
Clot Management Devices Market Report 2026
Global Outlook – By Product Type (Neurovascular Embolectomy Devices, Embolectomy Balloon Catheters, Percutaneous Thrombectomy Devices, Catheter-Directed Thrombolysis (CDT) devices, Inferior vena cava filters (IVCF)), By Percutaneous Thrombectomy Devices Type Outlook (Aspiration Thrombectomy Devices, Percutaneous Mechanical Thrombectomy Device), By End User (Diagnostic centers, Hospitals) – Market Size, Trends, Strategies, and Forecast to 2035
Clot Management Devices Market Overview
• Clot Management Devices market size has reached to $1.82 billion in 2025 • Expected to grow to $2.37 billion in 2030 at a compound annual growth rate (CAGR) of 5.3% • Growth Driver: Rising Hospital Demand Fuels Growth In The Clot Management Device Market • Market Trend: Unveils AI-Enhanced Lightning Flash 2.0 For Advanced Clot Management • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clot Management Devices Market?
A clot management device is a medical tool designed to prevent, manage, or remove blood clots within the vascular system. They are used for the treatment of blood clots, particularly in the context of vascular conditions such as deep vein thrombosis (DVT) or pulmonary embolism (PE). The main product types of the clot management devices market are neurovascular embolectomy devices, embolectomy balloon catheters, percutaneous thrombectomy devices, catheter-directed thrombolysis (CDT) devices, and inferior vena cava filters (IVCF). Neurovascular embolectomy devices are specialized tools used to remove clots from the blood vessels in the brain. The various percutaneous thrombectomy device type outlooks include aspiration thrombectomy devices, and percutaneous mechanical thrombectomy devices and are used by various end users such as diagnostic centers, and hospitals.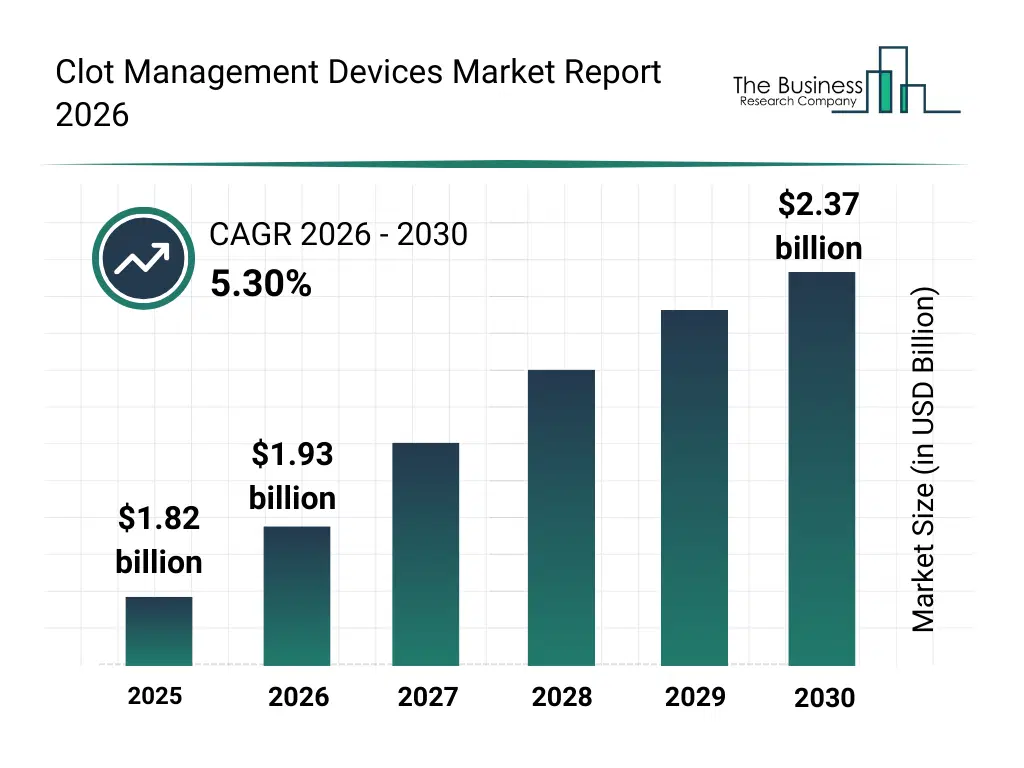
What Is The Clot Management Devices Market Size and Share 2026?
The clot management devices market size has grown strongly in recent years. It will grow from $1.82 billion in 2025 to $1.93 billion in 2026 at a compound annual growth rate (CAGR) of 6.0%. The growth in the historic period can be attributed to rising incidence of deep vein thrombosis and pulmonary embolism, increasing prevalence of cardiovascular diseases, growth in interventional radiology procedures, expanding hospital vascular care capabilities, improved awareness of clot-related complications.What Is The Clot Management Devices Market Growth Forecast?
