
Clinical Trial Biorepository And Archiving Solutions Market Report 2026
Global Outlook – By Product (Preclinical Products, Clinical Products), By Service (Biorepository Services, Archiving Solution Services), By Application (Research And Development, Clinical Trials, Regulatory Compliance, Drug Development), By End User (Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations, Academic Institutions) – Market Size, Trends, Strategies, and Forecast to 2035
Clinical Trial Biorepository And Archiving Solutions Market Overview
• Clinical Trial Biorepository And Archiving Solutions market size has reached to $4.43 billion in 2025 • Expected to grow to $6.54 billion in 2030 at a compound annual growth rate (CAGR) of 8.1% • Growth Driver: Rising Clinical Trials Driving The Growth Of The Market Due To Advancements In Biotechnology And Regulatory Support • Market Trend: Technological Advancements In Biorepository Facilities Enhancing Clinical Trial Storage And Data Management • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Biorepository And Archiving Solutions Market?
Clinical trial biorepository and archiving solutions are services that manage the collection, storage, preservation, and retrieval of biological samples, clinical data, and associated documentation during and after clinical trials. These solutions ensure biospecimens are stored under regulated conditions, maintaining their integrity for future research, regulatory audits, or additional analysis. The main clinical trial biorepository and archiving solution products are preclinical products and clinical products. Preclinical products refer to drug candidates, therapies, or medical devices that are in the research and testing phase before being tested in human clinical trials. These products undergo laboratory and animal testing to assess safety, efficacy, and potential side effects. Services offered include biorepository and archiving solutions. Key applications include research and development, clinical trials, regulatory compliance, and drug development. The solutions cater to various end uses, including pharmaceutical companies, biotechnology firms, contract research organizations, and academic institutions.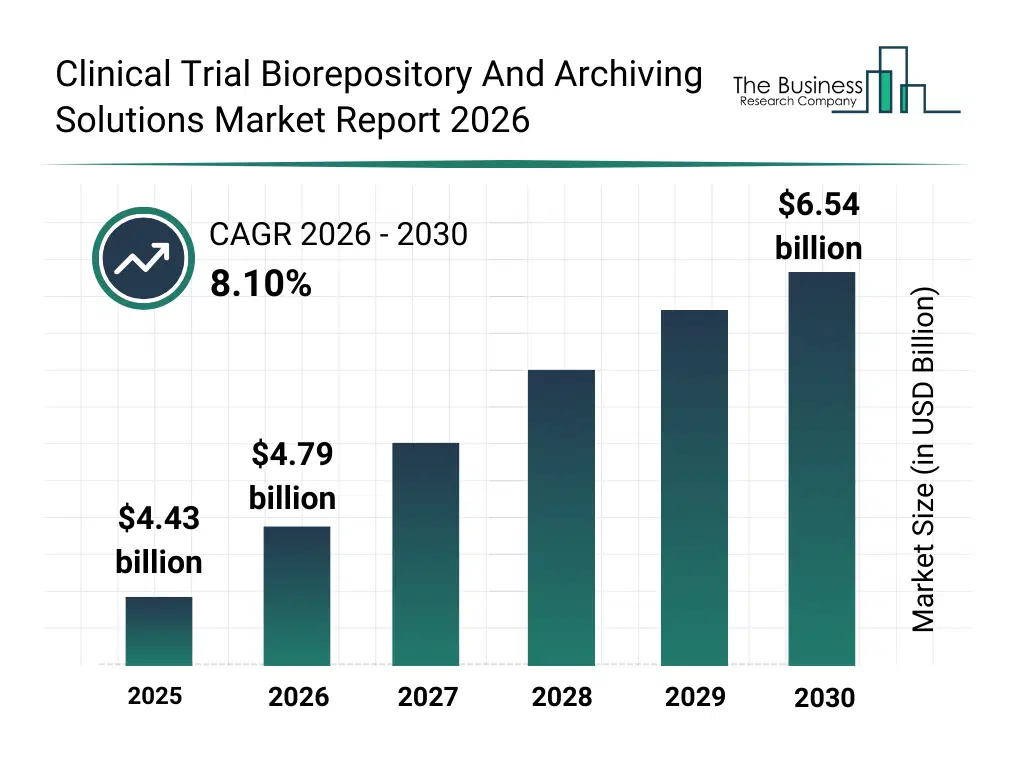
What Is The Clinical Trial Biorepository And Archiving Solutions Market Size and Share 2026?
The clinical trial biorepository and archiving solutions market size has grown strongly in recent years. It will grow from $4.43 billion in 2025 to $4.79 billion in 2026 at a compound annual growth rate (CAGR) of 8.1%. The growth in the historic period can be attributed to growth in clinical trial volumes, increasing regulatory documentation requirements, expansion of pharmaceutical r&d activities, rise in biobanking practices, globalization of clinical research.What Is The Clinical Trial Biorepository And Archiving Solutions Market Growth Forecast?
