
Clinical Trial Services Market Report 2026
Global Outlook – By Service Type (Clinical Trial Management (CTM), Planning And Design, Project Management, Monitoring And Data Management, Site Management, Regulatory Services, Regulatory Submission, Consulting, Protocol And Safety Consulting, Other Service Types ), By Phase (Phase I, Phase II, Phase III, Phase IV ), By Therapeutic Area (Oncology, Cardiology, Neurology, Infectious Diseases, Immunology, Respiratory, Dermatology ), By End-User (Pharmaceutical And Biotechnology Companies, Medical Device Companies, Contract Research Organizations, Academic And Research Institutes) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Services Market Overview
• Clinical Trial Services market size has reached to $56.17 billion in 2025 • Expected to grow to $95.23 billion in 2030 at a compound annual growth rate (CAGR) of 11.1% • Growth Driver: Impact Of Growing Demand For Personalized Medicine On The Market • Market Trend: Advancements In Data Solutions To Accelerate The Clinical Trial Process • North America was the largest region and fastest growing region.What Is Covered Under Clinical Trial Services Market?
Clinical trial services refer to the comprehensive range of activities and support provided throughout the process of designing, conducting, and analyzing clinical trials. They are used for assessing the safety, efficacy, and regulatory compliance of new medical treatments, drugs, or devices. These services include patient recruitment, data collection, monitoring, regulatory submissions, statistical analysis, and final reporting, ensuring that clinical studies are conducted in compliance with regulatory requirements and contribute to the advancement of medical science. The main service types in clinical trial services are clinical trial management (CTM), planning and design, project management, monitoring and data management, site management, regulatory services, regulatory submission, consulting, protocol and safety consulting, and other service types. Clinical trial management (CTM) involves overseeing all aspects of a clinical trial, including scheduling, resource management, regulatory compliance, and ensuring the trial runs smoothly. The phases of clinical trials include Phase I, Phase II, Phase III, and Phase IV. Therapeutic areas covered in clinical trials include oncology, cardiology, neurology, infectious diseases, immunology, respiratory, and dermatology. The key end-users of clinical trial services are pharmaceutical and biotechnology companies, medical device companies, contract research organizations, and academic and research institutes.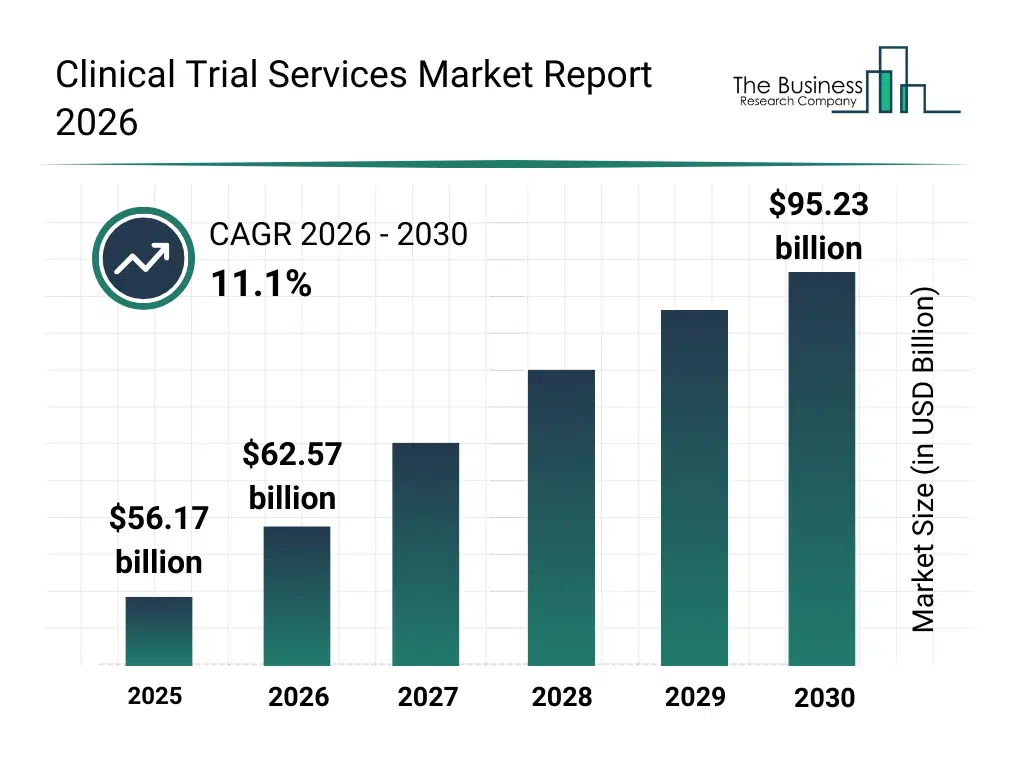
What Is The Clinical Trial Services Market Size and Share 2026?
The clinical trial services market size has grown rapidly in recent years. It will grow from $56.17 billion in 2025 to $62.57 billion in 2026 at a compound annual growth rate (CAGR) of 11.4%. The growth in the historic period can be attributed to increasing pharmaceutical R&D spending, expansion of global clinical trial pipelines, rising complexity of regulatory requirements, growth in multi-therapeutic clinical studies, availability of specialized CRO expertise.What Is The Clinical Trial Services Market Growth Forecast?
