
Collagen Booster Microneedle Device Market Report 2026
Global Outlook – By Product Type (Manual Microneedle Devices, Automated Microneedle Devices), By Distribution Channel (Online Retail, Pharmacies, Specialty Stores, Other Distribution Channels), By Application (Anti-Aging, Scar Treatment, Skin Rejuvenation, Hyperpigmentation, Other Applications), By End-User (Dermatology Clinics, Hospitals, Homecare Settings, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Collagen Booster Microneedle Device Market Overview
• Collagen Booster Microneedle Device market size has reached to $0.52 billion in 2025 • Expected to grow to $0.85 billion in 2030 at a compound annual growth rate (CAGR) of 10.2% • Growth Driver: Surging Demand For Minimally Invasive Cosmetic Procedures Driving The Market Growth Due To Enhancing Consumer Preference For Non-Surgical Treatments • Market Trend: Advanced Retraction Mechanisms Elevate Precision And Safety In Skin Treatments • North America was the largest region in 2025.What Is Covered Under Collagen Booster Microneedle Device Market?
A collagen booster microneedle device is a skincare tool designed to stimulate collagen production in the skin through controlled micro-injuries. It uses fine needles to promote natural healing processes, improving skin elasticity, firmness, and texture. The device enhances the absorption of active ingredients, supporting overall skin rejuvenation and a more youthful appearance. The main product types of collagen booster microneedle devices are manual microneedle devices and automated microneedle devices. A manual microneedle device is a hand-operated tool that precisely delivers tiny needles into the skin for cosmetic or therapeutic purposes without electronic assistance. They are distributed through various channels, including online retail, pharmacies, specialty stores, and other distribution channels. They are used for applications such as anti-aging, scar treatment, skin rejuvenation, hyperpigmentation, and other indications, and are utilized by end-users including dermatology clinics, hospitals, homecare settings, and other healthcare providers.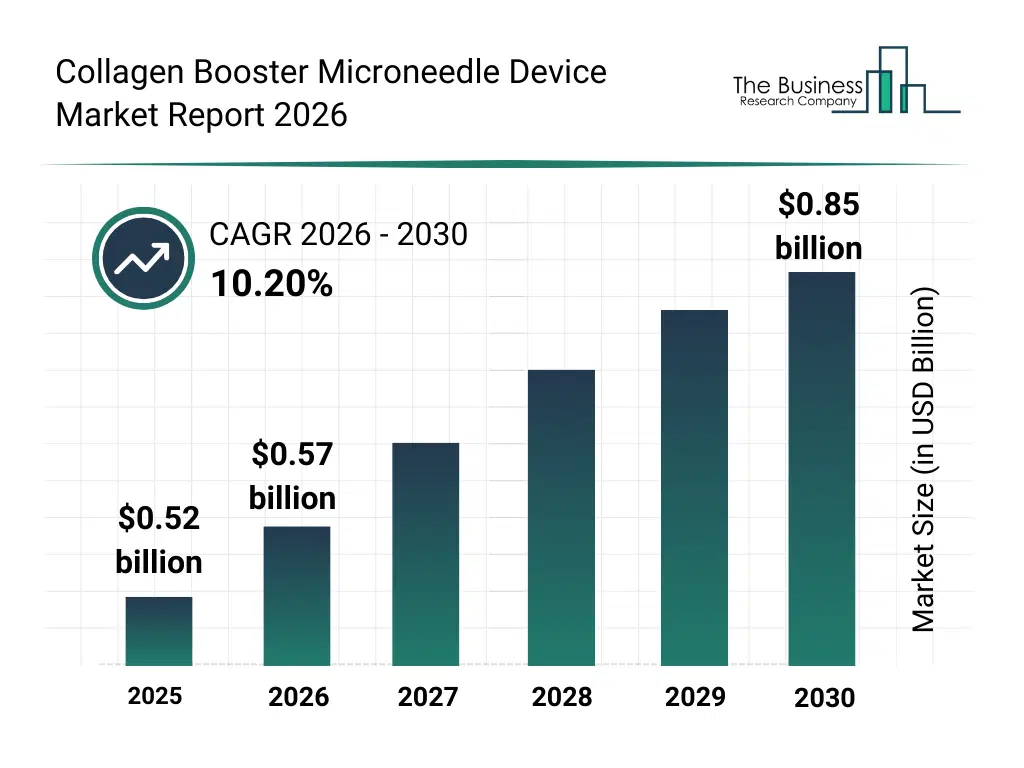
What Is The Collagen Booster Microneedle Device Market Size and Share 2026?
The collagen booster microneedle device market size has grown rapidly in recent years. It will grow from $0.52 billion in 2025 to $0.57 billion in 2026 at a compound annual growth rate (CAGR) of 10.5%. The growth in the historic period can be attributed to growing demand for anti-aging treatments, adoption of microneedling in dermatology, advances in manual microneedle devices, increasing homecare skincare routines, rising awareness of collagen stimulation therapies.What Is The Collagen Booster Microneedle Device Market Growth Forecast?
