
Clinical Trial Patient Recruitment Services Market Report 2026
Global Outlook – By Service Type (Patient Recruitment And Registry Services, Patient Retention Services, Other Service Types), By Phase (Phase I, Phase II, Phase III, Phase IV), By Therapeutic Area (Respiratory Diseases, Pain And Anesthesia, Oncology, Central Nervous System, Cardiovascular, Endocrine, Anti-Infective, Other Therapeutic Areas), By Recruitment Channel (Investigator-Site Led, Contract Research Organization (CRO) Led, Direct-To-Patient, Patient-Advocacy Group Partnerships), By End-User (Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations (CROs), Clinical Research Institutions) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Patient Recruitment Services Market Overview
• Clinical Trial Patient Recruitment Services market size has reached to $3.37 billion in 2025 • Expected to grow to $4.98 billion in 2030 at a compound annual growth rate (CAGR) of 8.1% • Growth Driver: Growing Number Of Clinical Trials Fueling The Growth Of The Market Due To Rising Demand For Efficient Patient Enrollment And Streamlined Trial Processes • Market Trend: AI-Powered Tools Enhance Eligibility Matching Accuracy In Trials • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Patient Recruitment Services Market?
Clinical trial patient recruitment services involve identifying, engaging, and enrolling suitable participants for clinical studies. Their primary purpose is to ensure timely and sufficient patient enrollment, which is essential for the success, validity, and efficiency of clinical trials. These services often include outreach, screening, and patient education to optimize recruitment and retention. The main types of clinical trial patient recruitment services are patient recruitment and registry services, patient retention services, and others. Patient recruitment and registry services involve identifying, enrolling, and maintaining databases of eligible participants for clinical trials or research studies to support efficient enrollment and long-term participant tracking. These services cover various phases such as Phase I, Phase II, Phase III, and Phase IV, and are used across different therapeutic areas, including respiratory diseases, pain and anesthesia, oncology, central nervous system, cardiovascular, endocrine, anti-infective, and others. Recruitment channels vary and include investigator-site-led, contract research organization (CRO)-led, direct-to-patient, and patient-advocacy group partnerships, with key end users such as pharmaceutical companies, biotechnology companies, contract research organizations (CROs), and clinical research institutions.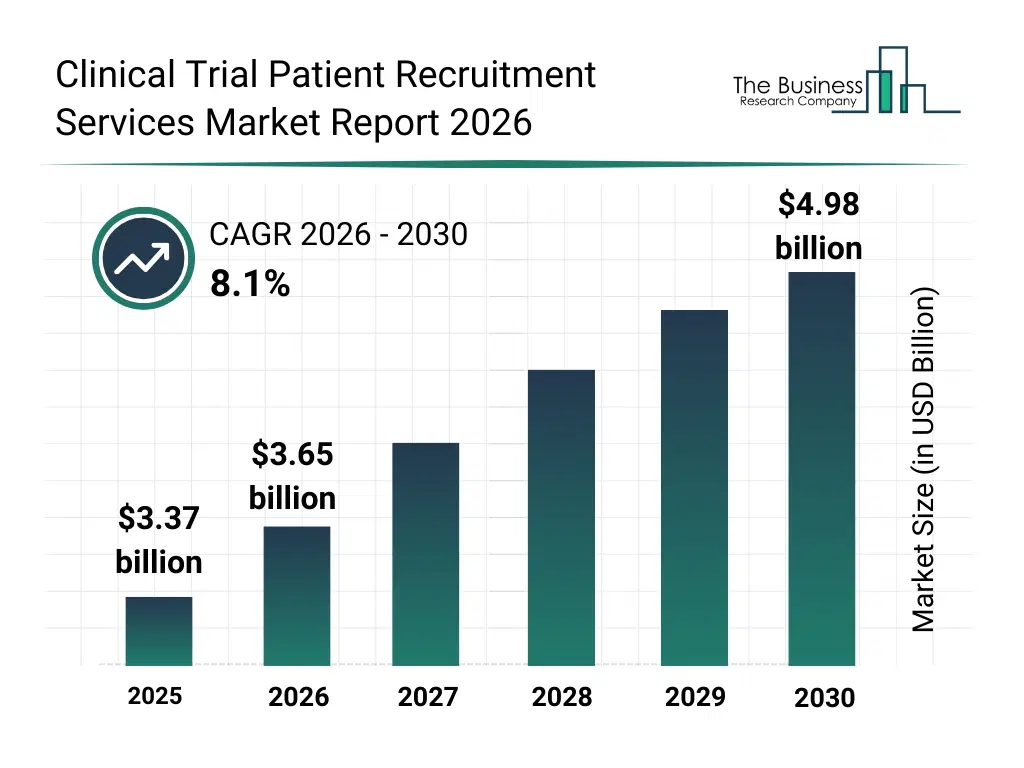
What Is The Clinical Trial Patient Recruitment Services Market Size and Share 2026?
The clinical trial patient recruitment services market size has grown strongly in recent years. It will grow from $3.37 billion in 2025 to $3.65 billion in 2026 at a compound annual growth rate (CAGR) of 8.3%. The growth in the historic period can be attributed to growth in global clinical trial volumes, increasing trial complexity across therapeutic areas, reliance on traditional site-based recruitment methods, rising regulatory requirements for patient diversity, expansion of CRO-led recruitment services.What Is The Clinical Trial Patient Recruitment Services Market Growth Forecast?
