
Clinical Trial Data Visualization Market Report 2026
Global Outlook – By Component (Software, Services), By Deployment Mode (On-Premises, Cloud), By End-User (Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations, Academic And Government Research Institutes, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Data Visualization Market Overview
• Clinical Trial Data Visualization market size has reached to $0.89 billion in 2025 • Expected to grow to $1.82 billion in 2030 at a compound annual growth rate (CAGR) of 15.4% • Growth Driver: Increasing Adoption Of Cloud-Based Solutions Fueling The Growth Of The Market Due To Scalability, Flexibility, And Real-Time Data Access • Market Trend: AI-Powered Platforms Streamline Clinical Trial Data Aggregation And Analysis • North America was the largest region in 2025 and Asia Pacific is the fastest growing region.What Is Covered Under Clinical Trial Data Visualization Market?
Clinical trial data visualization refers to the use of graphical and visual tools to represent data collected during clinical trials. Its primary purpose is to make complex trial data more understandable, enabling researchers, sponsors, and regulators to quickly identify trends, patterns, and anomalies. This aids in informed decision-making, monitoring trial progress, and communicating results effectively. The main components of clinical trial data visualization are software and services. Software refers to specialized applications that collect, analyze, and visually present clinical trial data to help researchers identify patterns, monitor study progress, and make informed decisions efficiently. These solutions are available in both on-premises and cloud deployment modes and serve a diverse range of end users, including pharmaceutical companies, biotechnology companies, contract research organizations, academic and government research institutes, and other end users.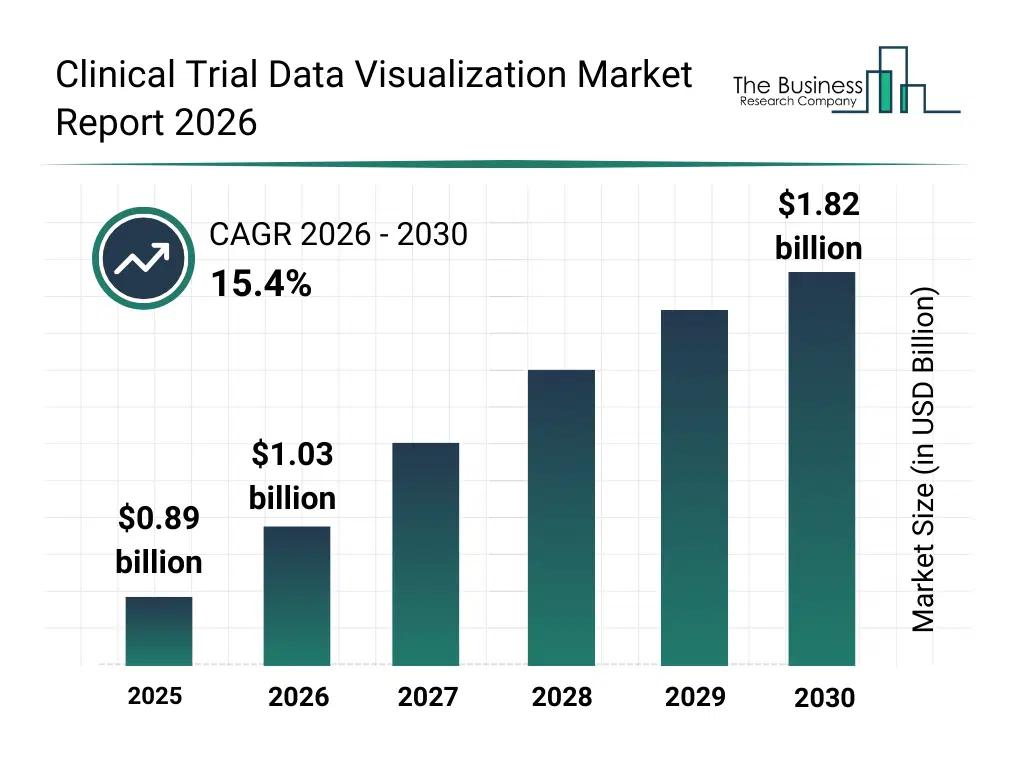
What Is The Clinical Trial Data Visualization Market Size and Share 2026?
The clinical trial data visualization market size has grown rapidly in recent years. It will grow from $0.89 billion in 2025 to $1.03 billion in 2026 at a compound annual growth rate (CAGR) of 15.7%. The growth in the historic period can be attributed to increasing complexity of clinical trial data, expansion of multi-site global trials, growing regulatory scrutiny, adoption of electronic data capture systems, rising demand for faster trial insights.What Is The Clinical Trial Data Visualization Market Growth Forecast?
