Clinical Trial Support Services Market Report 2026

Clinical Trial Support Services Market Report 2026
Global Outlook – By Service (Clinical Trial Site Management, Data Management, Patient Recruitment Management, Administrative Staff, Institutional Review Board (IRB), Other Services), By Phases (Preclinical Studies, Phase I, Phase II, Phase III, Phase IV), By Application (Oncology, Central Nervous System (CNS) And Mental Disorders, Cardiovascular Diseases, Infectious Diseases, Bood Disorders, Other Applications) - Market Size, Trends, And Global Forecast 2026-2035
Clinical Trial Support Services Market Overview
• Clinical Trial Support Services market size has reached to $24.48 billion in 2025 • Expected to grow to $35.97 billion in 2030 at a compound annual growth rate (CAGR) of 7.9% • Growth Driver: Tailored Treatments Drive Rising Demand For Support Services • Market Trend: Patient-Centric Trials Revolutionize Clinical Research • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Trial Support Services Market?
Clinical trial support services refer to a range of specialized services that assist in the planning, execution, and management of clinical trials, ensuring regulatory compliance, data integrity, patient recruitment, and overall trial efficiency. These services help streamline the drug development process, reduce operational complexities, and enhance the quality and reliability of trial outcomes. The main services in the clinical trial support services market are clinical trial site management, data management, patient recruitment management, administrative staff, institutional review board (IRB), and others. Clinical trial site management refers to the coordination and oversight of all activities at a clinical trial site to ensure compliance with regulatory requirements, protocol adherence, and efficient trial execution. The different phases encompass preclinical studies, Phase I, Phase II, Phase III, and Phase IV, covering various applications such as oncology, central nervous system (CNS) and mental disorders, cardiovascular diseases, infectious diseases, blood disorders, and others.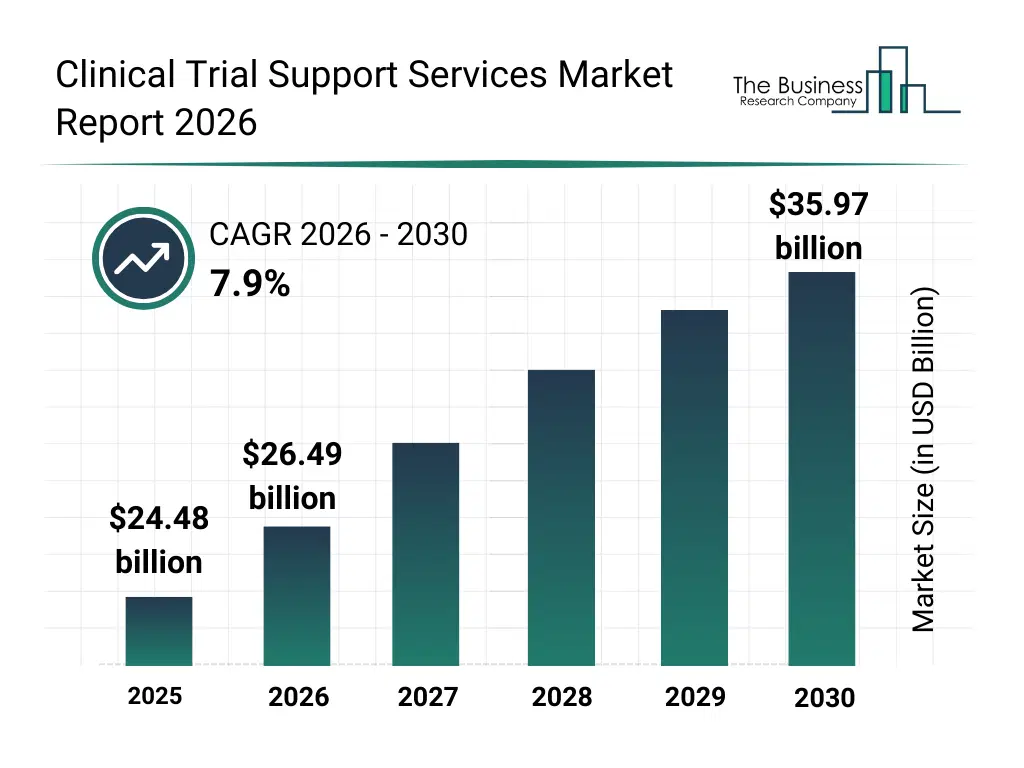
What Is The Clinical Trial Support Services Market Size and Share 2026?
