
Clinical Trial Management Systems Market Report 2026
Global Outlook – By Product (Enterprise-Based, Site-Based), By Components (Software, Services), By Delivery Mode (Web-based Clinical Trial Management Systems, On-Premise Clinical Trial Management Systems, Cloud-Based Clinical Trial Management Systems), By End-User (Pharmaceutical And Biotechnology Firms, Medical Device Firms, Clinical Research Organization (CROs), Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Clinical Trial Management Systems Market Overview
• Clinical Trial Management Systems market size has reached to $2.08 billion in 2025 • Expected to grow to $4.01 billion in 2030 at a compound annual growth rate (CAGR) of 14% • Growth Driver: R&D Investments Boom Accelerates Clinical Trial Management Systems Market • Market Trend: Industry Players Redefine Clinical Trial Management With Advanced Systems • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Management Systems Market?
Clinical trial management systems refer to a comprehensive cloud-based software platform for managing clinical studies from beginning to end. It is commonly used for clinical trial planning, monitoring, analysis, locating, and handling the participant patients to keep track of subjects' participation in clinical trials and manage the finances. The main products of clinical trial management systems are enterprise-based and site-based. Enterprise-based refers to a substantial software platform that is often created to function in a corporate setting, such as one found in business or government. The various components include software and services. The various delivery modes include web-based CTMS, on-premise CTMS, and cloud-based CTMS, which are used by various end-users such as pharmaceutical and biotechnology firms, medical device firms, clinical research organizations (CROs), and others.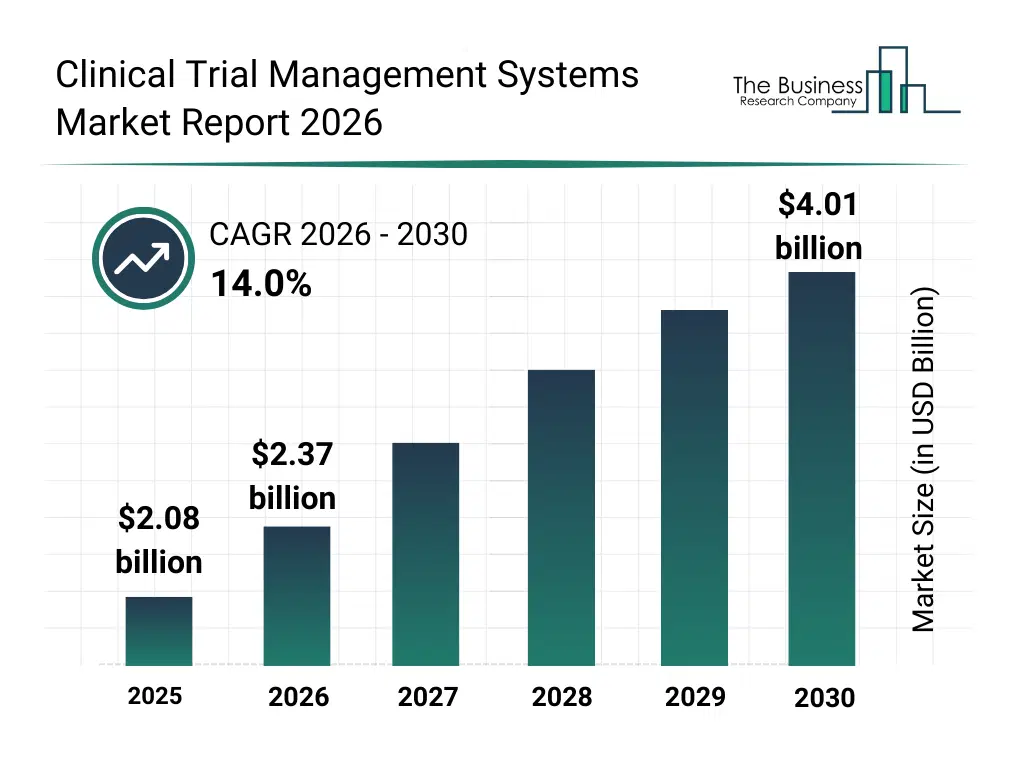
What Is The Clinical Trial Management Systems Market Size and Share 2026?
The clinical trial management systems market size has grown rapidly in recent years. It will grow from $2.08 billion in 2025 to $2.37 billion in 2026 at a compound annual growth rate (CAGR) of 13.9%. The growth in the historic period can be attributed to growth in clinical research activities, increasing complexity of clinical trials, demand for efficient trial data management, expansion of pharmaceutical r and d spending, need for regulatory compliance tracking.What Is The Clinical Trial Management Systems Market Growth Forecast?
