
Clinical Trial Data Cloud Market Report 2026
Global Outlook – By Component (Software, Services), By Data Type (Clinical Data, Operational Data, Imaging Data, Genomics And Biomarker Data), By Deployment Model (Private Cloud, Public Cloud, Hybrid Cloud), By Application (Drug Development, Clinical Research, Patient Data Management, Regulatory Compliance, Other Applications), By End-User (Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Academic And Research Institutes, Hospitals And Clinics, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Clinical Trial Data Cloud Market Overview
• Clinical Trial Data Cloud market size has reached to $3.82 billion in 2025 • Expected to grow to $7.59 billion in 2030 at a compound annual growth rate (CAGR) of 14.8% • Growth Driver: The Increasing Number Of Clinical Trials Driving The Growth Of The Market Due To Rising Prevalence Of Chronic And Lifestyle-Related Diseases • Market Trend: Innovative Platforms Streamline Trial Data Management And Accelerate Research Insights • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Data Cloud Market?
Clinical trial data cloud refers to a secure, cloud-based platform designed to store, manage, and analyze clinical trial data in real time. It enables researchers and healthcare organizations to access large volumes of patient and trial information efficiently, ensuring data integrity, compliance, and collaboration across multiple sites. The main components of clinical trial data cloud include Software and services. Software refers to cloud-based platforms that enable centralized storage, integration, and analysis of clinical trial data to improve efficiency and collaboration. These solutions manage data types such as clinical data, operational data, imaging data, and genomics and biomarker data, and are deployed through private cloud, public cloud, and hybrid cloud models. The various applications involved are drug development, clinical research, patient data management, regulatory compliance, and other applications, and they are utilized by end users including pharmaceutical and biotechnology companies, contract research organizations, academic and research institutes, hospitals and clinics, and other end-users.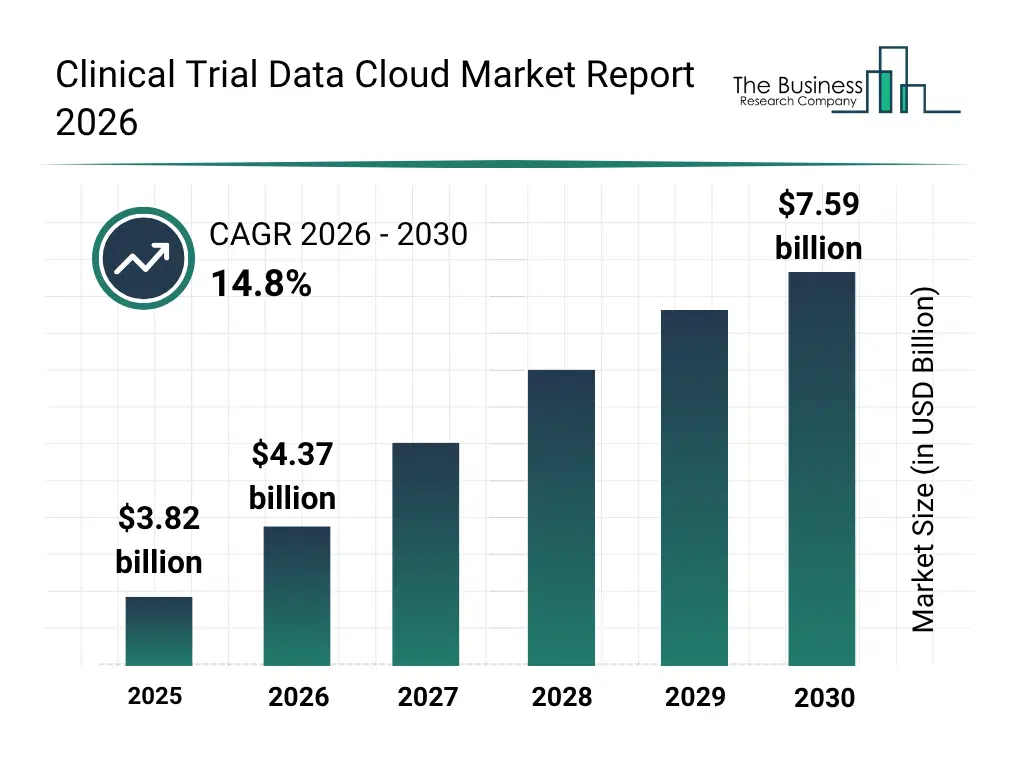
What Is The Clinical Trial Data Cloud Market Size and Share 2026?
The clinical trial data cloud market size has grown rapidly in recent years. It will grow from $3.82 billion in 2025 to $4.37 billion in 2026 at a compound annual growth rate (CAGR) of 14.5%. The growth in the historic period can be attributed to increasing digitization of clinical trials, adoption of electronic data capture systems, growing need for multi-site trial collaboration, rising focus on patient data integrity, expansion of cloud infrastructure in healthcare.What Is The Clinical Trial Data Cloud Market Growth Forecast?
