Disease Diagnosis, Drug Development, Precision Medicine, Other Applications ), By End User (Hospitals, Diagnostic Laboratories, Academic Institutes, Pharmaceutical Companies) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Reference Laboratory Service Market Overview
• Clinical Reference Laboratory Service market size has reached to $18.98 billion in 2025 • Expected to grow to $29.87 billion in 2030 at a compound annual growth rate (CAGR) of 9.4% • Growth Driver: Rising Demand For Personalized Medicine Fueling The Growth Of The Market Due To Increasing Need For Tailored Genetic Testing And Treatment • Market Trend: Advancements In Laboratory Automation Fully Automated Digi-Smart Reference Lab • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Reference Laboratory Service Market?
Clinical reference laboratory service refers to a specialized laboratory service that provides diagnostic testing and analytical services for healthcare providers, hospitals, clinics, and other laboratories. These services are typically performed by large, centralized laboratories that offer a wide array of high-complexity and specialty tests, which may not be available in smaller or on-site hospital labs. The main types of clinical reference laboratory service are clinical chemistry, hematology, microbiology, cytology, genetic testing, and others. Clinical Chemistry refers to a branch of laboratory medicine that focuses on the analysis of bodily fluids, primarily blood and urine, to diagnose and monitor diseases. The multiple sample types involved are blood samples, urine samples, tissue samples, saliva samples, and others are used for various technologies, including laboratory information systems (LIS), automated laboratory equipment, point-of-care testing, and others. The various applications include disease diagnosis, drug development, precision medicine, and others, and cater to several end users such as hospitals, diagnostic laboratories, academic institutes, and pharmaceutical companies.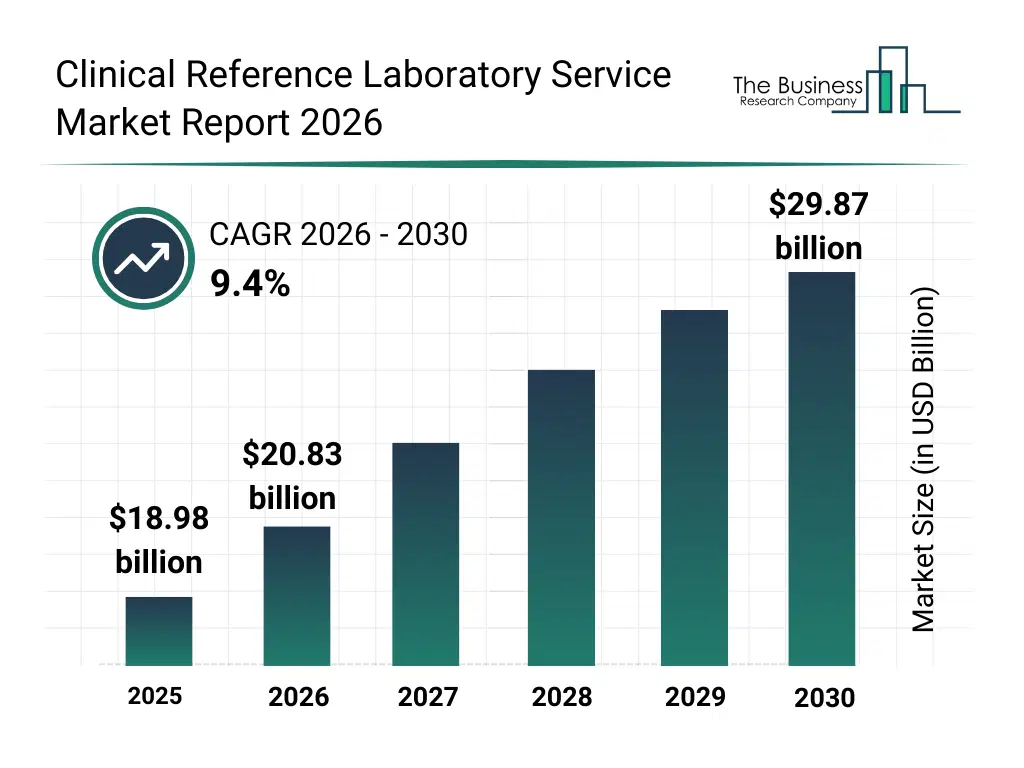
What Is The Clinical Reference Laboratory Service Market Size and Share 2026?
