
Closed Viral Vector Manufacturing Platforms Market Report 2026
Global Outlook – By Product Type (Plasmid Deoxyribonucleic Acid (DNA), Lentiviral Vectors, Adenoviral Vectors, Adeno-Associated Viral Vectors, Retroviral Vectors, Other Product Types), By Workflow (Upstream Processing, Downstream Processing, Fill-Finish), By Application (Gene Therapy, Vaccines, Cell Therapy, Other Applications), By End User (Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Manufacturing Organizations, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Closed Viral Vector Manufacturing Platforms Market Overview
• Closed Viral Vector Manufacturing Platforms market size has reached to $1.96 billion in 2025 • Expected to grow to $3.63 billion in 2030 at a compound annual growth rate (CAGR) of 13.1% • Growth Driver: Rise In Clinical Trials Driving The Growth Of The Market Due To Increasing Demand For Innovative Therapies • Market Trend: Advancements In Nitrogen Regulation Technology Enhancing Viral Vector And Vaccine Production • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Closed Viral Vector Manufacturing Platforms Market?
Closed viral vector manufacturing platforms are fully enclosed systems designed to produce viral vectors under tightly controlled and sterile conditions. They integrate various production steps, including cell growth, vector assembly, purification, and formulation, within a sealed environment to minimize contamination and human intervention. These platforms improve process consistency, scalability, and regulatory compliance, ensuring high-quality vector output while maintaining safety and efficiency throughout the production cycle. The main product types of closed viral vector manufacturing platforms include plasmid deoxyribonucleic acid (DNA), lentiviral vectors, adenoviral vectors, adeno-associated viral vectors, retroviral vectors, and others. Plasmid DNA consists of circular, double-stranded DNA molecules that serve as essential templates for producing viral vectors or as direct gene delivery tools in non-viral gene therapy. Different workflows include upstream processing, downstream processing, and fill-finish and are applied for gene therapy, vaccines, cell therapy, and others. It is used by several end users, including pharmaceutical and biotechnology companies, academic and research institutes, contract manufacturing organizations, and others.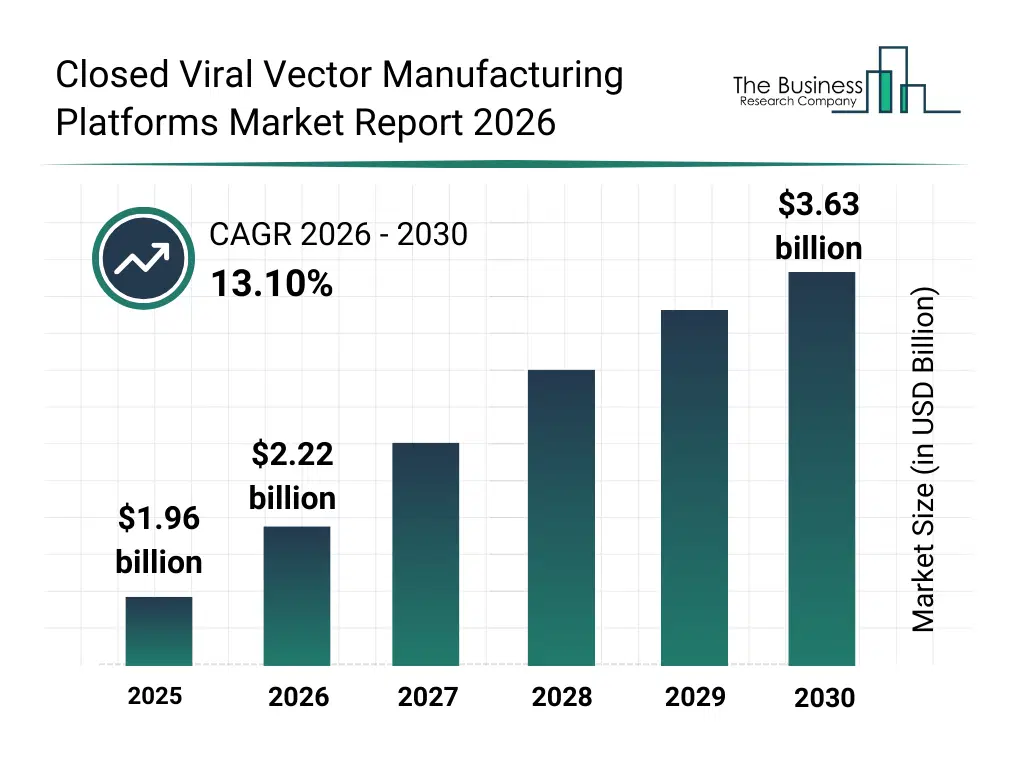
What Is The Closed Viral Vector Manufacturing Platforms Market Size and Share 2026?
