Clinical Trial Software Market Report 2026

Clinical Trial Software Market Report 2026
Global Outlook – By Deployment (On-Premises, Web-Based Clinical Trial Software, Cloud-Based Clinical Trial Software, Other Deployments), By Software (Electronic Data Capture (EDC), Electronic Clinical Outcome Assessment (eCOA) Or Electronic Patient-Reported Outcome (ePRO), Electronic Informed Consent), By End-User (Pharmaceutical And Biotechnology Companies, Contract Research Organizations (CROs), Medical Device Manufacturers, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Software Market Overview
• Clinical Trial Software market size has reached to $1.32 billion in 2025 • Expected to grow to $2.53 billion in 2030 at a compound annual growth rate (CAGR) of 13.8% • Growth Driver: Impact Of Increasing Research And Development Activities On The Growth Of The Market • Market Trend: AstraZeneca's Evinova Platform Enhances Efficiency And Patient Engagement Through AI Technology • North America was the largest region in 2025 and Europe is the fastest growing region.What Is Covered Under Clinical Trial Software Market?
Clinical trial software refers to specialized applications that support the planning, execution, and management of clinical trials. It is used for data management, patient recruitment, regulatory compliance, and monitoring trial progress to ensure accuracy and efficiency in the research process. The main deployment types of clinical trial software are on-premises, web-based clinical trial software, cloud-based clinical trial software, and other deployments. On-premises deployment involves installing and running clinical trial management software on the organization's local servers and computers. This approach provides complete control over the software and data but requires significant IT resources for maintenance and updates. The various software involved are electronic data capture (EDC), electronic clinical outcome assessment (ECOA) or electronic patient-reported outcome (EPRO), and electronic informed consent and are used by several end-users such as pharmaceutical and biotechnology companies, contract research organizations (CROs), medical device manufacturers, and others.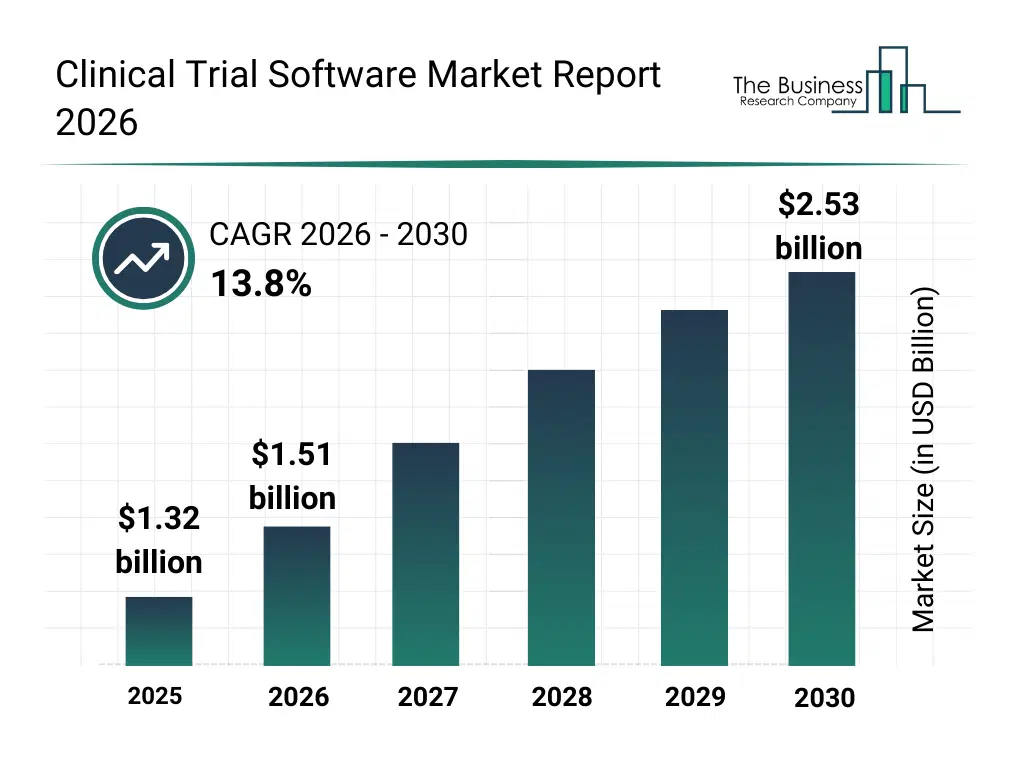
What Is The Clinical Trial Software Market Size and Share 2026?