The clinical trial support services market size has grown strongly in recent years. It will grow from $24.48 billion in 2025 to $26.49 billion in 2026 at a compound annual growth rate (CAGR) of 8.2%. The growth in the historic period can be attributed to growing volume of clinical research activity, increasing complexity of regulatory requirements, rising reliance on outsourced trial services, expansion of multi-site clinical trials, increasing demand for skilled trial personnel.What Is The Clinical Trial Support Services Market Growth Forecast?
The clinical trial support services market size is expected to see strong growth in the next few years. It will grow to $35.97 billion in 2030 at a compound annual growth rate (CAGR) of 7.9%. The growth in the forecast period can be attributed to increasing adoption of decentralized trial models, rising use of AI-enabled trial analytics, expansion of digital patient engagement tools, growing investments in end-to-end trial support platforms, increasing focus on operational efficiency optimization. Major trends in the forecast period include increasing adoption of centralized data management services, rising demand for advanced patient recruitment solutions, growing use of remote monitoring and virtual trial support, expansion of integrated trial management platforms, enhanced focus on regulatory compliance support.Global Clinical Trial Support Services Market Segmentation
1) By Service: Clinical Trial Site Management, Data Management, Patient Recruitment Management, Administrative Staff, Institutional Review Board (IRB), Other Services 2) By Phases: Preclinical Studies, Phase I, Phase II, Phase III, Phase IV 3) By Application: Oncology, Central Nervous System (CNS) And Mental Disorders, Cardiovascular Diseases, Infectious Diseases, Bood Disorders, Other Applications Subsegments: 1) By Clinical Trial Site Management: Site Selection, Site Initiation, Site Monitoring, Site Close-Out 2) By Data Management: Data Collection, Data Validation, Electronic Data Capture (EDC), Statistical Analysis 3) By Patient Recruitment Management: Patient Screening, Enrollment Strategy, Retention Services 4) By Administrative Staff: Clinical Research Coordinators, Project Managers, Regulatory Affairs Specialists 5) By Institutional Review Board (IRB): Ethical Review, Protocol Review, Compliance Monitoring 6) By Other Services: Regulatory Consulting, Quality Assurance, Medical Writing, Investigator TrainingWhat Is The Driver Of The Clinical Trial Support Services Market?
The emerging personalized medicine is expected to propel the growth of the clinical trial support services market going forward. Personalized medicine involve tailoring treatments to individual patients based on their genetic, environmental, and lifestyle factors to optimize outcomes and improve care. The use of personalized medicine is increasing due to factors such as advancements in genomics, growing availability of biomarker-based diagnostics, improved data analytics technologies and a rising demand for more targeted and effective therapies. Clinical trial support services facilitate personalized medicine by optimizing patient recruitment, implementing biomarker-driven trial designs, and ensuring regulatory compliance, enabling the development of targeted therapies tailored to individual genetic and disease profiles. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based nonprofit organization, the U.S. Food and Drug Administration (FDA) approved 26 new personalized treatments in 2023, a significant increase from the 12 approved in 2022. Therefore, the emerging personalized medicine is driving the growth of the clinical trial support services industry.Key Players In The Global Clinical Trial Support Services Market
Major companies operating in the clinical trial support services market are Thermo Fisher Scientific Inc., IQVIA Inc., Laboratory Corporation of America Holdings (Labcorp), ICON plc, SGS S.A., Eurofins Scientific SE, WuXi AppTec, Syneos Health, Catalent Inc., Charles River Laboratories International Inc., Medpace Holdings Inc., Pharmaron, MARKEN Ltd., Novotech, ClinChoice Private Limited, Medidata Solutions Inc., Premier Research, Quotient Sciences Ltd., Caidya, MMS Holdings Inc., Celerion Inc., Parexel International (MA) Corporation, Alcura Health, Almac GroupGlobal Clinical Trial Support Services Market Trends and Insights
