
Colorectal Cancer Molecular Diagnostics Market Report 2026
Global Outlook – By Type (Instruments, Reagents And Kits, Services), By Technology (Polymerase Chain Reaction (PCR), Sequencing, Mass Spectrometry, Transcription Mediated Amplification, Chips And Microarrays, Isothermal Nucleic Acid Amplification Technology (INAAT)), By End Users (Hospitals, Ambulatory Surgical Centers, Diagnostic Laboratories, Homecare Settings) - Market Size, Trends, And Global Forecast 2026-2035
Colorectal Cancer Molecular Diagnostics Market Overview
• Colorectal Cancer Molecular Diagnostics market size has reached to $2.78 billion in 2025 • Expected to grow to $4.15 billion in 2030 at a compound annual growth rate (CAGR) of 8.3% • Growth Driver: The Impact Of Increasing Colorectal Cancer Prevalence On The Colorectal Cancer Molecular Diagnostics Market • Market Trend: Advancements in Non-Invasive Early Detection for Colorectal Cancer • North America was the largest region in 2025 and Asia Pacific is the fastest growing region.What Is Covered Under Colorectal Cancer Molecular Diagnostics Market?
Colorectal cancer, also known as colon cancer or rectal cancer, is a malignant tumor that develops in the colon (the large intestine) or the rectum (the final portion of the digestive tract just before the anus). It is a type of cancer that arises from the inner lining of these parts of the gastrointestinal system. Colorectal cancer molecular diagnostics uses advanced techniques to identify specific genetic mutations, alterations, and biomarkers that play a role in the development, progression, and treatment response of colorectal cancer. The main types of colorectal cancer molecular diagnostics are instruments, reagents, kits, and services. An instrument refers to a specialized tool or device designed to analyze and detect molecular changes, such as genetic mutations, variations in gene expression, or specific biomolecules, within biological samples of colorectal cancer patients. The molecular diagnostic solutions are also based on technologies such as polymerase chain reaction (PCR), sequencing, mass spectrometry, transcription-mediated amplification, chips and microarrays, and isothermal nucleic acid amplification technology (INAAT) used by end users such as hospitals, ambulatory surgical centers, diagnostic laboratories, and homecare settings.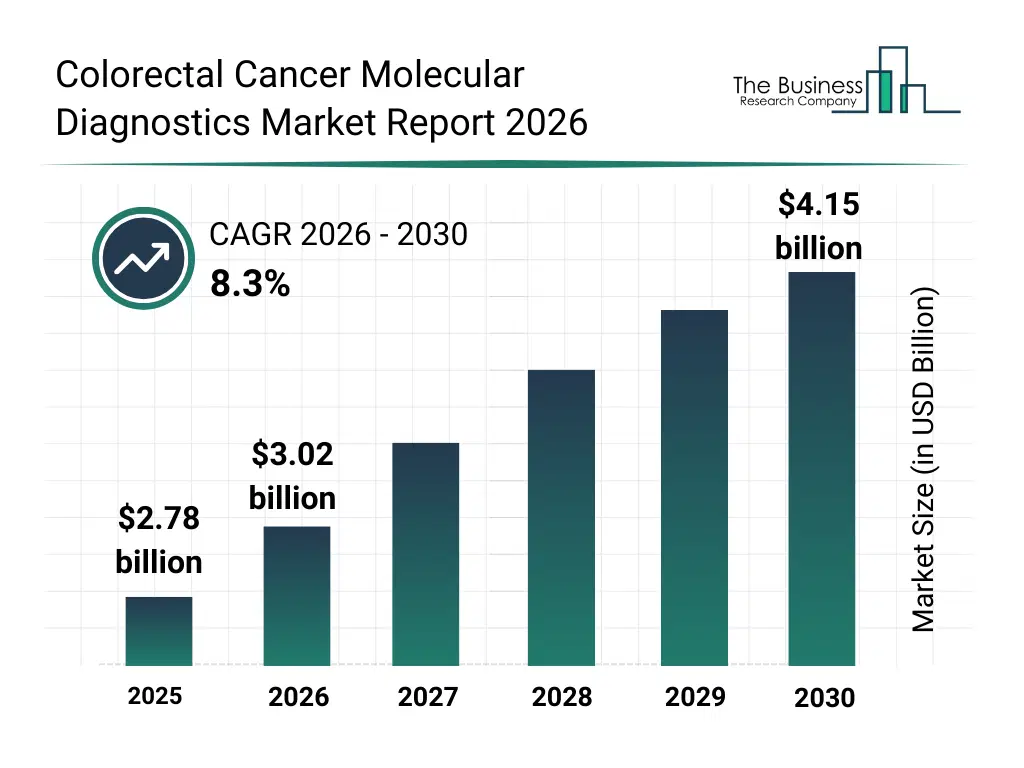
What Is The Colorectal Cancer Molecular Diagnostics Market Size and Share 2026?