The clinical trial management systems market size is expected to see rapid growth in the next few years. It will grow to $4.01 billion in 2030 at a compound annual growth rate (CAGR) of 14.0%. The growth in the forecast period can be attributed to adoption of decentralized trial models, integration of AI driven analytics, demand for faster trial execution, growth of personalized medicine research, increasing regulatory scrutiny. Major trends in the forecast period include shift toward cloud based ctms platforms, integration of real time trial monitoring, adoption of decentralized clinical trials, advanced data analytics for trial management, increased focus on regulatory compliance.Global Clinical Trial Management Systems Market Segmentation
1) By Product: Enterprise-Based, Site-Based 2) By Components: Software, Services 3) By Delivery Mode: Web-based Clinical Trial Management Systems, On-Premise Clinical Trial Management Systems, Cloud-Based Clinical Trial Management Systems 4) By End-User: Pharmaceutical And Biotechnology Firms, Medical Device Firms, Clinical Research Organization (CROs), Other End-Users Subsegments: 1) By Enterprise-Based: Centralized Clinical Trial Management Systems (CTMS), Cloud-Hosted Enterprise CTMS, Multi-Study Enterprise CTMS, Integrated Enterprise CTMS Solutions 2) By Site-Based: Local Site Clinical Trial Management Systems (CTMS), Standalone Site CTMS, Investigator-Site CTMS, Department-Level Site CTMSWhat Is The Driver Of The Clinical Trial Management Systems Market?
The rising research and development investments in healthcare is expected to propel the growth of the clinical trial management systems market going forward. Research and development (R&D) investments refer to the financial resources allocated by organizations or companies for discovering, creating, and enhancing new products, services, processes, or technologies. Expanded investments in research and development activities by pharmaceutical, biotechnology, and medical device companies drive the demand for efficient clinical trial management systems (CTMS) solutions to manage and optimize clinical trials. For instance, in February 2025, according to the National Center for Science and Engineering Statistics (NCSES), a US-based government statistical agency, U.S. research and development expenditures rose from $892 billion in 2022 to $940 billion in 2023, reflecting an increase of $48 billion. Therefore, the rising research and development investments in healthcare are driving the growth of the clinical trial management systems industry.Key Players In The Global Clinical Trial Management Systems Market
Major companies operating in the clinical trial management systems market are Thermo Fisher Scientific Inc., Oracle Corporation, Parexel International Corporation, Clario, Veeva Systems Inc., Medpace Holdings Inc., ERT Inc., Advarra Inc., Medidata Solutions Inc., ArisGlobal LLC, Anju Software Inc., MasterControl Inc., MedNet Solutions Inc., OmniComm Systems Inc., Bio-Optronics Inc., ClinCapture Inc., Forte Research Systems Inc., DataTRAK International Inc., OpenClinica LLC, SimpleTrialsGlobal Clinical Trial Management Systems Market Trends and Insights
Major companies operating in the clinical trial management systems market are focused on developing innovative solutions such as advanced clinical trial management systems. Advanced Clinical Trial Management Systems (CTMS) refer to sophisticated and comprehensive software solutions designed to streamline and optimize the planning, tracking, management, and reporting of clinical trials. For instance, in October 2023, BGO Software, a Bulgaria-based software company launched ‘Clinicubes CTMS’. It represents a sophisticated system that caters to fundamental study requirements, streamlines crucial study processes, furnishes research specialists with robust evaluation and standardization features, promotes seamless information sharing, ensures prompt delivery of results, guarantees cost-effectiveness, and offers additional benefits.What Are Latest Mergers And Acquisitions In The Clinical Trial Management Systems Market?
In August 2024, Valsoft Corporation Inc., a Canada-based company that acquires and develops vertical market software businesses, acquired Anju Software for an undisclosed amount. This acquisition aims to preserve the strong legacy Anju has established in life sciences, while continuing to build innovative technology for existing and new customers. Anju Software is a US-based provider of clinical trial management system.Regional Insights
North America was the largest region in the clinical trial management systems market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Management Systems Market?
The clinical trial management systems market includes revenues earned by entities by providing treatment trials, prevention trials, screening trials, supportive and palliative care trials management services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Management Systems Market Report 2026?
The clinical trial management systems market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial management systems industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Management Systems Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.37 billion |
| Revenue Forecast In 2035 | $4.01 billion |
| Growth Rate | CAGR of 13.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Components, Delivery Mode, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Oracle Corporation, Parexel International Corporation, Clario, Veeva Systems Inc., Medpace Holdings Inc., ERT Inc., Advarra Inc., Medidata Solutions Inc., ArisGlobal LLC, Anju Software Inc., MasterControl Inc., MedNet Solutions Inc., OmniComm Systems Inc., Bio-Optronics Inc., ClinCapture Inc., Forte Research Systems Inc., DataTRAK International Inc., OpenClinica LLC, SimpleTrials |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
