
Clinical Trial Platform Market Report 2026
Global Outlook – By Product Type (Software Solutions, Services, Hardware), By Deployment Mode (Cloud-Based, On-Premises), By Phase (Phase I, Phase II, Phase III, Phase IV), By Application (Oncology, Cardiovascular Diseases, Neurology, Infectious Diseases, Rare Diseases), By End-User (Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Medical Device Companies, Hospitals And Clinics, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Platform Market Overview
• Clinical Trial Platform market size has reached to $2.69 billion in 2025 • Expected to grow to $5.1 billion in 2030 at a compound annual growth rate (CAGR) of 13.6% • Growth Driver: The Growing Investment In Research And Development Is Fueling The Growth Of The Market Due To The Need For Managing Complex Research, Data, And Regulatory Compliance • Market Trend: Advancements In AI-Driven Clinical Trial Platforms For Enhanced Decision-Making And Efficiency • North America was the largest region in 2025 and Asia Pacific is the fastest growing region.What Is Covered Under Clinical Trial Platform Market?
The clinical trial platform is a digital solution that optimizes the planning, management, and execution of clinical studies. It combines data collection, monitoring, and analytics to boost operational efficiency. The platform provides real-time insights, supporting faster decision-making and enhancing trial accuracy. It simplifies task management and ensures seamless coordination across study processes, resulting in quicker and more reliable outcomes in clinical research. The main product types of clinical trial platforms are software solutions, services, and hardware. Software solutions refer to digital tools and systems that streamline trial design, patient recruitment, data collection, monitoring, and analysis to enhance efficiency and compliance. The deployment modes are cloud-based and on-premises, and the clinical phases include phase I, phase II, phase III, and phase IV. It is applied in various applications such as oncology, cardiovascular diseases, neurology, infectious diseases, and rare diseases, and is used by several end-users, including pharmaceutical and biotechnology companies, contract research organizations, medical device companies, hospitals and clinics, and others.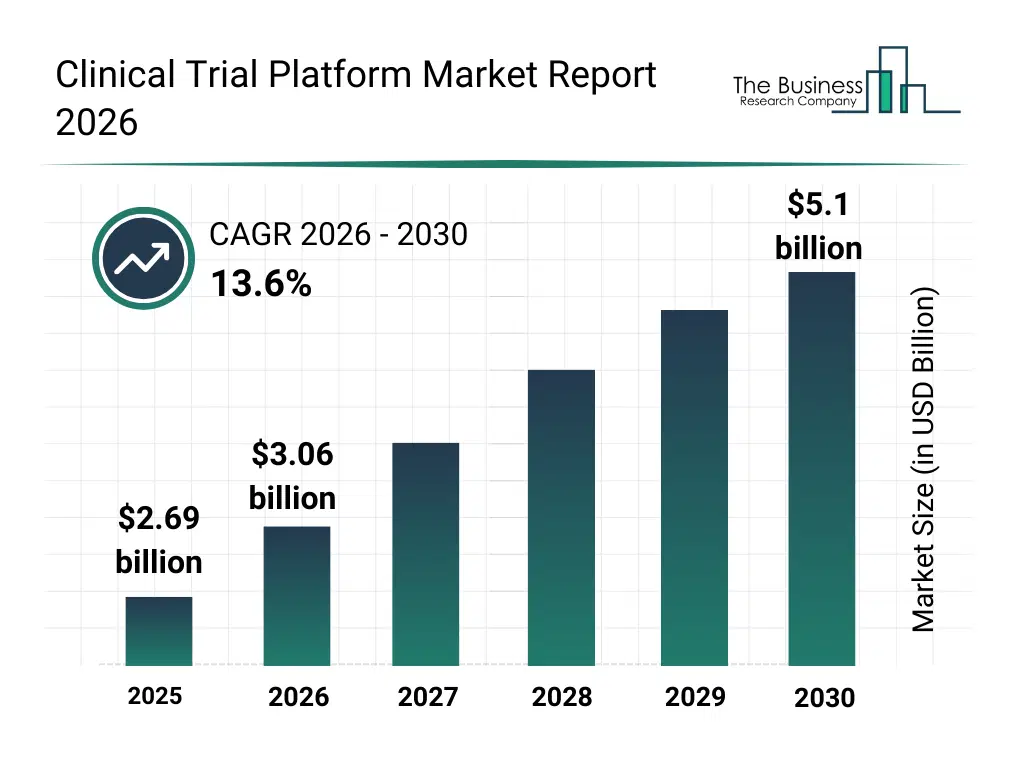
What Is The Clinical Trial Platform Market Size and Share 2026?
The clinical trial platform market size has grown rapidly in recent years. It will grow from $2.69 billion in 2025 to $3.06 billion in 2026 at a compound annual growth rate (CAGR) of 13.9%. The growth in the historic period can be attributed to increasing number of global clinical trials, rising need for operational standardization, expansion of contract research organizations, growing adoption of electronic data capture systems, increasing regulatory complexity.What Is The Clinical Trial Platform Market Growth Forecast?
