
Clinical Trial Investigative Site Network Market Report 2026
Global Outlook – By Type Of Clinical Trials (Interventional Trials, Observational Trials, Expanded Access Trials, Adaptive Trials), By Site Type (Academic Medical Centers, Community Hospitals, Specialized Research Institutions, Private Practices, Site Management Organizations (SMOs)), By Application (Oncology, Cardiology, Central Nervous System, Pain Management, Endocrine, Other Applications), By End-Use (Pharmaceutical And Biopharmaceutical Companies, Medical Device Companies, Other End-Uses) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Investigative Site Network Market Overview
• Clinical Trial Investigative Site Network market size has reached to $8.71 billion in 2025 • Expected to grow to $13.01 billion in 2030 at a compound annual growth rate (CAGR) of 8.3% • Growth Driver: Rising Demand For Clinical Research Activity Fueling The Growth Of The Market Due To The Need For Innovative Therapies • Market Trend: Innovative Platform Launches Transform Clinical Trial Site Operations • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Investigative Site Network Market?
Clinical trial investigative site network refers to a group or consortium of clinical research sites that collaborate to conduct clinical trials under a unified framework. These networks streamline study execution by providing standardized procedures, centralized management, and faster patient recruitment across multiple locations. The main clinical trial types of clinical trial investigative site networks include interventional trials, observational trials, expanded access trials, and adaptive trials. Interventional trials are clinical studies in which researchers actively assign participants to receive specific treatments or interventions to evaluate their effects on health outcomes. The site types typically include academic medical centers, community hospitals, specialized research institutions, private practices, and site management organizations (SMOs). It is used for a wide range of applications, including oncology, cardiology, central nervous system disorders, pain management, endocrine disorders, and others. The key end-users of these trials are pharmaceutical and biopharmaceutical companies, medical device companies, and other healthcare-related organizations.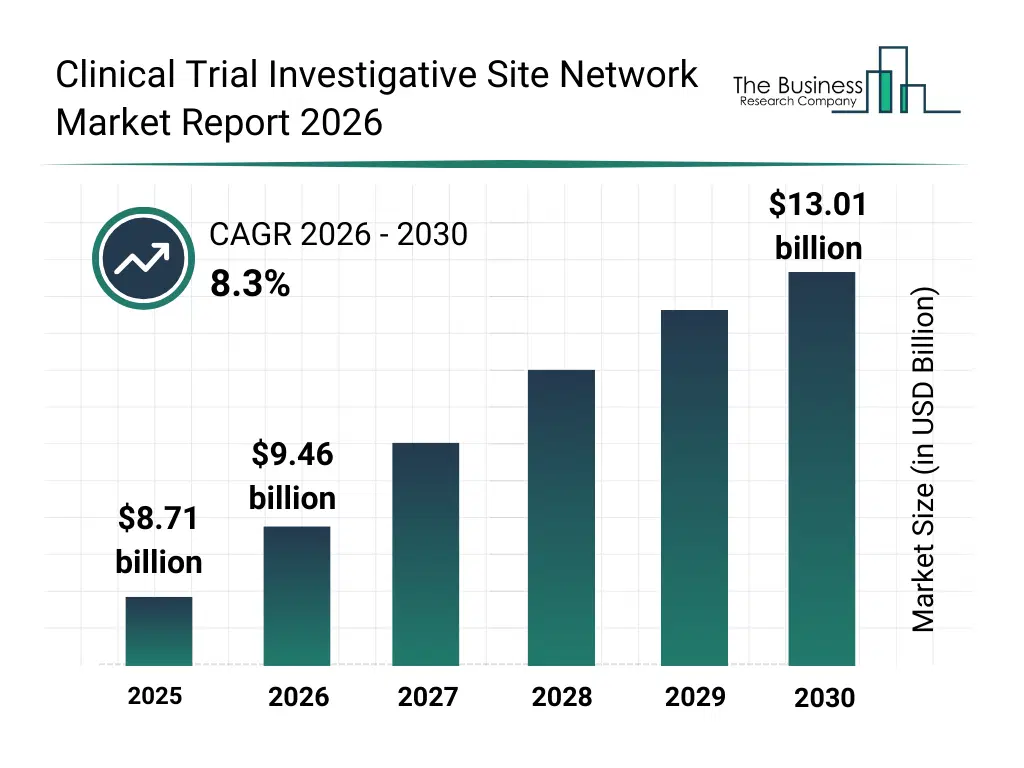
What Is The Clinical Trial Investigative Site Network Market Size and Share 2026?
The clinical trial investigative site network market size has grown strongly in recent years. It will grow from $8.71 billion in 2025 to $9.46 billion in 2026 at a compound annual growth rate (CAGR) of 8.6%. The growth in the historic period can be attributed to increasing number of clinical trials globally, expansion of multi-site research studies, rising pharmaceutical r&d spending, adoption of standardized clinical protocols, growth of site management organizations.What Is The Clinical Trial Investigative Site Network Market Growth Forecast?