The closed viral vector manufacturing platforms market size has grown rapidly in recent years. It will grow from $1.96 billion in 2025 to $2.22 billion in 2026 at a compound annual growth rate (CAGR) of 13.4%. The growth in the historic period can be attributed to rising gene therapy clinical trials, increased use of viral vectors in vaccine development, expansion of cell and gene therapy research programs, growing regulatory focus on sterile manufacturing, early adoption of closed processing technologies.What Is The Closed Viral Vector Manufacturing Platforms Market Growth Forecast?
The closed viral vector manufacturing platforms market size is expected to see rapid growth in the next few years. It will grow to $3.63 billion in 2030 at a compound annual growth rate (CAGR) of 13.1%. The growth in the forecast period can be attributed to increasing commercialization of gene therapies, rising demand for large-scale viral vector supply, growing investments in advanced biomanufacturing infrastructure, expansion of contract manufacturing capacity, increasing focus on process standardization and automation. Major trends in the forecast period include increasing adoption of fully closed and automated production systems, rising demand for scalable viral vector manufacturing platforms, growing integration of single-use and modular components, expansion of end-to-end closed processing workflows, enhanced focus on regulatory compliance and contamination control.Global Closed Viral Vector Manufacturing Platforms Market Segmentation
1) By Product Type: Plasmid Deoxyribonucleic Acid (DNA), Lentiviral Vectors, Adenoviral Vectors, Adeno-Associated Viral Vectors, Retroviral Vectors, Other Product Types 2) By Workflow: Upstream Processing, Downstream Processing, Fill-Finish 3) By Application: Gene Therapy, Vaccines, Cell Therapy, Other Applications 4) By End User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Manufacturing Organizations, Other End Users Subsegments: 1) By Plasmid Deoxyribonucleic Acid (DNA): Supercoiled DNA, Linearized DNA, Minicircle DNA 2) By Lentiviral Vectors: Integrating Lentiviral Vectors, Non-Integrating Lentiviral Vectors 3) By Adenoviral Vectors: Replication-Deficient Adenoviral Vectors, Replication-Competent Adenoviral Vectors 4) By Adeno-Associated Viral Vectors (AAV): Single-Stranded AAV (ssAAV), Self-Complementary AAV (scAAV) 5) By Retroviral Vectors: Gamma-Retroviral Vectors, Alpha-Retroviral Vectors 6) By Other Product Types: Herpes Simplex Virus (HSV) Vectors, Vaccinia Virus Vectors, Baculovirus VectorsWhat Is The Driver Of The Closed Viral Vector Manufacturing Platforms Market?