The clinical trial software market size has grown rapidly in recent years. It will grow from $1.32 billion in 2025 to $1.51 billion in 2026 at a compound annual growth rate (CAGR) of 14.1%. The growth in the historic period can be attributed to increasing digitization of clinical trial processes, rising volume of clinical trial data, growing need for real-time trial visibility, expansion of global clinical research activities, availability of advanced software platforms.What Is The Clinical Trial Software Market Growth Forecast?
The clinical trial software market size is expected to see rapid growth in the next few years. It will grow to $2.53 billion in 2030 at a compound annual growth rate (CAGR) of 13.8%. The growth in the forecast period can be attributed to increasing adoption of virtual and decentralized trials, rising investments in saas-based trial solutions, growing integration of ai in trial workflows, expansion of remote patient monitoring, increasing regulatory focus on data integrity. Major trends in the forecast period include increasing adoption of cloud-based clinical trial software, rising use of AI-powered data analytics tools, growing demand for decentralized trial software platforms, expansion of integrated edc and ctms solutions, enhanced focus on data security and compliance.Global Clinical Trial Software Market Segmentation
1) By Deployment: On-Premises, Web-Based Clinical Trial Software, Cloud-Based Clinical Trial Software, Other Deployments 2) By Software: Electronic Data Capture (EDC), Electronic Clinical Outcome Assessment (eCOA) Or Electronic Patient-Reported Outcome (ePRO), Electronic Informed Consent 3) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations (CROs), Medical Device Manufacturers, Other End-Users Subsegments: 1) By On-Premises: Enterprise Clinical Trial Management Systems, Site-Based Clinical Trial Solutions 2) By Web-Based Clinical Trial Software: Electronic Data Capture (EDC) Systems, Clinical Trial Management Systems (CTMS), Randomization And Trial Supply Management (RTSM) 3) By Cloud-Based Clinical Trial Software: Software-as-a-Service (SaaS) Platforms, AI-Powered Clinical Trial Solutions, Remote Monitoring And Decentralized Trial Systems 4) By Other Deployments: Hybrid Clinical Trial Solutions, Custom-Built Clinical Trial SoftwareWhat Is The Driver Of The Clinical Trial Software Market?
Increasing research and development activities is expected to propel the growth of the clinical trial software market. Research and development activities refer to the systematic process of investigating, designing, and testing new products or technologies to innovate and improve existing solutions or create entirely new offerings. Research and development activities are increasing due to the growing demand for innovation across industries, fueled by advancements in technology, competitive market pressures, and the need for sustainable solutions, which compel organizations to invest more resources in developing new products, processes, and services. Clinical trial software is used for research and development activities by streamlining the planning, execution, and management of clinical trials, which are crucial for testing new drugs and therapies. For instance, in 2023, according to Office for National Statistics, a UK-based, government agency, the UK government’s net spending on research and development (R&D) increased to £17.4 ($22.1 billion) billion in 2023, up from £16.1 billion ($20.45 billion) in 2022, marking an 8.2% rise. Therefore, the increasing research and development activities drive the growth of the clinical trial software industry.Key Players In The Global Clinical Trial Software Market
Major companies operating in the clinical trial software market are Oracle Corporation, Veeva Systems, Medidata Solutions, Signant Health Inc., Clario Inc., Greenphire Inc., Medrio Inc., Arisglobal LLC, Anju Software Inc., ClinCapture Inc., OpenClinica LLC, Castor, Viedoc, Clinion, OmniComm (TrialMaster), Curebase, Clinevo, RealTime-CTMS, REDCap Cloud, Zelta, Trialytix, Florence eBinders, Dot Compliance, ClinPlus CTMS, nQuery, Jeeva Clinical TrialsGlobal Clinical Trial Software Market Trends and Insights
Major companies operating in the clinical trial software market are focused on developing innovative technologies, such as unified trial solutions to enhance data collection and integration, improve patient engagement, streamline trial processes, reduce costs, and accelerate the overall drug development timeline. Unified Trial Solution refers to a comprehensive digital platform designed to streamline and enhance the clinical trial process. For instance, in November 2023, AstraZeneca plc, a UK-based pharmaceutical company, introduced Evinova to enable AI in clinical research. This innovative platform boasts unique features such as automated patient recruitment, which leverages AI algorithms to swiftly match candidates with trial criteria, thereby significantly reducing enrollment times. It also includes predictive modeling tools that analyze historical and real-time data to forecast patient responses, enhancing the design and efficiency of trials. Its user-friendly interface fosters better patient engagement by providing digital platforms for communication and feedback, allowing participants to share their experiences more easily.What Are Latest Mergers And Acquisitions In The Clinical Trial Software Market?