The clinical reference laboratory service market size has grown strongly in recent years. It will grow from $18.98 billion in 2025 to $20.83 billion in 2026 at a compound annual growth rate (CAGR) of 9.7%. The growth in the historic period can be attributed to increasing outsourcing of diagnostic testing, expansion of centralized laboratory networks, rising demand for specialty diagnostics, growth in hospital referral testing volumes, availability of advanced laboratory technologies.What Is The Clinical Reference Laboratory Service Market Growth Forecast?
The clinical reference laboratory service market size is expected to see strong growth in the next few years. It will grow to $29.87 billion in 2030 at a compound annual growth rate (CAGR) of 9.4%. The growth in the forecast period can be attributed to increasing adoption of precision medicine diagnostics, expansion of genomic and molecular testing services, rising investments in laboratory automation, growing demand for high-throughput testing capacity, increasing integration of digital laboratory platforms. Major trends in the forecast period include increasing centralization of high-complexity diagnostic testing, rising adoption of automated reference laboratory systems, growing demand for specialized and genetic testing services, expansion of integrated laboratory information systems, enhanced focus on quality and regulatory compliance.Global Clinical Reference Laboratory Service Market Segmentation
1) By Service Type: Clinical Chemistry, Hematology, Microbiology, Cytology, Genetic Testing, Other Service Types 2) By Sample Type: Blood Samples, Urine Samples, Tissue Samples, Saliva Samples, Other Sample Types 3) By Technology: Laboratory Information Systems (LIS), Automated Laboratory Equipment, High-Throughput Centralized Testing Platforms, Other Technologies 4) By Application: Disease Diagnosis, Drug Development, Precision Medicine, Other Applications 5) By End User: Hospitals, Diagnostic Laboratories, Academic Institutes, Pharmaceutical Companies Subsegments: 1) By Clinical Chemistry: Blood Glucose Testing, Electrolyte Panels, Liver Function Tests, Renal Function Panels 2) By Hematology: Complete Blood Count (CBC), Coagulation Tests, Hemoglobin Testing, Erythrocyte Sedimentation Rate (ESR) 3) By Microbiology: Bacterial Culture And Sensitivity Testing, Viral Detection Assays, Fungal Diagnostics, Parasitology Tests 4) By Cytology: Pap Smears, Fine-Needle Aspiration Cytology (FNAC), Fluid Cytology (E.G., Pleural, Ascitic), Liquid-Based Cytology 5) By Genetic Testing: Carrier Screening, Predictive And Pre-Symptomatic Testing, Pharmacogenomics, Whole Genome Or Exome Sequencing 6) By Other Service Types: Immunology Or Serology, Toxicology Screening, Allergy Testing, Drug Of Abuse TestingWhat Is The Driver Of The Clinical Reference Laboratory Service Market?
The increasing demand for personalized medicine is expected to propel the growth of the clinical reference laboratory service market going forward. Personalized medicine refers to a medical model that uses information about a person’s genes and lifestyle environment to tailor treatments and healthcare to their specific needs. The rise of personalized medicine is due to people preferring more tailored and individualized healthcare that considers their unique genetic makeup and environmental factors. Clinical reference laboratory services help personalize medicine by allowing precise genetic testing and pharmacogenomic analysis to tailor treatments to individual genetic profiles. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based nonprofit organization, the U.S. Food and Drug Administration (FDA) approved 26 new personalized medicines in 2023, a significant increase from the 12 approved in 2022. Therefore, the increasing demand for personalized medicine is driving the growth of the clinical reference laboratory service industry.Key Players In The Global Clinical Reference Laboratory Service Market
Major companies operating in the clinical reference laboratory service market are Laboratory Corporation of America Holdings, Mayo Clinic Laboratories, Quest Diagnostics Incorporated, Eurofins Scientific SE, Sonic Healthcare Limited, SYNLAB International GmbH, OPKO Health Inc, Unilabs AB, LifeLabs Medical Laboratory Services, ARUP Laboratories, Dr Lal PathLabs Limited, PathGroup Inc, Clinical Reference Laboratory Inc, ACCU Reference Medical Lab LLC, ACM Global Laboratories, Genova Diagnostics Inc, Cerba HealthCare Group, MedLabs Diagnostics, Apollo Diagnostics, Health Services LaboratoriesGlobal Clinical Reference Laboratory Service Market Trends and Insights