The collagen booster microneedle device market size is expected to see rapid growth in the next few years. It will grow to $0.85 billion in 2030 at a compound annual growth rate (CAGR) of 10.2%. The growth in the forecast period can be attributed to innovation in automated microneedle devices, integration with cosmetic serums, expansion in dermatology clinics, rise of personalized skincare treatments, adoption of smart microneedle systems. Major trends in the forecast period include motorized microneedle pens, smart microneedle systems, electronic derma rollers, pneumatic microneedle devices, enhanced skin absorption techniques.Global Collagen Booster Microneedle Device Market Segmentation
1) By Product Type: Manual Microneedle Devices, Automated Microneedle Devices 2) By Distribution Channel: Online Retail, Pharmacies, Specialty Stores, Other Distribution Channels 3) By Application: Anti-Aging, Scar Treatment, Skin Rejuvenation, Hyperpigmentation, Other Applications 4) By End-User: Dermatology Clinics, Hospitals, Homecare Settings, Other End Users Subsegments: 1) By Manual Microneedle Devices: Derma Rollers, Stamp Microneedles, Pen Type Microneedles, Adjustable Depth Microneedles 2) By Automated Microneedle Devices: Motorized Microneedle Pens, Electronic Derma Rollers, Pneumatic Microneedle Devices, Smart Microneedle SystemsWhat Is The Driver Of The Collagen Booster Microneedle Device Market?
The surging demand for minimally invasive cosmetic procedures is expected to propel the growth of the collagen booster microneedle device market going forward. Minimally invasive cosmetic procedures are non-surgical treatments that enhance appearance with reduced recovery time and lower risks compared to traditional surgical methods. The growing demand for these procedures is due to consumer preference for safer alternatives with minimal downtime, as modern lifestyles increasingly prioritize quick recovery and natural-looking results without the complications associated with invasive surgery. Collagen booster microneedle devices facilitate minimally invasive cosmetic procedures by creating controlled micro-injuries in the skin that trigger the body's natural wound-healing processes, resulting to increased collagen production and skin rejuvenation without the need for surgical intervention. For instance, in June 2024, according to the American Society of Plastic Surgeons, a US-based non-profit organization, in 2023, there were 347,782 liposuction procedures, marking a 7% increase from 2022. Additionally, plastic surgeries increased by 5% year-over-year, while minimally invasive procedures rose by 7%. Therefore, the surging demand for minimally invasive cosmetic procedures is driving the growth of the collagen booster microneedle device industry.Key Players In The Global Collagen Booster Microneedle Device Market
Major companies operating in the collagen booster microneedle device market are Shiseido Company Limited, Cynosure Inc., Cutera Inc., Sincoheren Co. Ltd., Crown Aesthetics Inc., Amiea Med GmbH, Beijing Sanhe Beauty Science & Technology Co. Ltd., EndyMed Medical Ltd., Eclipse MedCorp LLC, Bomtech Electronics Co. Ltd., DermaQuip Inc., Aesthetics Biomedical Inc., RegenOMedix Inc., Emage Medical Inc., Medicube Co. Ltd., FotroMed Co. Ltd., Accio Biotech Inc., Shenzhen GSD Technology Co. Ltd., Dermapen World, Dr Pen USA, Exceed Medical, Lumenis Ltd, Candela Medical, Mesoestetic Pharma GroupGlobal Collagen Booster Microneedle Device Market Trends and Insights
Major companies operating in the collagen booster microneedle device market are focusing on leveraging innovative technologies such as advanced retraction technology to enhance treatment precision, improve patient safety, and deliver more consistent aesthetic outcomes. Advanced retraction technology is a microneedling innovation that automatically and smoothly retracts the needles after each insertion, helping to reduce tissue trauma, enhance patient comfort, and ensure safer, more precise treatments. For instance, in September 2024, Crown Aesthetics Inc., a US-based medical aesthetics company, received U.S. Food and Drug Administration (FDA) clearance for its new SkinPen Precision Elite, featuring ActiSine technology with active retraction, an RFID-enabled cartridge system, and enhanced safety features. The device works by creating controlled micro-injuries in the skin that stimulate the body’s natural wound-healing process, triggering collagen and elastin production to improve the appearance of acne scars, wrinkles, and overall skin texture. This innovation is designed to improve both provider experience and patient outcomes by offering greater precision, reliability, and comfort during microneedling procedures, while also setting a new benchmark in the collagen booster microneedle device market by combining advanced safety mechanisms, ease of use, and clinically proven skin rejuvenation benefits.What Are Latest Mergers And Acquisitions In The Collagen Booster Microneedle Device Market?
In June 2025, Richards Packaging Income Fund, a Canada-based company, acquired DermapenWorld Pty Ltd. for US$40.0 million. With this acquisition, Richards Packaging Income Fund aims to expand its healthcare portfolio by integrating DermapenWorld, a leading provider of microneedling devices and dermatological cosmetics, leveraging its brand and products to strengthen its healthcare offerings alongside Richards’ existing lines. DermapenWorld Pty Ltd. is an Australia-based manufacturing company that specializes in providing collagen booster microneedle devices.Regional Outlook
North America was the largest region in the collagen booster microneedle device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Collagen Booster Microneedle Device Market?
The collagen booster microneedle device market consists of sales of microneedling patches, collagen-boosting serums and ampoules, at-home microneedling devices, and rf microneedling devices. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Collagen Booster Microneedle Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.57 billion |
| Revenue Forecast In 2035 | $0.85 billion |
| Growth Rate | CAGR of 10.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Shiseido Company Limited, Cynosure Inc., Cutera Inc., Sincoheren Co. Ltd., Crown Aesthetics Inc., Amiea Med GmbH, Beijing Sanhe Beauty Science & Technology Co. Ltd., EndyMed Medical Ltd., Eclipse MedCorp LLC, Bomtech Electronics Co. Ltd., DermaQuip Inc., Aesthetics Biomedical Inc., RegenOMedix Inc., Emage Medical Inc., Medicube Co. Ltd., FotroMed Co. Ltd., Accio Biotech Inc., Shenzhen GSD Technology Co. Ltd., Dermapen World, Dr Pen USA, Exceed Medical, Lumenis Ltd, Candela Medical, Mesoestetic Pharma Group |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