In October 2023, Certara Inc., a US-based software company, acquired Formedix LLC for an undisclosed amount. With this acquisition, Certara aims to enhance its clinical trial automation capabilities by integrating Formedix’s expertise in data standardization and CDISC compliance, streamlining regulatory submissions, improving trial efficiency, and accelerating drug development processes for pharmaceutical companies and regulatory agencies. Formedix LLC is a US-based company that specializes in providing cloud-based software solutions designed to streamline clinical trial processes.Regional Outlook
North America was the largest region in the clinical trial software market in 2025. Europe is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Software Market?
The clinical trial software market includes revenues earned by entities by providing services such as site management and monitoring, patient recruitment and retention solutions, clinical trial management systems (CTMS), and risk-based monitoring. The market value includes the value of related goods sold by the service provider or included within the service offering. The clinical trial software market consists of sales of electronic data capture (EDC) systems, patient recruitment platforms, randomization and trial supply management (RTSM), and risk-based monitoring solutions. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Software Market Report 2026?
The clinical trial software market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial software industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Software Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.51 billion |
| Revenue Forecast In 2035 | $2.53 billion |
| Growth Rate | CAGR of 14.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Deployment, Software, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Oracle Corporation, Veeva Systems, Medidata Solutions, Signant Health Inc., Clario Inc., Greenphire Inc., Medrio Inc., Arisglobal LLC, Anju Software Inc., ClinCapture Inc., OpenClinica LLC, Castor, Viedoc, Clinion, OmniComm (TrialMaster), Curebase, Clinevo, RealTime-CTMS, REDCap Cloud, Zelta, Trialytix, Florence eBinders, Dot Compliance, ClinPlus CTMS, nQuery, Jeeva Clinical Trials |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Clinical Trial Software market was valued at $1.32 billion in 2025, increased to $1.51 billion in 2026, and is projected to reach $2.53 billion by 2030.
The global Clinical Trial Software market is expected to grow at a CAGR of 13.8% from 2026 to 2035 to reach $2.53 billion by 2035.
Some Key Players in the Clinical Trial Software market Include, Oracle Corporation, Veeva Systems, Medidata Solutions, Signant Health Inc., Clario Inc., Greenphire Inc., Medrio Inc., Arisglobal LLC, Anju Software Inc., ClinCapture Inc., OpenClinica LLC, Castor, Viedoc, Clinion, OmniComm (TrialMaster), Curebase, Clinevo, RealTime-CTMS, REDCap Cloud, Zelta, Trialytix, Florence eBinders, Dot Compliance, ClinPlus CTMS, nQuery, Jeeva Clinical Trials .
Major trend in this market includes: AstraZeneca's Evinova Platform Enhances Efficiency And Patient Engagement Through AI Technology. For further insights on this market.
Request for SampleNorth America was the largest region in the clinical trial software market in 2025. Europe is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial software market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