The clinical trial biorepository and archiving solutions market size is expected to see strong growth in the next few years. It will grow to $6.54 billion in 2030 at a compound annual growth rate (CAGR) of 8.1%. The growth in the forecast period can be attributed to increase in precision medicine trials, growing biopharmaceutical pipelines, rising need for long-term sample retention, expansion of decentralized clinical trials, stricter regulatory audit requirements. Major trends in the forecast period include growing demand for long-term biospecimen storage, rising importance of regulatory-compliant archiving, expansion of global clinical trial activities, increasing use of centralized biorepositories, focus on data integrity and sample traceability.Global Clinical Trial Biorepository And Archiving Solutions Market Segmentation
1) By Product: Preclinical Products, Clinical Products 2) By Service: Biorepository Services, Archiving Solution Services 3) By Application: Research And Development, Clinical Trials, Regulatory Compliance, Drug Development 4) By End User: Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations, Academic Institutions Subsegments: 1) By Preclinical Products: Biospecimen Storage, Sample Processing And Preservation, Inventory Management Solutions, Data Management And Informatics 2) By Clinical Products: Biological Sample Archiving, Drug Compound Storage, Regulatory Compliance Solutions, Temperature-Controlled StorageWhat Is The Driver Of The Clinical Trial Biorepository And Archiving Solutions Market?
The increasing number of clinical trials is expected to propel the growth of the clinical trial biorepository and archiving solutions market in the future. Clinical trials are research studies conducted on humans to test new medical treatments or drugs' safety, effectiveness, and side effects. The increasing number of clinical trials is due to advancements in medical research and the expansion of personalized medicine, which requires more targeted and diverse clinical testing. Clinical trial biorepository and archiving solutions help drive the increasing number of clinical trials by ensuring secure long-term storage, easy retrieval of biological samples, and compliance with regulatory standards, which enhances data reliability and accelerates future research. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry (ABPI), a UK-based association, for the second consecutive year, industry-led clinical trial activity in the UK grew, rising from 411 trials in 2022 to 426 in 2023. Therefore, the increasing number of clinical trials is driving the growth of the clinical trial biorepository and archiving solutions industry.Key Players In The Global Clinical Trial Biorepository And Archiving Solutions Market
Major companies operating in the clinical trial biorepository and archiving solutions market are Thermo Fisher Scientific Inc., GE HealthCare, Labcorp Holdings Inc., IQVIA, ICON plc, Avantor Inc., VWR International LLC, Charles River Laboratories, Parexel, Clinigen Limited, Inotiv Inc., Medpace Holdings Inc., LabConnect LLC, Azenta Life Sciences, HSRL Holdings LLC, EPL Archives, Manuh Solutions LLC, EMINENT Services Corporation, Boca Biolistics LLC, AstoriomGlobal Clinical Trial Biorepository And Archiving Solutions Market Trends and Insights
Major companies operating in the clinical trial biorepository and archiving solutions market are focusing on developing innovative solutions, such as state-of-the-art biorepository facilities, to improve storage, data management, and compliance in clinical research. A state-of-the-art biorepository facility refers to an advanced storage and management center equipped with cutting-edge technology for preserving biological samples and clinical trial data. For instance, in January 2025, Azenta Life Sciences, a US-based biotech company, inaugurated a state-of-the-art biorepository facility in Greater Boston to support biotech and life sciences industries with advanced sample storage and management. The facility enables efficient handling of research, clinical trials, and biobanking while offering secure, compliant offsite storage for clinical trials.What Are Latest Mergers And Acquisitions In The Clinical Trial Biorepository And Archiving Solutions Market?
In July 2024, Tobin Scientific Inc., a US-based laboratory and provider of biopharma cold chain, ambient storage, transport, and logistics services, acquired BioRepository Resources LLC for an undisclosed amount. With this acquisition, Tobin Scientific Inc. aims to enhance its biopharmaceutical storage capabilities, expand its market presence, and strengthen its position as a leader in the life sciences industry. BioRepository Resources LLC is a US-based life sciences service provider specializing in clinical trial biorepository and archiving solutions.Regional Insights
North America was the largest region in the clinical trial biorepository and archiving solutions market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Biorepository And Archiving Solutions Market?
The clinical trial biorepository and archiving solutions market consists of revenues earned by entities by providing services such as long-term archiving, regulatory compliance, data management, inventory tracking and sample retrieval. The market value includes the value of related goods sold by the service provider or included within the service offering. The clinical trial biorepository and archiving solutions market also includes sales of cryogenic freezers, biobanking software, automated sample storage systems and liquid nitrogen storage tanks. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Biorepository And Archiving Solutions Market Report 2026?
The clinical trial biorepository and archiving solutions market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial biorepository and archiving solutions industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Biorepository And Archiving Solutions Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.79 billion |
| Revenue Forecast In 2035 | $6.54 billion |
| Growth Rate | CAGR of 8.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Service, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., GE HealthCare, Labcorp Holdings Inc., IQVIA, ICON plc, Avantor Inc., VWR International LLC, Charles River Laboratories, Parexel, Clinigen Limited, Inotiv Inc., Medpace Holdings Inc., LabConnect LLC, Azenta Life Sciences, HSRL Holdings LLC, EPL Archives, Manuh Solutions LLC, EMINENT Services Corporation, Boca Biolistics LLC, Astoriom |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
