
Clinical Trial Supplies Market Report 2026
Global Outlook – By Services (Logistics And Distribution, Storage And Retention, Supply Chain Management, Packaging, Labeling, And Blinding, Comparator Sourcing), By Clinical Phases (Phase I, Phase II, Phase III, Phase IV), By Therapeutic Use (Oncology, Central Nervous System (CNS), Cardiovascular, Infectious Disease, Metabolic Disorders, Other Therapeutic Uses), By End User (Pharmaceutical And Biotech companies, Contract Research Organization (CRO)) – Market Size, Trends, Strategies, and Forecast to 2035
Clinical Trial Supplies Market Overview
• Clinical Trial Supplies market size has reached to $2.86 billion in 2025 • Expected to grow to $4.33 billion in 2030 at a compound annual growth rate (CAGR) of 8.7% • Growth Driver: The Surge In Registered Clinical Trials Driving Growth In The Clinical Trial Supplies Market • Market Trend: The Integration Of Real-World Evidence Platforms Shapes The Landscape Of The Clinical Trial Supplies Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Supplies Market?
Clinical trial supplies refer to the materials, products, and equipment used during clinical trials. Clinical trial supplies play a crucial role in the successful execution of clinical trials by providing the necessary resources for testing and evaluation. The main types of services of clinical trial supplies are logistics and distribution, storage and retention, supply chain management, packaging, labeling, blinding manufacturing, and comparator sourcing. Logistics and distribution refer to the management and transportation of trial supplies from manufacturing to trial sites, ensuring timely availability and clinical trial supplies are efficiently delivered to the designated sites, ensuring the timely availability of necessary materials for the trial. These supplies are utilized in different clinical phases, including Phase I, Phase II, Phase III, and Phase IV, for therapeutic uses such as oncology, the central nervous system (CNS), cardiovascular disease, infectious diseases, metabolic disorders, and others. The end users includes pharmaceutical and biotech companies, contract research organizations (CROs), and medical device companies.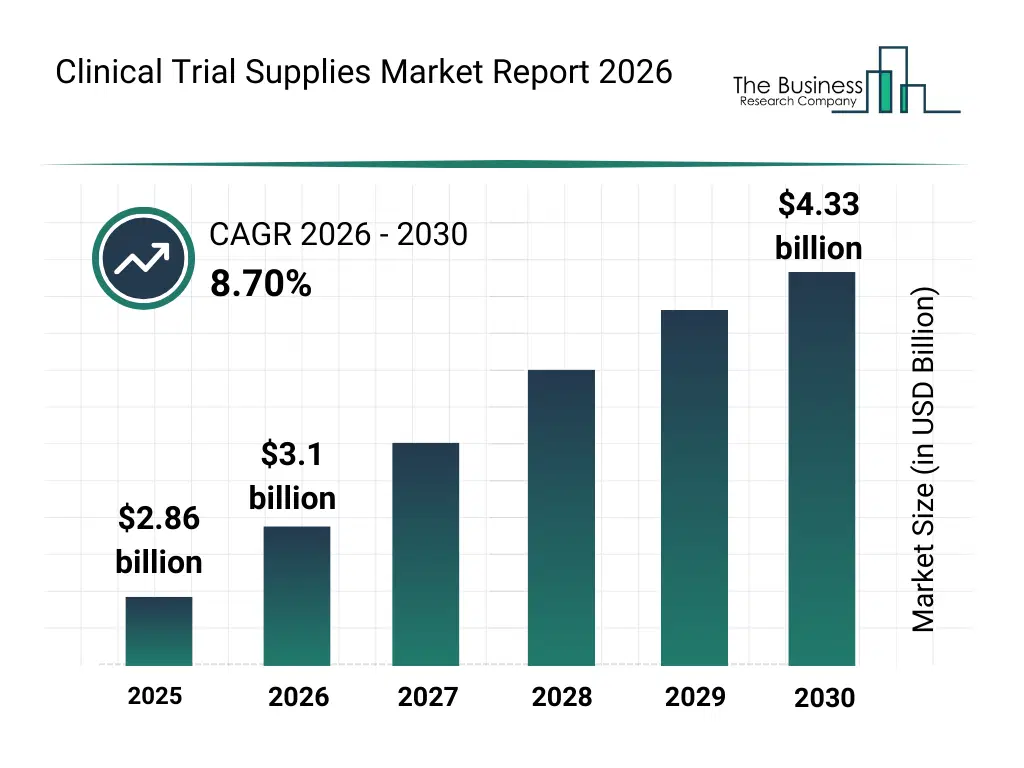
What Is The Clinical Trial Supplies Market Size and Share 2026?
The clinical trial supplies market size has grown strongly in recent years. It will grow from $2.86 billion in 2025 to $3.1 billion in 2026 at a compound annual growth rate (CAGR) of 8.3%. The growth in the historic period can be attributed to increase in global clinical trial activity, expansion of multicenter trial designs, growth of biologics and specialty drugs, rising outsourcing to CROs, development of cold-chain logistics infrastructure.What Is The Clinical Trial Supplies Market Growth Forecast?
