
Clinical Trial Imaging Market Report 2026
Global Outlook – By Product And Services (Services, Software), By Modality (Computed Tomography, Magnetic Resonance Imaging, Echocardiography, Nuclear Medicine, Positron Emission Tomography, X-Ray, Ultrasound, Optical Coherence Tomography, Other Modalities), By Application (Oncology, Neurology, Cardiology, Endocrinology, Dermatology, Hematology, Other Applications), By End User (Contract Research Organizations, Pharmaceutical And Biotechnology Companies, Medical Device Manufacturers, Academic And Government Research Institutes, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Imaging Market Overview
• Clinical Trial Imaging market size has reached to $1.58 billion in 2025 • Expected to grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 8.2% • Growth Driver: The Rising Prevalence Of Chronic Diseases Fuels Growth In The Clinical Trial Imaging Market • Market Trend: Innovations In Cloud-Based Image Viewers Drive Efficiency In Clinical Trial Imaging Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Imaging Market?
Clinical trial imaging refers to the use of medical imaging techniques, such as magnetic resonance imaging (MRI), computed tomography scan (CT) scans, and X-rays, to capture detailed internal images of participants during clinical trials. These images help monitor disease progression, assess treatment efficacy, and ensure patient safety. It is a crucial component in evaluating the biological impact of new therapies and interventions. The main products and services of clinical trial imaging are services and software. Clinical trial imaging services involve the provision of specialized imaging techniques and methodologies to support the research and development phases of clinical trials. The various modalities include computed tomography, magnetic resonance imaging, echocardiography, nuclear medicine, positron emission tomography, x-ray, ultrasound, optical coherence tomography, and others. These are used by various applications such as oncology, neurology, cardiology, endocrinology, dermatology, hematology, and others distributed through direct sales, and tender sales. These are also used by end users such as contract research organizations, pharmaceutical and biotechnology companies, medical device manufacturers, academic and government research institutes, others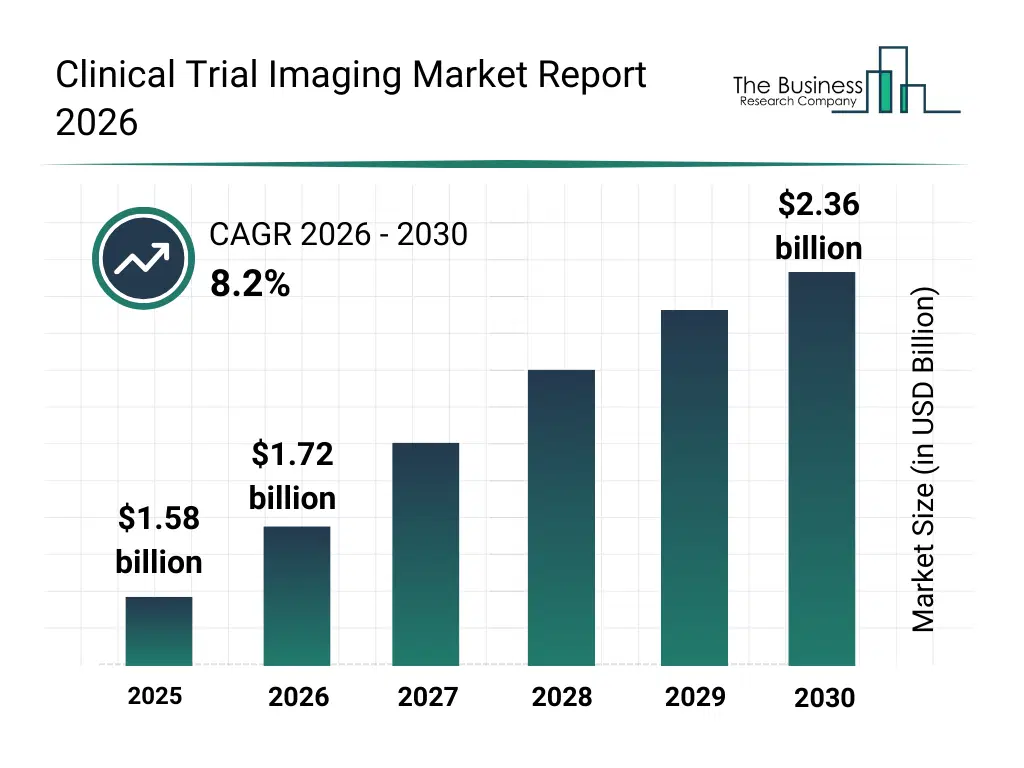
What Is The Clinical Trial Imaging Market Size and Share 2026?
The clinical trial imaging market size has grown strongly in recent years. It will grow from $1.58 billion in 2025 to $1.72 billion in 2026 at a compound annual growth rate (CAGR) of 9.3%. The growth in the historic period can be attributed to increasing complexity of clinical trial protocols, growing use of imaging endpoints in oncology trials, expansion of global clinical trials, rising regulatory emphasis on imaging accuracy, availability of advanced imaging modalities.What Is The Clinical Trial Imaging Market Growth Forecast?
