
Clinical Trial Management System Market Report 2026
Global Outlook – By System Type (Enterprise Based Clinical Trial Management System, Site Based Clinical Trial Management System), By Component (Software, Services, Hardware), By Mode Of Delivery (Cloud Based Clinical Trial Management System, Web Based Clinical Trial Management System, On-Premise Clinical Trial Management System), By End-User (Pharmaceutical And Biopharmaceutical Companies, Medical Device Manufacturers, Clinical Research Organizations (CRO), Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Management System Market Overview
• Clinical Trial Management System market size has reached to $2.25 billion in 2025 • Expected to grow to $4.2 billion in 2030 at a compound annual growth rate (CAGR) of 13.4% • Growth Driver: Clinical Trial Management Systems (CTMS) Market Surges As The Number Of Clinical Trials Soars Worldwide • Market Trend: Innovative Clinical Trial Technology Platforms Streamline Processes And Enhance Efficiency • North America was the largest region in 2025 and Asia Pacific is the fastest growing region.What Is Covered Under Clinical Trial Management System Market?
A clinical trial management system (CTMS) is a software system used by the biotechnology and pharmaceutical industries to manage clinical trials in clinical research. It is a comprehensive solution to centralize, organize, and automate various aspects of the clinical trial process, enabling efficient study planning, execution, monitoring, and reporting. The main types of clinical trial management systems are enterprise-based and site-based. An enterprise-based clinical trial management system is a software solution that is implemented and operated across an entire organization or enterprise and provides a centralized platform for managing all clinical trials conducted by the organization. The components of a clinical trial management system include software, services, and hardware delivered in cloud-based, web-based, and on-premises modes to be used by pharmaceutical and biopharmaceutical companies, medical device manufacturers, clinical research organizations (CROs), and others.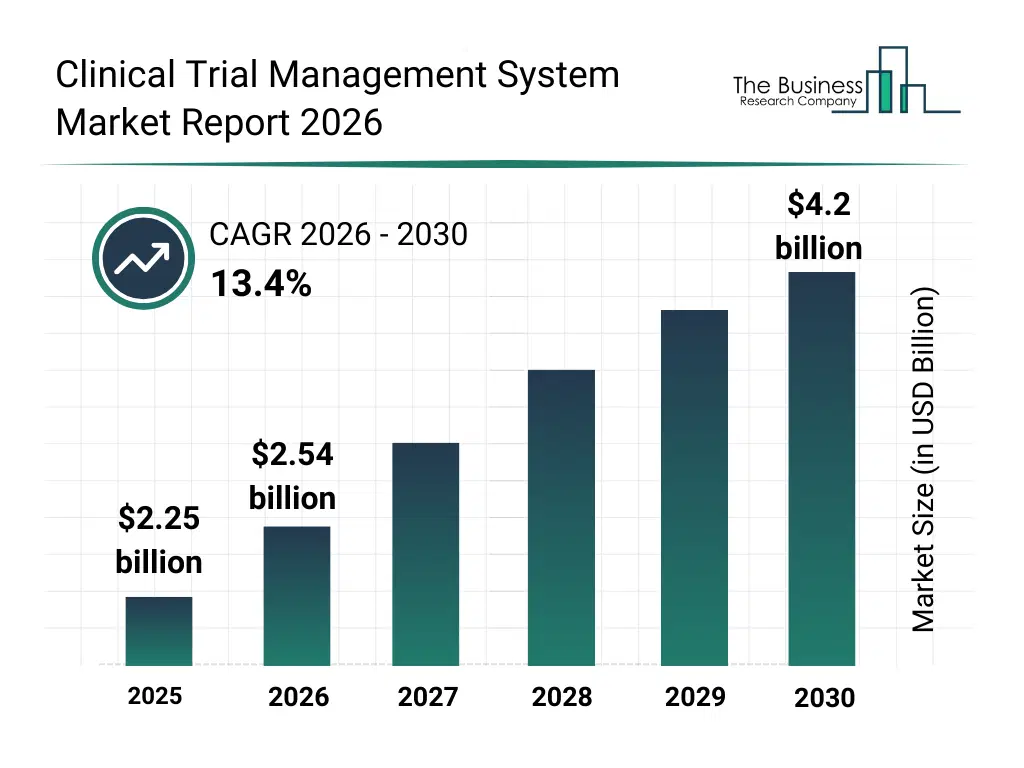
What Is The Clinical Trial Management System Market Size and Share 2026?
The clinical trial management system market size has grown rapidly in recent years. It will grow from $2.25 billion in 2025 to $2.54 billion in 2026 at a compound annual growth rate (CAGR) of 13.1%. The growth in the historic period can be attributed to increasing number of global clinical trials, rising complexity of multi-site trial management, growing need for centralized trial oversight, expansion of outsourced clinical research, availability of enterprise software solutions.What Is The Clinical Trial Management System Market Growth Forecast?