The clinical trial platform market size is expected to see rapid growth in the next few years. It will grow to $5.1 billion in 2030 at a compound annual growth rate (CAGR) of 13.6%. The growth in the forecast period can be attributed to increasing investments in decentralized clinical trials, rising demand for real-time trial insights, expansion of wearable-enabled data collection, growing focus on patient-centric trial design, increasing use of predictive analytics in trial planning. Major trends in the forecast period include increasing adoption of cloud-based trial platforms, rising use of decentralized trial technologies, growing integration of AI-driven trial management, expansion of remote patient monitoring capabilities, enhanced focus on operational efficiency.Global Clinical Trial Platform Market Segmentation
1) By Product Type: Software Solutions, Services, Hardware 2) By Deployment Mode: Cloud-Based, On-Premises 3) By Phase: Phase I, Phase II, Phase III, Phase IV 4) By Application: Oncology, Cardiovascular Diseases, Neurology, Infectious Diseases, Rare Diseases 5) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Medical Device Companies, Hospitals And Clinics, Other End-Users Subsegments: 1) By Software Solutions: Electronic Data Capture (EDC) Systems, Clinical Trial Management System (CTMS), Randomization And Trial Supply Management (RTSM) Software, eConsent Software, Safety And Pharmacovigilance Software, Data Analytics And Reporting Tools, Patient Recruitment And Retention Software, Remote Monitoring And Decentralized Trial Software 2) By Services: Clinical Trial Design And Consulting Services, Site Management Organization (SMO) Services, Contract Research Organization (CRO) Services, Patient Recruitment And Retention Services, Data Management Services, Regulatory And Compliance Services, Training And Support Services 3) By Hardware: Wearable Health Monitoring Devices, Infusion Pumps And Devices, Biometric Sensors, Mobile Health Devices, Imaging And Diagnostic Devices, Laboratory Equipment And InstrumentsWhat Is The Driver Of The Clinical Trial Platform Market?
The growing investment in research and development is expected to propel the growth of the clinical trial platform market going forward. Research and development investment refers to the total expenditure by companies and organizations on innovative activities aimed at developing new technologies, products, and solutions. The rising investment in research and development is largely driven by intensifying global competition and rapid technological advancements, as organizations recognize that sustained innovation is essential for maintaining a competitive advantage in an increasingly dynamic marketplace. Growing research and development spending fuels demand for comprehensive clinical trial platforms that can efficiently manage complex research processes, data collection, and regulatory compliance requirements across multiple studies and therapeutic areas. For instance, in January 2025, according to People’s Daily Online, the official online news portal of the China-based People’s Daily newspaper, reported that China’s expenditure on R&D exceeds $495 billion in 2024, up 8.3% from 2023. Therefore, growing investment in research and development is driving the growth of the clinical trial platform industry.Key Players In The Global Clinical Trial Platform Market
Major companies operating in the clinical trial platform market are Oracle Corporation, Tata Consultancy Services Limited, IQVIA Holdings Inc., Labcorp Holdings Inc., ICON Public Limited Company, Veeva Systems Inc., Medpace Holdings Inc., Clario Inc., Medidata Solutions Inc., Medable Inc., South Australian Health and Medical Research Institute, Medrio Inc., Ciwit B.V., Obvio Health USA Inc., Curebase Inc., DATATRAK International Inc., OpenClinica LLC, Infiuss Health Inc., PhaseV Inc., Everest Global Inc.Global Clinical Trial Platform Market Trends and Insights
Major companies operating in the clinical trial platform market are focusing on developing advanced solutions such as machine learning-based clinical trial platforms to enable predictive analytics for better decision-making throughout the clinical trial process. A machine learning-based clinical trial platform uses artificial intelligence to analyze clinical data and forecast patient eligibility, recruitment patterns, and trial results while optimizing study design and accelerating participant enrollment for enhanced efficiency and informed decision-making. For instance, in July 2025, PhaseV, a US-based provider of AI/ML-driven clinical development solutions, launched the ClinOps AI platform, a next-generation solution designed to transform clinical trial operations by enabling precision-guided site selection and dynamic real-time performance monitoring. It leverages causal machine learning and real-world patient-level data to enable trial teams to make quicker, more informed decisions by providing deeper insights into site performance and patient variability, enhancing recruitment outcomes, participant diversity, and overall operational efficiency across the trial lifecycle.What Are Latest Mergers And Acquisitions In The Clinical Trial Platform Market?
In December 2024, OpenClinica Inc., a US-based provider of software-as-a-service clinical trial solutions, acquired BuildClinical Inc. for an undisclosed amount. Through this acquisition, OpenClinica aims to accelerate patient recruitment and reduce enrollment bottlenecks in clinical trials by leveraging BuildClinical’s data-driven digital engagement and enrollment strategies. BuildClinical Inc.is a US-based health technology company that provides a clinical trial platform to accelerate patient recruitment for academic research studies.Regional Outlook
North America was the largest region in the clinical trial platform market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Platform Market?
The clinical trial platform market consists of revenues earned by entities by providing services such as patient recruitment and retention, data management and analytics, remote monitoring and telemedicine support, regulatory compliance and trial management, and training and consulting services. The market value includes the value of related products sold by the service provider or included within the service offering. The clinical trial platform market also includes sales of electronic data capture (EDC) systems, wearable health monitoring devices, biosensors, and clinical trial software solutions. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream service providers, distributors, and healthcare institutions) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Platform Market Report 2026?
The clinical trial platform market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial platform industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Platform Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.06 billion |
| Revenue Forecast In 2035 | $5.1 billion |
| Growth Rate | CAGR of 13.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Deployment Mode, Phase, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Oracle Corporation, Tata Consultancy Services Limited, IQVIA Holdings Inc., Labcorp Holdings Inc., ICON Public Limited Company, Veeva Systems Inc., Medpace Holdings Inc., Clario Inc., Medidata Solutions Inc., Medable Inc., South Australian Health and Medical Research Institute, Medrio Inc., Ciwit B.V., Obvio Health USA Inc., Curebase Inc., DATATRAK International Inc., OpenClinica LLC, Infiuss Health Inc., PhaseV Inc., Everest Global Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