Major companies operating in the clinical trial support services market are focusing on developing patient-centric trials to enhance recruitment, improve retention, minimize participant burden, and ensure a more inclusive and positive experience for diverse patient populations. Patient-centric trials are clinical studies designed with a focus on the needs, preferences, and experiences of patients, aiming to improve accessibility, engagement, and overall participant satisfaction throughout the trial process. For instance, in March 2023, Leal Health, a US-based patient-first digital health company, launched first-of-its-kind patient-centric program-level analytics platform designed to be integrated with pharmaceutical companies' online clinical trial websites that enables to develop and execute effective, patient-focused programs to ensure high-quality patient candidates, reduce recruitment timelines and achieve operational efficiencies for a better end-to-end patient experience. The platform focuses on developing effective, patient-focused programs that ensure high-quality patient candidates for clinical trials.What Are Latest Mergers And Acquisitions In The Clinical Trial Support Services Market?
In February 2024, ICON plc, an Ireland-based clinical research organization, acquired Clinical Research Management Inc. for an undisclosed amount. The acquisition will enhance ICON’s ability to access the market for government sponsored research and further enhances ICON’s capabilities in the areas of vaccines and infectious disease. Clinical Research Management Inc. is a US-based contract research organization (CRO) that offers clinical research and trial services.Regional Outlook
North America was the largest region in the clinical trial support services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Clinical Trial Support Services Market?
The clinical trial support services market includes revenues earned by entities by providing services such as regulatory affairs and compliance, medical writing and documentation, pharmacovigilance and safety monitoring, biostatistics and data analysis, quality assurance and auditing, and eClinical solutions. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Clinical Trial Support Services Market Report 2026?
The clinical trial support services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial support services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Clinical Trial Support Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $26.49 billion |
| Revenue Forecast In 2035 | $35.97 billion |
| Growth Rate | CAGR of 8.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service, Phases, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., IQVIA Inc., Laboratory Corporation of America Holdings (Labcorp), ICON plc, SGS S.A., Eurofins Scientific SE, WuXi AppTec, Syneos Health, Catalent Inc., Charles River Laboratories International Inc., Medpace Holdings Inc., Pharmaron, MARKEN Ltd., Novotech, ClinChoice Private Limited, Medidata Solutions Inc., Premier Research, Quotient Sciences Ltd., Caidya, MMS Holdings Inc., Celerion Inc., Parexel International (MA) Corporation, Alcura Health, Almac Group |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Clinical Trial Support Services market was valued at $24.48 billion in 2025, increased to $26.49 billion in 2026, and is projected to reach $35.97 billion by 2030.
The global Clinical Trial Support Services market is expected to grow at a CAGR of 7.9% from 2026 to 2035 to reach $35.97 billion by 2035.
Some Key Players in the Clinical Trial Support Services market Include, Thermo Fisher Scientific Inc., IQVIA Inc., Laboratory Corporation of America Holdings (Labcorp), ICON plc, SGS S.A., Eurofins Scientific SE, WuXi AppTec, Syneos Health, Catalent Inc., Charles River Laboratories International Inc., Medpace Holdings Inc., Pharmaron, MARKEN Ltd., Novotech, ClinChoice Private Limited, Medidata Solutions Inc., Premier Research, Quotient Sciences Ltd., Caidya, MMS Holdings Inc., Celerion Inc., Parexel International (MA) Corporation, Alcura Health, Almac Group .
Major trend in this market includes: Patient-Centric Trials Revolutionize Clinical Research. For further insights on this market.
Request for SampleNorth America was the largest region in the clinical trial support services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial support services market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