The clinical trial management system market size is expected to see rapid growth in the next few years. It will grow to $4.2 billion in 2030 at a compound annual growth rate (CAGR) of 13.4%. The growth in the forecast period can be attributed to increasing adoption of decentralized trial models, rising investments in digital clinical infrastructure, growing focus on predictive trial management, expansion of interoperability across clinical systems, increasing regulatory digitization requirements. Major trends in the forecast period include increasing adoption of cloud-based ctms platforms, rising use of AI-driven trial analytics, growing demand for real-time trial monitoring tools, expansion of integrated clinical data management systems, enhanced focus on regulatory compliance automation.Global Clinical Trial Management System Market Segmentation
1) By System Type: Enterprise Based Clinical Trial Management System, Site Based Clinical Trial Management System 2) By Component: Software, Services, Hardware 3) By Mode Of Delivery: Cloud Based Clinical Trial Management System, Web Based Clinical Trial Management System, On-Premise Clinical Trial Management System 4) By End-User: Pharmaceutical And Biopharmaceutical Companies, Medical Device Manufacturers, Clinical Research Organizations (CRO), Other End-Users Subsegments: 1) By Enterprise Based Clinical Trial Management System: Comprehensive CTMS Solutions, Modular CTMS Solutions 2) By Site Based Clinical Trial Management System: Site-Level Data Management Tools, Study Management Solutions For SitesWhat Is The Driver Of The Clinical Trial Management System Market?
The increasing number of clinical trials is expected to propel the growth of the clinical trial management system market. Clinical trials are research studies conducted on human participants to evaluate the safety, efficacy, and potential benefits of new medical treatments, interventions, or drugs. Clinical Trials Management Systems (CTMS) are software platforms designed to streamline and facilitate the management of clinical trials. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry (ABPI), a UK-based trade association, the number of industry clinical trials initiated in the UK increased to 426 in 2023, from 411 in 2022. Therefore, an increasing number of clinical trials will drive the growth of the clinical trial management system industry.Key Players In The Global Clinical Trial Management System Market
Major companies operating in the clinical trial management system market are Oracle Corporation, Veeva Systems Inc, BioClinica Inc, Calyx, ArisGlobal LLC, Medrio Inc, Anju Software Inc, Forte Research System Inc, OmniComm Systems Inc, SimpleTrials, Castor, Viedoc, Cloudbyz, Crucial Data Solutions, eClinPro, Ennov, EvidentIQ, ClinPlus, Clinion, MedNet Solutions, DataTrak International, eClinForce, Flex Databases, Axiom, ERT ClinicalGlobal Clinical Trial Management System Market Trends and Insights
Major companies operating in the clinical trial management system market are focusing on developing technologically innovative solutions, such as clinical trial technology platforms to streamline trial processes, enhance data accuracy, and ensure regulatory compliance. A clinical trial technology platform refers to a comprehensive, digital ecosystem designed to manage and streamline the complex processes involved in clinical trials. For instance, in June 2024, IQVIA, a US-based health information technology company, launched the One Home for Sites platform, a comprehensive technology solution designed to streamline clinical trial management by integrating multiple applications into a single sign-on dashboard. This platform reduces the administrative burdens faced by clinical research sites, allowing staff to focus more on patient care and recruitment while managing multiple trials efficiently. The platform aims to enhance operational efficiency, accelerate trial processes, and ultimately increase the availability of new treatments for patients.What Are Latest Mergers And Acquisitions In The Clinical Trial Management System Market?
In July 2023, RealTime Software Solutions, LLC, a US-based company and prominent provider of cloud-based software solutions, acquired Devana Solutions, LLC, for an undisclosed amount. This acquisition is strategically focused on leveraging complementary technologies to improve clinical trial processes, enhance operational efficiencies, and better support researchers and participants alike. Devana Solutions, LLC is a US-based cloud-based software company specializing in clinical trial workflow and analytics.Regional Outlook
North America was the largest region in the Asia Pacific market in 2025. Asia Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Management System Market?
The clinical trials management system market consists of revenues earned by entities by providing Web-based clinical trials management system, End-to-end clinical trials management system, Integrated clinical trials management system, and Cloud-based clinical trials management system. The market value includes the value of related goods sold by the service provider or included within the service offering. The clinical trials management system market also includes sales of clinical trial analytics tools, scanners, printers, and electronic data capture devices that are used in providing clinical trials management services. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Management System Market Report 2026?
The clinical trial management system market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial management system industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Management System Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.54 billion |
| Revenue Forecast In 2035 | $4.2 billion |
| Growth Rate | CAGR of 13.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | System Type, Component, Mode Of Delivery, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Oracle Corporation, Veeva Systems Inc, BioClinica Inc, Calyx, ArisGlobal LLC, Medrio Inc, Anju Software Inc, Forte Research System Inc, OmniComm Systems Inc, SimpleTrials, Castor, Viedoc, Cloudbyz, Crucial Data Solutions, eClinPro, Ennov, EvidentIQ, ClinPlus, Clinion, MedNet Solutions, DataTrak International, eClinForce, Flex Databases, Axiom, ERT Clinical |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