The clinical trial imaging market size is expected to see strong growth in the next few years. It will grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 8.2%. The growth in the forecast period can be attributed to increasing adoption of decentralized clinical trials, rising investments in ai-enabled imaging platforms, growing demand for real-time imaging analytics, expansion of precision medicine trials, increasing integration of imaging with clinical data platforms. Major trends in the forecast period include increasing adoption of AI-assisted image analysis, rising use of multi-modality imaging in trials, growing demand for centralized image management services, expansion of remote imaging review capabilities, enhanced focus on imaging standardization and quality control.Global Clinical Trial Imaging Market Segmentation
1) By Product And Services: Services, Software 2) By Modality: Computed Tomography, Magnetic Resonance Imaging, Echocardiography, Nuclear Medicine, Positron Emission Tomography, X-Ray, Ultrasound, Optical Coherence Tomography, Other Modalities 3) By Application: Oncology, Neurology, Cardiology, Endocrinology, Dermatology, Hematology, Other Applications 4) By End User: Contract Research Organizations, Pharmaceutical And Biotechnology Companies, Medical Device Manufacturers, Academic And Government Research Institutes, Other End-Users Subsegments: 1) By Services: Imaging Services For Diagnostics, Image Acquisition Services, Image Analysis Services, Image Management And Archiving Services 2) By Software: Imaging Software For Analysis, Image Processing Software, Radiology Information Systems (RIS), Picture Archiving And Communication Systems (PACS)What Is The Driver Of The Clinical Trial Imaging Market?
The increasing prevalence of chronic diseases is expected to propel the growth of the clinical trial imaging market going forward. Chronic disease is a long-lasting condition that typically requires ongoing medical attention and can limit daily activities or quality of life. The increasing prevalence of chronic diseases is due to exposure to air pollution, chemicals, and toxins, lifestyle changes, and genetic factors. Clinical trial imaging provides efficient monitoring through imaging that can help patients understand their condition and the impact of treatments, improving adherence and satisfaction. For instance, in January 2023, according to the National Center for Biotechnology Information (NCBI), a US-based National Library of Medicine, it is projected that by the year 2050, the number of individuals aged 50 years and above who have at least one chronic illness will surge by 99.5%, rising from 71.522 million in 2020 to 142.66 million. Therefore, the increasing prevalence of chronic diseases drives the clinical trial imaging industry.Key Players In The Global Clinical Trial Imaging Market
Major companies operating in the clinical trial imaging market are Koninklijke Philips N.V., Labcorp Drug Development, ICON Plc, PPD Inc., Syneos Health, Charles River Laboratories, Parexel International Corporation, Clario, Medpace Inc., Medidata Solutions Inc., Biotelemetry Inc., ProScan Imaging LLC, Invicro LLC, Perspectum, Intrinsic Imaging LLC, Datatrak Inc., IXICO Plc, Calyx Inc., Resonance Health Ltd., Anagram-ESIC SLGlobal Clinical Trial Imaging Market Trends and Insights
Major companies operating in the clinical trial imaging market are developing cloud-based image tools to provide better solutions and improve the efficiency of clinical trials. Cloud-based imaging tools are software applications and platforms that utilize cloud computing technology to store, manage, and analyze medical images. For instance, in March 2023, Clarion, a US-based manufacturer of advanced power distribution systems, launched a cutting-edge cloud-based image viewer tool. This innovative tool offers real-time, on-demand viewing and manipulation of high-resolution images, providing read-quality access to images in near real-time while eliminating manual steps that were previously required. It streamlines the image viewing process, eliminating manual steps and making image accessibility easy, secure and data privacy compliant. The tool offers full-resolution viewing and manipulation capabilities, empowering end users to gain valuable insights into their trial subject.What Are Latest Mergers And Acquisitions In The Clinical Trial Imaging Market?
In March 2023, Medica, a UAE-based medical company acquired RadMD for an undisclosed amount. The acquisition of RadMD by Medica represents a strategic move to enter the clinical trials market, leveraging RadMD's expertise and market experience to reduce costs and improve efficiency in drug and device development for pharmaceutical, biotech, and contract research organizations (CROs). RadMD is a US-based company that offers clinical trial imaging.Regional Outlook
North America was the largest region in the clinical trial imaging market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Imaging Market?
The clinical trial imaging market consists of revenues earned by entities by providing services such as image protocol development, data management and integration, image processing, and analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. The clinical trial imaging market also includes sales of analysis tools, clinical trial management systems, and compliance tools. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Imaging Market Report 2026?
The clinical trial imaging market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial imaging industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Imaging Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.72 billion |
| Revenue Forecast In 2035 | $2.36 billion |
| Growth Rate | CAGR of 9.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product And Services, Modality, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Koninklijke Philips N.V., Labcorp Drug Development, ICON Plc, PPD Inc., Syneos Health, Charles River Laboratories, Parexel International Corporation, Clario, Medpace Inc., Medidata Solutions Inc., Biotelemetry Inc., ProScan Imaging LLC, Invicro LLC, Perspectum, Intrinsic Imaging LLC, Datatrak Inc., IXICO Plc, Calyx Inc., Resonance Health Ltd., Anagram-ESIC SL |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