The clinical trial patient recruitment services market size is expected to see strong growth in the next few years. It will grow to $4.98 billion in 2030 at a compound annual growth rate (CAGR) of 8.1%. The growth in the forecast period can be attributed to increasing adoption of decentralized clinical trials, rising demand for faster trial timelines, growing emphasis on patient-centric trial designs, increased use of digital recruitment platforms, expanding clinical research activity in emerging markets. Major trends in the forecast period include increasing use of data-driven patient targeting, rising adoption of decentralized and virtual trial recruitment, growing focus on patient retention and engagement programs, expansion of direct-to-patient recruitment channels, enhanced use of real-world data for eligibility screening.Global Clinical Trial Patient Recruitment Services Market Segmentation
1) By Service Type: Patient Recruitment And Registry Services, Patient Retention Services, Other Service Types 2) By Phase: Phase I, Phase II, Phase III, Phase IV 3) By Therapeutic Area: Respiratory Diseases, Pain And Anesthesia, Oncology, Central Nervous System, Cardiovascular, Endocrine, Anti-Infective, Other Therapeutic Areas 4) By Recruitment Channel: Investigator-Site Led, Contract Research Organization (CRO) Led, Direct-To-Patient, Patient-Advocacy Group Partnerships 5) By End-User: Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations (CROs), Clinical Research Institutions Subsegments: 1) By Patient Recruitment And Registry Services Type: Patient Outreach, Site Identification, Eligibility Assessment, Patient Database Management 2) By Patient Retention Services Type: Patient Engagement, Appointment Reminders, Incentive Management, Progress Tracking 3) By Other Service Types: Regulatory Support, Data Management, Site Monitoring, Protocol DevelopmentWhat Is The Driver Of The Clinical Trial Patient Recruitment Services Market?
The growing number of clinical trials is expected to propel the growth of the clinical trial patient recruitment services market going forward. Clinical trials refer to research studies conducted with human participants to evaluate the safety, effectiveness, and outcomes of medical interventions such as drugs, therapies, or devices. The number of clinical trials is increasing due to advancements in biotechnology, which enable the development of more precise and innovative therapies requiring clinical evaluation. Clinical trial patient recruitment services support the growing number of clinical trials by streamlining the process of identifying, engaging, and enrolling suitable participants. They reduce delays and enhance trial efficiency by ensuring timely and adequate patient enrollment, improving overall study success and data reliability. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry (ABPI), a UK-based non-profit organization, the total number of industry-sponsored clinical trials initiated increased from 411 trials in 2022 to 426 in 2023. Therefore, the growing number of clinical trials is driving the growth of the clinical trial patient recruitment services industry.Key Players In The Global Clinical Trial Patient Recruitment Services Market
Major companies operating in the clinical trial patient recruitment services market are IQVIA Holdings Inc., Labcorp Drug Development, ICON plc, Syneos Health Inc., MMG Inc., Medpace Holdings Inc., WCG Clinical, Worldwide Clinical Trials LLC, Elligo Health Research Inc., Science 37 Holdings Inc., SubjectWell Inc., Trialbee AB, AutoCruitment LLC, StudyKIK LLC, BBK Worldwide LLC, Antidote Technologies Inc., Clariness GmbH, Trialfacts Pty Ltd, Praxis Communications LLC, Ardent Clinical Research Services Pvt Ltd.Global Clinical Trial Patient Recruitment Services Market Trends and Insights
Major companies operating in the clinical trial patient recruitment services market are focusing on the adoption of artificial intelligence, such as artificial intelligence (AI) agents, to streamline patient pre-screening, enhance eligibility matching accuracy, and accelerate enrollment timelines. An AI agent refers to a software program that can independently perceive information, make decisions, and perform tasks to achieve specific goals using artificial intelligence techniques. For instance, in January 2025, Inato, a France-based technology company, launched an AI-powered patient pre-screening tool. This expansion leverages advanced AI technologies to automate and accelerate the patient eligibility screening process, significantly reducing site workload and speeding up clinical trial enrollment while ensuring data privacy and high accuracy. This enhanced AI-powered pre-screening tool improves patient matching accuracy, streamlines site operations, and accelerates enrollment timelines for clinical trials.What Are Latest Mergers And Acquisitions In The Clinical Trial Patient Recruitment Services Market?
In December 2024, OpenClinica LLC, a US-based healthcare technology company, acquired BuildClinical LLC for an undisclosed amount. With this acquisition, OpenClinica aims to accelerate patient recruitment and integrate BuildClinical’s data-driven digital recruitment capabilities into its end-to-end clinical trial platform to speed enrolment, improve participant matching, and reduce engagement costs. BUILD CLINICAL LLC is a US-based health technology company that specialises in engaging and pre-qualifying targeted populations through digital advertising and data-driven strategies to accelerate clinical trial participant recruitment.Regional Outlook
North America was the largest region in the clinical trial patient recruitment services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Patient Recruitment Services Market?
The clinical trial patient recruitment services market includes revenues earned by entities through patient identification, pre-screening and eligibility assessment, site feasibility support, multi-channel patient outreach campaigns, and database management. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Patient Recruitment Services Market Report 2026?
The clinical trial patient recruitment services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial patient recruitment services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Patient Recruitment Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.65 billion |
| Revenue Forecast In 2035 | $4.98 billion |
| Growth Rate | CAGR of 8.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Phase, Therapeutic Area, Recruitment Channel, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | IQVIA Holdings Inc., Labcorp Drug Development, ICON plc, Syneos Health Inc., MMG Inc., Medpace Holdings Inc., WCG Clinical, Worldwide Clinical Trials LLC, Elligo Health Research Inc., Science 37 Holdings Inc., SubjectWell Inc., Trialbee AB, AutoCruitment LLC, StudyKIK LLC, BBK Worldwide LLC, Antidote Technologies Inc., Clariness GmbH, Trialfacts Pty Ltd, Praxis Communications LLC, Ardent Clinical Research Services Pvt Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