The clinical trial data visualization market size is expected to see rapid growth in the next few years. It will grow to $1.82 billion in 2030 at a compound annual growth rate (CAGR) of 15.4%. The growth in the forecast period can be attributed to increasing use of ai-driven data visualization, expansion of decentralized and hybrid trials, rising demand for cloud-based analytics platforms, growing focus on predictive trial analytics, increasing investment in data transparency tools. Major trends in the forecast period include increasing adoption of interactive trial dashboards, rising use of advanced data analytics tools, growing demand for real-time trial monitoring, expansion of visual risk detection capabilities, enhanced focus on regulatory reporting accuracy.Global Clinical Trial Data Visualization Market Segmentation
1) By Component: Software, Services 2) By Deployment Mode: On-Premises, Cloud 3) By End-User: Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations, Academic And Government Research Institutes, Other End-Users Subsegments: 1) By Software: Data Visualization Tools, Data Analytics Platforms, Reporting Dashboards 2) By Services: Data Management Services, Consulting Services, Training And Support ServicesWhat Is The Driver Of The Clinical Trial Data Visualization Market?
The increasing adoption of cloud-based solutions is expected to propel the growth of the clinical trial data visualization market going forward. Cloud-based solutions are internet-delivered services that offer access to computing power, storage, and applications remotely, eliminating the need for on-site hardware or infrastructure. The cloud-based solutions are rising due to scalability and flexibility, as they allow businesses to easily adjust resources on demand without heavy upfront infrastructure costs. Cloud-based solutions enhance clinical trial data visualization by offering scalable and accessible platforms, making them ideal for geographically distributed study teams. They reduce manual data handling by providing real-time aggregation, analysis, and visualization, improving trial efficiency and decision-making. For instance, in December 2023, according to Eurostat, a Luxembourg-based government agency, in 2023, 42.5% of EU enterprises purchased cloud computing services mainly for email, file storage, and office software representing a 4.2-percentage-point increase compared with 2021. Therefore, the increasing adoption of cloud-based solutions is driving the growth of the clinical trial data visualization industry.Key Players In The Global Clinical Trial Data Visualization Market
Major companies operating in the clinical trial data visualization market are ICON plc, Dassault Systèmes European Company, Oracle Corporation, Thermo Fisher Scientific Inc., IQVIA Inc., Syneos Health Inc., Veeva Systems Inc., Medpace Holdings Inc., Medidata Solutions, Signant Health, QlikTech International AB, ArisGlobal LLC, Rho Inc., YPrime LLC, Anju Software Inc., TriNetX LLC, Quanticate International Limited., Thread Research Inc., Spotfire LLC, Datacubed Health Inc., Castor EDC Inc., Clinion Healthcare Technologies Pvt. Ltd.Global Clinical Trial Data Visualization Market Trends and Insights
Major companies operating in the clinical trial data visualization market are focusing on developing innovative solutions, such as artificial intelligence-powered data integration platforms, to streamline data aggregation, enhance real-time insights, and improve decision-making in clinical trials. Artificial Intelligence-powered data integration platforms are software solutions that use AI to automatically aggregate, analyze, and harmonize clinical trial data from multiple sources, enabling faster and more accurate insights. For instance, in June 2024, Medidata Solutions Inc., a US-based technology and life sciences company, launched Medidata Clinical Data Studio, an AI-driven data integration platform aimed at transforming the clinical trial data experience. The platform streamlines the aggregation, standardization, and review of clinical data, helping study teams cut data review cycle times by up to 80% and reduce patient profile review by 50%. It offers automated data ingestion and reconciliation, anomaly detection, risk-based quality management dashboards, and real-time collaborative insights, all within a single unified workspace, enhancing data quality, minimizing risk, and accelerating trial execution.What Are Latest Mergers And Acquisitions In The Clinical Trial Data Visualization Market?
In January 2023, Phastar CRO Private Limited, a UK-based biometrics contract research organization, acquired S-cubed ApS for an undisclosed amount. With this acquisition, Phastar aims to strengthen its position in data visualization and expand its global footprint. S-cubed ApS is a Denmark-based company that specializes in clinical trial data visualization services.Regional Outlook
North America was the largest region in the clinical trial data visualization market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Data Visualization Market?
The clinical trial data visualization market includes revenues earned by entities through data management services, visualization software solutions, analytics and reporting services, consulting services, and training and support services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Data Visualization Market Report 2026?
The clinical trial data visualization market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial data visualization industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Data Visualization Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.03 billion |
| Revenue Forecast In 2035 | $1.82 billion |
| Growth Rate | CAGR of 15.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Component, Deployment Mode, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | ICON plc, Dassault Systèmes European Company, Oracle Corporation, Thermo Fisher Scientific Inc., IQVIA Inc., Syneos Health Inc., Veeva Systems Inc., Medpace Holdings Inc., Medidata Solutions, Signant Health, QlikTech International AB, ArisGlobal LLC, Rho Inc., YPrime LLC, Anju Software Inc., TriNetX LLC, Quanticate International Limited., Thread Research Inc., Spotfire LLC, Datacubed Health Inc., Castor EDC Inc., Clinion Healthcare Technologies Pvt. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