The growing number of clinical trials is expected to propel the growth of the closed viral vector manufacturing platforms market going forward. Clinical trials are carefully designed studies carried out in people to assess the safety, effectiveness, and appropriate use of new medical therapies or interventions. The demand for clinical trials is rising due to the growing need for innovative therapies, as increasing prevalence of chronic and complex diseases drives the development and testing of new drugs and treatments to address unmet medical needs. Closed viral vector manufacturing platforms support clinical trials by providing consistent, sterile production of viral vectors, ensuring the safety and reliability of the therapies under investigation. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry (ABPI), a UK-based trade association, the total number of industry-sponsored clinical trials launched in the UK rose from 411 in 2022 to 426 in 2023. Therefore, the growing number of clinical trials is driving the growth of the closed viral vector manufacturing platforms industry.Key Players In The Global Closed Viral Vector Manufacturing Platforms Market
Major companies operating in the closed viral vector manufacturing platforms market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec, Samsung Biologics Co. Ltd., FUJIFILM Biotechnologies, Oxford Biomedica Plc, Hillgene, Takara Bio Inc., SkyPharma Production SAS, GeneScript ProBio, Obio Technology Corp. Ltd., VectorBuilder, Charles River Laboratories Pvt. Ltd., Creative Biogene, Esco Aster Pte. Ltd., Karma Biotechnologies, Cell and Gene Therapy Catapult, CoJourney, Applied Biological Laboratories Inc., CEVEC Pharmaceuticals, Genesail Biotech Co. Ltd.Global Closed Viral Vector Manufacturing Platforms Market Trends and Insights
Major companies operating in the closed viral vector manufacturing platforms market are focusing on developing advanced solutions, such as nitro controllers, to prevent nitrogen-induced stress or toxicity in cell cultures. Nitro controllers are devices or systems used to monitor and regulate nitrogen levels or reactive nitrogen species in bioprocesses, ensuring optimal conditions for cell growth and product formation. For instance, in May 2025, Univercells Technologies, a Belgium-based biotechnology company, launched the scale-X nitro controller. It is a compact and economical system engineered for large-scale viral vector and vaccine production. It is compatible with 200 m² and 600 m² scale-X nitro bioreactors and supports both adherent and suspension cell cultures. The system enables efficient production of AAV, lentiviral vectors, exosomes, and oncolytic viruses. Additionally, it allows rapid scale-up, facilitating the transition from research and development to commercial manufacturing within eight months.What Are Latest Mergers And Acquisitions In The Closed Viral Vector Manufacturing Platforms Market?
In November 2023, Ajinomoto Co. Inc., a Japan-based biotechnology company, acquired Forge Biologics for $545 million. Following this acquisition, Ajinomoto obtained Forge’s 200,000-square-foot cGMP facility, the Hearth, in Columbus, Ohio, along with a workforce of over 300 employees. This expansion strengthens Ajinomoto’s global gene therapy manufacturing capabilities, enhancing its contract development and manufacturing services for AAV and plasmid-based therapies. Forge Biologics is a US-based company specializing in closed viral vector manufacturing platforms.Regional Insights
North America was the largest region in the Closed Viral Vector Manufacturing Platforms market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Closed Viral Vector Manufacturing Platforms Market?
The closed viral vector manufacturing platforms market includes revenues earned by entities by providing services such as upstream process development, downstream process development, analytical development, and formulation development. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Closed Viral Vector Manufacturing Platforms Market Report 2026?
The closed viral vector manufacturing platforms market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the closed viral vector manufacturing platforms industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Closed Viral Vector Manufacturing Platforms Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.22 billion |
| Revenue Forecast In 2035 | $3.63 billion |
| Growth Rate | CAGR of 13.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Workflow, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec, Samsung Biologics Co. Ltd., FUJIFILM Biotechnologies, Oxford Biomedica Plc, Hillgene, Takara Bio Inc., SkyPharma Production SAS, GeneScript ProBio, Obio Technology Corp. Ltd., VectorBuilder, Charles River Laboratories Pvt. Ltd., Creative Biogene, Esco Aster Pte. Ltd., Karma Biotechnologies, Cell and Gene Therapy Catapult, CoJourney, Applied Biological Laboratories Inc., CEVEC Pharmaceuticals, Genesail Biotech Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