The clinical trial investigative site network market size is expected to see strong growth in the next few years. It will grow to $13.01 billion in 2030 at a compound annual growth rate (CAGR) of 8.3%. The growth in the forecast period can be attributed to increasing demand for faster trial execution, rising focus on rare and complex disease trials, expansion of adaptive trial designs, growing use of digital patient engagement tools, increasing collaboration between sponsors and site networks. Major trends in the forecast period include expansion of decentralized and hybrid clinical trials, growing use of centralized patient recruitment models, increasing adoption of standardized trial operations, rising integration of digital trial management platforms, enhanced focus on data quality and compliance.Global Clinical Trial Investigative Site Network Market Segmentation
1) By Type Of Clinical Trials: Interventional Trials, Observational Trials, Expanded Access Trials, Adaptive Trials 2) By Site Type: Academic Medical Centers, Community Hospitals, Specialized Research Institutions, Private Practices, Site Management Organizations (SMOs) 3) By Application: Oncology, Cardiology, Central Nervous System, Pain Management, Endocrine, Other Applications 4) By End-Use: Pharmaceutical And Biopharmaceutical Companies, Medical Device Companies, Other End-Uses Subsegments: 1) By Type of Interventional Trials: Phase I, Phase II, Phase III, Phase IV 2) By Type of Observational Trials: Cohort Studies, Case-Control Studies, Cross-Sectional Studies, Longitudinal Studies 3) By Type of Expanded Access Trials: Individual Patient Access, Intermediate-Size Patient Populations, Treatment IND Or Protocol Programs, Emergency Use Access 4) By Type of Adaptive Trials: Dose-Finding Adaptive Trials, Sample Size Re-estimation Trials, Adaptive Randomization Trials, Seamless Phase II Or III TrialsWhat Is The Driver Of The Clinical Trial Investigative Site Network Market?
The increasing demand for clinical research activity is expected to propel the growth of the clinical trial investigative site network market going forward. Clinical research activity refers to any organized efforts related to designing and analyzing clinical studies that aim to improve medical knowledge or public health. The demand for clinical research activity is driven by the growing need for innovative therapies to treat chronic and complex diseases. This has intensified efforts to develop safe and effective drugs and medical devices through rigorous clinical trials. A clinical trial investigative site network helps clinical research activity by allowing centralized coordination, faster execution, and consistent quality across multiple trial sites. For instance, in December 2024, according to the National Library of Medicine (NLM), a US-based biomedical library, there were 520,884 registered clinical trials worldwide, a significant rise from approximately 477,207 in mid-2023. This represents an increase of nearly 20% in just over one year, highlighting a continued and robust expansion in global clinical research activity. Therefore, the increasing demand for clinical research activity is driving the growth of the clinical trial investigative site network industry.Key Players In The Global Clinical Trial Investigative Site Network Market
Major companies operating in the clinical trial investigative site network market are IQVIA Inc, ICON Plc, Fortrea Holdings Inc, Medpace Holdings Inc, ClinChoice Inc, Velocity Clinical Research Inc, NVISION Clinical Research LLC, George Clinical, Medical Research Network Limited, MAC Clinical Research Ltd, FOMAT Medical Research Inc, WCG Clinical, Access Clinical Trials Inc, KV Clinical Research, SMO Pharmina, Clinical Research Network LLC, CenExel Clinical Research, EMS Healthcare Ltd, PPD Inc, PRA Health Sciences, Parexel International Ltd, Syneos Health, Altasciences Clinical Research, Worldwide Clinical TrialsGlobal Clinical Trial Investigative Site Network Market Trends and Insights
Major companies operating in the clinical trial investigative site network market are focusing on developing innovative solutions, such as single sign-on platforms, to streamline site operations, enhance data access, and improve trial efficiency. Single sign-on platforms refer to authentication systems that allow users to access multiple applications or systems with one set of login credentials. These platforms simplify user access, enhance security, and reduce the administrative burden of managing multiple logins. For instance, in June 2024, IQVIA Inc., a US-based company specializing in clinical research and health technology, launched One Home for Sites, a unified clinical trial platform designed to simplify site workflows and enhance trial efficiency. Its key features include single sign-on access to streamline user login across multiple trial systems, centralized study dashboards for better oversight of ongoing trials, and integrated communication tools to enhance collaboration between sponsors, CROs, and site teams. The platform also supports automated workflows, document management, and real-time performance analytics, enabling sites to reduce administrative complexity, improve regulatory compliance, and accelerate trial execution with greater efficiency and transparency.What Are Latest Mergers And Acquisitions In The Clinical Trial Investigative Site Network Market?
In January 2025, Medical Research Network (MRN), a UK-based company that specializes in accelerating clinical trials, acquired Volunteer Centre for Trials & Clinical Research (VCTC) for an undisclosed amount. With this acquisition, Medical Research Network (MRN) aims to expand its UK footprint and enhance its global community-based clinical trial capabilities by integrating VCTC's multi-therapeutic site network and advancing virtual trial delivery models. VCTC is a UK-based clinical trial site network specializing in patient-centric and decentralized clinical research solutions.Regional Outlook
North America was the largest region in the clinical trial investigative site network market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Investigative Site Network Market?
The clinical trial investigative site network market consists of revenues earned by entities by providing services such as patient recruitment and enrollment, study conduct and management, data collection and management, site monitoring and quality assurance, and patient care and follow-up. The market value includes the value of related goods sold by the service provider or included within the service offering. The clinical trial investigative site network market also includes sales of clinical trial data management systems, patient recruitment tools, standardized protocol templates, electronic case report forms and monitoring and reporting tools. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Investigative Site Network Market Report 2026?
The clinical trial investigative site network market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial investigative site network industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Investigative Site Network Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $9.46 billion |
| Revenue Forecast In 2035 | $13.01 billion |
| Growth Rate | CAGR of 8.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type Of Clinical Trials, Site Type, Application, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | IQVIA Inc, ICON Plc, Fortrea Holdings Inc, Medpace Holdings Inc, ClinChoice Inc, Velocity Clinical Research Inc, NVISION Clinical Research LLC, George Clinical, Medical Research Network Limited, MAC Clinical Research Ltd, FOMAT Medical Research Inc, WCG Clinical, Access Clinical Trials Inc, KV Clinical Research, SMO Pharmina, Clinical Research Network LLC, CenExel Clinical Research, EMS Healthcare Ltd, PPD Inc, PRA Health Sciences, Parexel International Ltd, Syneos Health, Altasciences Clinical Research, Worldwide Clinical Trials |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