The clinical trial supplies market size is expected to see strong growth in the next few years. It will grow to $4.33 billion in 2030 at a compound annual growth rate (CAGR) of 8.7%. The growth in the forecast period can be attributed to increasing complexity of clinical trial protocols, expansion of decentralized and virtual trials, rising focus on patient-centric trial models, growing investments in digital trial management tools, increasing regulatory scrutiny across regions. Major trends in the forecast period include increasing adoption of integrated supply chain platforms, rising use of temperature-controlled logistics solutions, growing demand for end-to-end trial visibility, expansion of adaptive packaging and labeling services, enhanced focus on compliance and traceability.Global Clinical Trial Supplies Market Segmentation
1) By Services: Logistics And Distribution, Storage And Retention, Supply Chain Management, Packaging, Labeling, And Blinding, Comparator Sourcing 2) By Clinical Phases: Phase I, Phase II, Phase III, Phase IV 3) By Therapeutic Use: Oncology, Central Nervous System (CNS), Cardiovascular, Infectious Disease, Metabolic Disorders, Other Therapeutic Uses 4) By End User: Pharmaceutical And Biotech companies, Contract Research Organization (CRO) Subsegments: 1) By Logistics And Distribution: Transportation, Inventory Management, Temperature-Controlled Shipping, Customs Clearance 2) By Storage And Retention: Short-term Storage, Long-term Storage, Controlled Environment Storage 3) By Supply Chain Management: Planning And Forecasting, Supplier Management, Risk Management, Compliance Management 4) By Packaging, Labeling, And Blinding: Primary Packaging, Secondary Packaging, Label Design And Printing, Blinding Techniques 5) By Comparator Sourcing: Sourcing Of Reference Products, Quality Control And Assurance, Regulatory Compliance For ComparatorsWhat Is The Driver Of The Clinical Trial Supplies Market?
The increasing number of registered clinical trials is expected to propel the growth of the clinical trial supplies market going forward. Clinical trials are research studies conducted with human participants to evaluate the safety and effectiveness of new medical treatments, interventions, or diagnostic procedures. Clinical trial supplies, including pharmaceuticals, medical devices, and other essential materials, are crucial for conducting successful clinical trials, supporting the research process, ensuring accurate data collection, and maintaining the patients' safety. For instance, in May 2023, according to ClinicalTrials.gov, a part of the National Institutes of Health (NIH), a US-based government agency responsible for conducting and supporting medical research, in 2023, around 437,533 clinical trials were registered, compared to 399,499 registered studies in 2022 in all 50 states of the United States and across 221 countries. Therefore, the increasing number of registered clinical trials is driving the growth of the clinical trial supplies industry.Key Players In The Global Clinical Trial Supplies Market
Major companies operating in the clinical trial supplies market are Thermo Fisher Scientific Inc., IQVIA, Eurofins Scientific SE, Parexel International Corporation, ICON PLC, Catalent Inc., Intertek Group PLC, Recipharm AB, World Courier, Almac Group Ltd., Piramal Pharma Solutions, Clinigen Group PLC, Movianto GmbH, Marken Limited, PCI Pharma Services, Rubicon Research Private Limited, Bionical Ltd., Durbin PLC, SIRO Clinpharm Pvt. Ltd., Biocair International Ltd., Ancillare LP., Myonex, Klifo A/S, Alium Medical Limited, ADAllen Pharma, Sharp Services LLCGlobal Clinical Trial Supplies Market Trends and Insights
Major companies operating in the clinical trial supplies market are adopting innovative technologies to transform healthcare decision-making, drug development, and patient care by leveraging real-world data to generate evidence-based insights and sustain their position in the market. For instance, in April 2023, Komodo Health, a US-based healthcare technology company that specializes in services related to patient recruitment, site selection, and real-time data monitoring for clinical trials, launched MapEnhance, a network of specialty data partners integrated into a real-world evidence (RWE) technology platform. MapEnhance empowers customers with a comprehensive and detailed real-world perspective on clinical events and patient populations, enabling unprecedented insight. MapEnhance includes precision molecular diagnostics for various therapeutic areas, high-volume standard laboratory diagnostics for earlier disease visibility, and electronic medical records capturing clinical encounters. It also offers inpatient and outpatient facility chargemasters for detailed cost and utilization insights across different hospital settings.What Are Latest Mergers And Acquisitions In The Clinical Trial Supplies Market?
In May 2024, Myonex, a US-based pharmaceutical company, acquired Creapharm’s pharmaceutical services for an undisclosed amount. As a result of the acquisition, Creapharm's capabilities in clinical packaging, distribution, commercial packaging, and bioservices will now operate under the name Creapharm, a Myonex company. This merger enhances flexibility and strengthens service offerings for pharmaceutical and biotech companies at both clinical and commercial stages, including those handling Advanced Therapies. Creapharm is a France-based manufacturer of clinical trial supplies.Regional Outlook
North America was the largest region in the clinical trial supplies market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Clinical Trial Supplies Market?
The clinical trial supplies market consists of revenues earned by entities by providing quality control, regulatory compliance, and return and destruction management services. The market value includes the value of related goods sold by the service provider or included within the service offering. The clinical trial supplies market also includes sales of investigational products, comparator products, and clinical trial kits. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Clinical Trial Supplies Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.1 billion |
| Revenue Forecast In 2035 | $4.33 billion |
| Growth Rate | CAGR of 8.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Services, Clinical Phases, Therapeutic Use, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., IQVIA, Eurofins Scientific SE, Parexel International Corporation, ICON PLC, Catalent Inc., Intertek Group PLC, Recipharm AB, World Courier, Almac Group Ltd., Piramal Pharma Solutions, Clinigen Group PLC, Movianto GmbH, Marken Limited, PCI Pharma Services, Rubicon Research Private Limited, Bionical Ltd., Durbin PLC, SIRO Clinpharm Pvt. Ltd., Biocair International Ltd., Ancillare LP., Myonex, Klifo A/S, Alium Medical Limited, ADAllen Pharma, Sharp Services LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