The clinical trial services market size is expected to see rapid growth in the next few years. It will grow to $95.23 billion in 2030 at a compound annual growth rate (CAGR) of 11.1%. The growth in the forecast period can be attributed to increasing adoption of decentralized and hybrid trials, rising focus on patient recruitment optimization, expansion of real-world evidence integration, growing use of advanced analytics in trial execution, increasing demand for faster trial timelines. Major trends in the forecast period include increasing outsourcing of end-to-end clinical trial services, rising demand for data-driven trial monitoring, growing adoption of risk-based monitoring models, expansion of patient-centric trial services, enhanced focus on regulatory and compliance support.Global Clinical Trial Services Market Segmentation
1) By Service Type: Clinical Trial Management (CTM), Planning And Design, Project Management, Monitoring And Data Management, Site Management, Regulatory Services, Regulatory Submission, Consulting, Protocol And Safety Consulting, Other Service Types 2) By Phase: Phase I, Phase II, Phase III, Phase IV 3) By Therapeutic Area: Oncology, Cardiology, Neurology, Infectious Diseases, Immunology, Respiratory, Dermatology 4) By End-User: Pharmaceutical And Biotechnology Companies, Medical Device Companies, Contract Research Organizations, Academic And Research Institutes Subsegments: 1) By Clinical Trial Management (CTM): Study Setup And Execution, Recruitment And Patient Enrollment, Trial Monitoring And Reporting, Trial Close-Out And Data Lock 2) By Planning And Design: Protocol Development, Clinical Trial Design, Feasibility Analysis, Site Selection And Initiation 3) By Project Management: Budgeting And Resource Allocation, Risk Management, Timeline Management, Stakeholder Coordination 4) By Monitoring And Data Management: Data Collection And Validation, Statistical Analysis, Quality Control And Assurance, Data Reporting And Interpretation 5) By Site Management: Site Initiation And Training, Site Monitoring And Audits, Site Close-Out And Documentation 6) By Regulatory Services: Regulatory Compliance And Consultation, Regulatory Strategy Development, Preparation For Audits And Inspections 7) By Regulatory Submission: Submission Of Investigational New Drug (IND) Applications, New Drug Application (NDA) Submission, Submission Of Clinical Study Reports (CSRs) 8) By Consulting: Strategy And Planning Consulting, Operational And Performance Consulting, Data Analytics And Interpretation Consulting 9) By Protocol And Safety Consulting: Clinical Trial Protocol Consulting, Safety Management And Risk Assessment, Safety Reporting And Adverse Event Management 10) By Other Service Types: Medical Writing, Biostatistics And Data Analysis Services, Patient Recruitment Services, Post-Approval StudiesWhat Is The Driver Of The Clinical Trial Services Market?
Growing demand for personalized medicine is expected to propel the growth of the clinical trial services market going forward. Personalized medicine refers to a medical approach that tailors treatment and healthcare strategies to individual patients based on their genetic, environmental, and lifestyle factors. Personalized medicine is increasing due to advances in genomic technologies, the rise of targeted therapies, growing patient demand for tailored treatments, and the integration of AI, biomarkers, and real-world data to improve treatment efficacy and safety. Clinical trial services play a crucial role in advancing personalized medicine by facilitating the development, testing, and validation of targeted therapies through the use of genetic data, biomarkers, and tailored treatment protocols to ensure more effective and individualized patient outcomes. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based hospital and health care organization, in 2023, the FDA approved 16 new personalized treatments for rare disease patients, up from six in 2022. Therefore, the growing demand for personalized medicine is driving the growth of the clinical trial services industry.Key Players In The Global Clinical Trial Services Market
Major companies operating in the clinical trial services market are IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc, Parexel International Corporation, Medpace Holdings Inc, BioClinica Inc, Worldwide Clinical Trials Inc, Medidata Solutions Inc, Precision Medicine Group Inc, Celerion Inc, MakroCare Clinical Research Limited, PCI Pharma Services, Ecron Acunova Limited, PPD LLC., Clinipace Inc., Syntactx LLC., Veristat LLC., AutoCruitment Inc.Global Clinical Trial Services Market Trends and Insights
Major companies operating in the clinical trial services market are developing data solutions to accelerate the clinical trial process. Data solutions refer to technologies and tools that manage, analyze, and optimize data to enhance efficiency, accuracy, and decision-making in processes such as clinical trials. For instance, in July 2024, Laboratory Corporation, a US-based life sciences company, launched Labcorp Global Trial Connect, a comprehensive suite of central laboratory solutions designed to streamline and enhance the clinical trial process. This integrated platform connects central laboratories, clinical trial sites, and study sponsors, enabling seamless communication and real-time data sharing. This helps to streamline the clinical trial process, reducing delays and improving trial timelines. It enhances patient access by utilizing advanced technologies to identify suitable participants and facilitate remote monitoring.What Are Latest Mergers And Acquisitions In The Clinical Trial Services Market?
In February 2024, ICON plc, an Ireland-based clinical research company, acquired Clinical Research Management Inc. for an undisclosed amount. With this acquisition, ICON aims to enhance its clinical trial services capabilities by expanding its expertise in clinical research management, improving operational efficiency, and providing a broader range of solutions to its clients. Clinical Research Management Inc. is a US-based company that provides a range of services in the field of clinical research.Regional Outlook
North America was the largest region in the clinical trial services market in 2025. North America is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Services Market?
The clinical trial services market includes revenues earned by entities by project management, clinical trial documentation, and safety monitoring. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Services Market Report 2026?
The clinical trial services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $62.57 billion |
| Revenue Forecast In 2035 | $95.23 billion |
| Growth Rate | CAGR of 11.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Phase, Therapeutic Area, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc, Parexel International Corporation, Medpace Holdings Inc, BioClinica Inc, Worldwide Clinical Trials Inc, Medidata Solutions Inc, Precision Medicine Group Inc, Celerion Inc, MakroCare Clinical Research Limited, PCI Pharma Services, Ecron Acunova Limited, PPD LLC., Clinipace Inc., Syntactx LLC., Veristat LLC., AutoCruitment Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