The clot management devices market size is expected to see strong growth in the next few years. It will grow to $2.37 billion in 2030 at a compound annual growth rate (CAGR) of 5.3%. The growth in the forecast period can be attributed to aging population with higher thrombotic risk, rising demand for minimally invasive vascular interventions, increasing focus on early diagnosis and treatment of clotting disorders, expansion of advanced stroke and thrombosis centers, growing adoption of endovascular treatment approaches. Major trends in the forecast period include rising adoption of minimally invasive clot removal procedures, increasing use of mechanical and aspiration thrombectomy devices, growing focus on rapid intervention for acute thrombotic events, expansion of catheter-based treatments for dvt and pe, increasing preference for retrievable inferior vena cava filters.Global Clot Management Devices Market Segmentation
1) By Product Type: Neurovascular Embolectomy Devices, Embolectomy Balloon Catheters, Percutaneous Thrombectomy Devices, Catheter-Directed Thrombolysis (CDT) devices, Inferior vena cava filters (IVCF) 2) By Percutaneous Thrombectomy Devices Type Outlook: Aspiration Thrombectomy Devices, Percutaneous Mechanical Thrombectomy Device 3) By End User: Diagnostic centers, Hospitals Subsegments: 1) By Neurovascular Embolectomy Devices: Aspiration Embolectomy Devices, Mechanical Embolectomy Devices, Embolectomy Balloon Catheters 2) By Embolectomy Balloon Catheters: High-Pressure Balloon Catheters, Low-Pressure Balloon Catheters 3) By Percutaneous Thrombectomy Devices: Rotational Thrombectomy Devices, Aspiration Thrombectomy Devices 4) By Catheter-Directed Thrombolysis (CDT) Devices: Infusion Catheters, Guidewires For CDT 5) By Inferior Vena Cava Filters (IVCF): Permanent IVC Filters, Retrievable IVC FiltersWhat Is The Driver Of The Clot Management Devices Market?
The increasing demand for hospitals is expected to propel the growth of the clot management device market going forward. Hospitals are healthcare facilities specifically designed to provide medical treatment, care, and services to individuals suffering from various illnesses, injuries, or medical conditions. Hospitals are increasing due to various factors, such as aging populations, urbanization, healthcare access, population expansion, and medical tourism. Clot management devices in the healthcare sector serve as instrumental components for treating conditions such as deep vein thrombosis, pulmonary embolism, and ischemic strokes, providing patients with effective and minimally invasive solutions and enhancing overall treatment outcomes and patient safety. For instance, in May 2024, according to the American Health Care Association, a US-based nonprofit organization, the number of hospitals in the US increased to 6,120 from 5,129 in 2022. Therefore, the increasing demand for hospitals is driving the growth of the clot management device industry.Key Players In The Global Clot Management Devices Market
Major companies operating in the clot management devices market are Bayer AG, Abbott Laboratories, Medtronic plc, Boston Scientific Corporation, B. Braun Melsungen AG, Terumo Corporation, Acandis GmbH & Co KG, Cook Medical, Merit Medical Systems Inc., Penumbra Inc., MicroVention Inc., Inari Medical Inc., AngioDynamics Inc., Argon Medical Devices Inc., LeMaitre Vascular Inc., iVascular SLU, Straub Medical AG, Rapid Medical, C.R. Bard Inc., InspireMD Inc.Global Clot Management Devices Market Trends and Insights
Major companies operating in the clot management device market are developing innovative technologies, such as computer-assisted vacuum thrombectomy (CAVT) systems, to serve customers with advanced features. Computer-assisted vacuum thrombectomy (CAVT) systems are medical devices used in the treatment of blood clots, particularly in conditions such as deep vein thrombosis (DVT) and pulmonary embolism (PE). For instance, in April 2024, Penumbra Inc., a US-based medical company, launched the Lightning Flash 2.0. It is the latest clot aspiration and vacuum thrombectomy (CAVT) technology designed to remove blood clots rapidly. The lightning flash 2.0 represents a significant advancement in clot management devices, offering enhanced performance and efficiency. This state-of-the-art device integrates cutting-edge AI and sophisticated engineering to improve patient outcomes in acute and chronic care settings. It includes advanced imaging and suction capabilities, allowing for precise and effective clot removal to meet the demands of various medical environments.What Are Latest Mergers And Acquisitions In The Clot Management Devices Market?
In December 2023, Johnson & Johnson (J&J) MedTech a US-based healthcare company, acquired Laminar Ltd. for $400 Million. With this acquisition, Johnson & Johnson MedTech aims to strengthen its presence in the clot management devices market by integrating Laminar’s innovative left atrial appendage (LAA) closure technology, which helps reduce stroke risk in atrial fibrillation patients who cannot take long-term blood thinners, thereby enhancing its cardiovascular product portfolio and advancing stroke prevention solutions. Laminar Inc. is a US-based medical device company that develops clot management devices, and heart implants to reduce the risk of stroke.Regional Outlook
North America was the largest region in the clot management devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Clot Management Devices Market?
The clot management devices market consists of sales of catheter-directed thrombolysis systems, thrombectomy devices, and inferior vena cava (IVC) filters. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Clot Management Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.93 billion |
| Revenue Forecast In 2035 | $2.37 billion |
| Growth Rate | CAGR of 6.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Percutaneous Thrombectomy Devices Type Outlook, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Bayer AG, Abbott Laboratories, Medtronic plc, Boston Scientific Corporation, B. Braun Melsungen AG, Terumo Corporation, Acandis GmbH & Co KG, Cook Medical, Merit Medical Systems Inc., Penumbra Inc., MicroVention Inc., Inari Medical Inc., AngioDynamics Inc., Argon Medical Devices Inc., LeMaitre Vascular Inc., iVascular SLU, Straub Medical AG, Rapid Medical, C.R. Bard Inc., InspireMD Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