The clinical trial data cloud market size is expected to see rapid growth in the next few years. It will grow to $7.59 billion in 2030 at a compound annual growth rate (CAGR) of 14.8%. The growth in the forecast period can be attributed to growing demand for AI-driven clinical analytics, increasing adoption of genomics and biomarker data integration, rising need for real-time patient monitoring, expansion of hybrid and public cloud solutions, growing focus on regulatory and compliance automation. Major trends in the forecast period include increasing adoption of hybrid cloud deployment models, rising demand for real-time clinical data analytics, growing integration of patient recruitment and engagement tools, expansion of regulatory compliance and safety management solutions, rising focus on managed services and technical support.Global Clinical Trial Data Cloud Market Segmentation
1) By Component: Software, Services 2) By Data Type: Clinical Data, Operational Data, Imaging Data, Genomics And Biomarker Data 3) By Deployment Model: Private Cloud, Public Cloud, Hybrid Cloud 4) By Application: Drug Development, Clinical Research, Patient Data Management, Regulatory Compliance, Other Applications 5) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Academic And Research Institutes, Hospitals And Clinics, Other End-Users Subsegments: 1) By Software: Clinical Data Management Software, Electronic Data Capture Software, Clinical Trial Management Software, Safety And Pharmacovigilance Software, Randomization And Trial Supply Management Software, Clinical Analytics And Reporting Software, Regulatory And Compliance Management Software, Patient Recruitment And Engagement Software, Trial Master File Software 2) By Services: Implementation And Integration Services, Consulting Services, Data Migration Services, Validation Services, Training And Education Services, Technical Support And Maintenance Services, Managed Services, Cloud Hosting And Infrastructure Services, Security And Compliance ServicesWhat Is The Driver Of The Clinical Trial Data Cloud Market?
The increasing number of clinical trials is expected to propel the growth of the clinical trial data cloud market. Clinical trials are systematically designed research studies conducted in humans to evaluate the safety and efficacy of new medical interventions. The growing volume of clinical trials is driven by factors such as the rising prevalence of chronic and lifestyle-related diseases, along with increasing investments in drug development and precision medicine. As the number and complexity of trials increase, sponsors and research organizations require advanced data management solutions to handle large volumes of diverse, real-time data. Clinical trial data cloud platforms address this need by centralizing, harmonizing, and analyzing trial data in real time, enabling faster decision-making, improved collaboration, and enhanced operational efficiency. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry, a UK-based trade association, the number of industry-initiated clinical trials in the UK rose from 411 in 2022 to 426 in 2023, reflecting a broader upward trend in clinical research activity. Therefore, the increasing number of clinical trials is driving the growth of the clinical trial data cloud industry.Key Players In The Global Clinical Trial Data Cloud Market
Major companies operating in the clinical trial data cloud market are Oracle Corporation, Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., ICON Public Limited Company, Veeva Systems Inc., Signant Health Inc., Advarra Inc., eClinical Solutions LLC, Medable Inc., Anju Software Solutions Pvt. Ltd., Medrio Inc., Florence Healthcare Inc., Calyx Inc., Castor Electronic Data Capture B.V., Viedoc Technologies AB, CluePoints Ltd., OpenClinica LLC, BioPharm Systems LLC, Delve Health Inc., and RealTime Software Solutions Inc.Global Clinical Trial Data Cloud Market Trends and Insights
Major companies operating in the clinical trial data cloud market are focusing on developing innovative solutions, such as integrated data harmonization platforms, to streamline data management, enhance trial efficiency, and support real-world evidence generation. Integrated data harmonization platforms are software solutions that combine and standardize data from multiple sources, such as clinical trials and real-world evidence, into a unified, consistent format for easier analysis and decision-making. For instance, in September 2025, nPhase Inc., a US based clinical research technology company, launched REDCap Cloud 2.0, an integrated data harmonization platform and innovative clinical trial data and analytics system designed to unify real-world evidence and clinical trial data within one interoperable environment. These platform features open architecture, standards-based data harmonization, automated workflows, and advanced analytics that streamline data management, shorten timelines, and support universal trial scalability from single-site pilots to complex, multi-phase programs. Its real-time data insights and integration with external sources enable researchers to make faster, more informed decisions across the study lifecycle.What Are Latest Mergers And Acquisitions In The Clinical Trial Data Cloud Market?
In February 2025, MMS Holdings Inc., a US-based data focused clinical research organization (CRO), acquired Exploristics for an undisclosed amount. With this acquisition, MMS aims to expand its biostatistics, advanced clinical trial design, and data science capabilities by integrating Exploristics’ flagship KerusCloud cloud based statistical modeling and simulation platform into its service offerings, enhancing its global footprint and technological depth in efficient drug development. Exploristics Ltd is a UK-based healthcare technology company that offers expert biostatistics consultancy and the KerusCloud platform for advanced clinical trial planning, analysis, and optimization.Regional Insights
North America was the largest region in the clinical trial data cloud market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Data Cloud Market?
The clinical trial data cloud market inclides revenues earned by entities by providing services such as cloud-based data storage and management, data analytics and visualization, electronic data capture (EDC) solutions, regulatory compliance and reporting services, and data anonymization and de-identification services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Clinical Trial Data Cloud Market Report 2026?
The clinical trial data cloud market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial data cloud industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Data Cloud Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.37 billion |
| Revenue Forecast In 2035 | $7.59 billion |
| Growth Rate | CAGR of 14.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Component, Data Type, Deployment Model, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Oracle Corporation, Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., ICON Public Limited Company, Veeva Systems Inc., Signant Health Inc., Advarra Inc., eClinical Solutions LLC, Medable Inc., Anju Software Solutions Pvt. Ltd., Medrio Inc., Florence Healthcare Inc., Calyx Inc., Castor Electronic Data Capture B.V., Viedoc Technologies AB, CluePoints Ltd., OpenClinica LLC, BioPharm Systems LLC, Delve Health Inc., and RealTime Software Solutions Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