Major companies operating in the clinical reference laboratory service market are focusing on advancements in diagnostic technologies such as next-generation laboratory automation to enhance testing accuracy, operational efficiency, and turnaround time. Next-generation laboratory automation refers to the integration of advanced technologies such as robotics, artificial intelligence (AI), machine learning, and data analytics to streamline and optimize laboratory workflows. For instance, in May 2025, Apollo Diagnostics, an India-based provider of comprehensive diagnostic and pathology testing services, launched the fully automated Digi-Smart central reference laboratory in Chennai. The new facility, spanning 45,000 square feet, is fully automated and aims to reduce laboratory sample turnaround time (TAT) by 60%. It offers a range of advanced features designed to enhance diagnostic efficiency and precision. Equipped with cutting-edge robotics and next-generation laboratory automation, the facility ensures seamless sample processing with minimal human intervention. It integrates AI-powered diagnostic tools, real-time data tracking, and end-to-end digital workflow management, resulting in faster turnaround times, improved accuracy, and reduced errors. The lab also supports high-throughput testing, enabling it to handle large volumes of diagnostic samples efficiently while maintaining stringent quality standards.What Are Latest Mergers And Acquisitions In The Clinical Reference Laboratory Service Market?
In April 2024, Eurofins Scientific SE, a Luxembourg-based testing laboratory company, acquired Ascend Clinical for an undisclosed amount. Through this acquisition, Eurofins aims to strengthen its footprint in the U.S. renal and transplant diagnostics market by integrating Ascend Clinical’s highly automated laboratory services for kidney dialysis and related testing. Ascend Clinical LLC is a US-based clinical reference laboratory company.Regional Outlook
North America was the largest region in the clinical reference laboratory service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Reference Laboratory Service Market?
The clinical reference laboratory service market consists of revenues earned by entities by providing services such as routine clinical testing, microbiology and infectious disease testing, molecular diagnostics, immunology and serology, and toxicology and drug testing. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Reference Laboratory Service Market Report 2026?
The clinical reference laboratory service market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical reference laboratory service industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Reference Laboratory Service Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $20.83 billion |
| Revenue Forecast In 2035 | $29.87 billion |
| Growth Rate | CAGR of 9.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Sample Type, Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Laboratory Corporation of America Holdings, Mayo Clinic Laboratories, Quest Diagnostics Incorporated, Eurofins Scientific SE, Sonic Healthcare Limited, SYNLAB International GmbH, OPKO Health Inc, Unilabs AB, LifeLabs Medical Laboratory Services, ARUP Laboratories, Dr Lal PathLabs Limited, PathGroup Inc, Clinical Reference Laboratory Inc, ACCU Reference Medical Lab LLC, ACM Global Laboratories, Genova Diagnostics Inc, Cerba HealthCare Group, MedLabs Diagnostics, Apollo Diagnostics, Health Services Laboratories |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Clinical Reference Laboratory Service market was valued at $18.98 billion in 2025, increased to $20.83 billion in 2026, and is projected to reach $29.87 billion by 2030.
The global Clinical Reference Laboratory Service market is expected to grow at a CAGR of 9.4% from 2026 to 2035 to reach $29.87 billion by 2035.
Some Key Players in the Clinical Reference Laboratory Service market Include, Laboratory Corporation of America Holdings, Mayo Clinic Laboratories, Quest Diagnostics Incorporated, Eurofins Scientific SE, Sonic Healthcare Limited, SYNLAB International GmbH, OPKO Health Inc, Unilabs AB, LifeLabs Medical Laboratory Services, ARUP Laboratories, Dr Lal PathLabs Limited, PathGroup Inc, Clinical Reference Laboratory Inc, ACCU Reference Medical Lab LLC, ACM Global Laboratories, Genova Diagnostics Inc, Cerba HealthCare Group, MedLabs Diagnostics, Apollo Diagnostics, Health Services Laboratories .
Major trend in this market includes: Advancements In Laboratory Automation Fully Automated Digi-Smart Reference Lab. For further insights on this market.
Request for SampleNorth America was the largest region in the clinical reference laboratory service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical reference laboratory service market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