The colorectal cancer molecular diagnostics market size has grown strongly in recent years. It will grow from $2.78 billion in 2025 to $3.02 billion in 2026 at a compound annual growth rate (CAGR) of 8.5%. The growth in the historic period can be attributed to increasing colorectal cancer incidence rates, expansion of diagnostic laboratory infrastructure, rising adoption of molecular testing protocols, improved awareness of early screening, growing use of precision medicine approaches.What Is The Colorectal Cancer Molecular Diagnostics Market Growth Forecast?
The colorectal cancer molecular diagnostics market size is expected to see strong growth in the next few years. It will grow to $4.15 billion in 2030 at a compound annual growth rate (CAGR) of 8.3%. The growth in the forecast period can be attributed to increasing investments in precision oncology, rising demand for minimally invasive diagnostics, expansion of genomic testing programs, growing integration of ai in cancer diagnostics, increasing focus on treatment response monitoring. Major trends in the forecast period include increasing adoption of molecular biomarker testing, rising use of pcr and sequencing technologies, growing demand for personalized oncology diagnostics, expansion of companion diagnostic development, enhanced focus on early cancer detection.Global Colorectal Cancer Molecular Diagnostics Market Segmentation
1) By Type: Instruments, Reagents And Kits, Services 2) By Technology: Polymerase Chain Reaction (PCR), Sequencing, Mass Spectrometry, Transcription Mediated Amplification, Chips And Microarrays, Isothermal Nucleic Acid Amplification Technology (INAAT) 3) By End Users: Hospitals, Ambulatory Surgical Centers, Diagnostic Laboratories, Homecare Settings Subsegments: 1) By Instruments: PCR Machines, Sequencers, Microarrays 2) By Reagents And Kits: DNA Or RNA Extraction Kits, Assay Kits, Reagent Packs 3) By Services: Diagnostic Testing Services, Genetic Testing Services, Consultation ServicesWhat Is The Driver Of The Colorectal Cancer Molecular Diagnostics Market?
The rising prevalence of colorectal cancer is expected to propel the growth of the colorectal cancer molecular diagnostics market going forward. Colorectal cancer is a disease that affects the colon or rectum, which are parts of the digestive system. Colorectal cancer occurs when cells in the colon or rectum grow out of control and form a tumor. The increase in colorectal cancer increases the demand for molecular diagnostics for cancer detection and treatment. For instance, in March 2023, according to the American Cancer Society, a US-based nationwide voluntary health organization dedicated to eliminating cancer, roughly 153,020 people are expected to receive a diagnosis of CRC (colorectal cancer), and out of that number, approximately 52,550 individuals are projected to succumb to the disease. Among those affected, there will be 19,550 cases and 3,750 deaths occurring in individuals under the age of 50. Therefore, the rising prevalence of colorectal cancer is driving the growth of the colorectal cancer molecular diagnostics industry.Key Players In The Global Colorectal Cancer Molecular Diagnostics Market
Major companies operating in the colorectal cancer molecular diagnostics market are Johnson & Johnson, Bayer AG, Abbott Laboratories, Danaher Corporation, Becton Dickinson and Company, Biocartis Group NV, Agilent Technologies Inc., Grifols S.A, Hologic Inc., Biomérieux SA, Quidel Corporation, Bio-Rad Laboratories Inc., Qiagen N.V., Exact Sciences Corporation, Illumina Inc., Cepheid, Myriad Genetics Inc., Guardant Health, Luminex Corporation, GenMark Diagnostics, Amoy Diagnostics Co. Ltd., MDxHealth Inc., HTG Molecular Diagnostics Inc., GenPath Diagnostics, geneOmbio Technologies Pvt.Global Colorectal Cancer Molecular Diagnostics Market Trends and Insights
Major companies operating in the colorectal cancer molecular diagnostics market are focusing on developing innovative solutions, such as blood-based liquid biopsy screening tests, to meet the rising demand for non-invasive, early detection tools, improved patient compliance versus colonoscopy, and broader population screening as guidelines and reimbursement evolve. A liquid biopsy is a blood test that detects tumour-derived biomarkers from a simple blood draw; its key features include minimal invasiveness, the ability to screen asymptomatic populations, and molecular-level sensitivity that complements. For instance, in July 2024, Guardant Health, a United States-based precision oncology company, launched Shield, a blood-based colorectal cancer screening test that was approved by the U.S. Food and Drug Administration as a primary screening option for average-risk adults 45 and older. Shield is a multi-marker blood assay that analyses circulating tumour signals to identify people likely to have colorectal cancer; it was developed and validated in the large ECLIPSE clinical programme and demonstrated high sensitivity and specificity sufficient for FDA primary-screening designation. The test’s composition and functionality combine sequencing and proprietary bioinformatic classifiers to detect methylation and other tumour-derived signals in plasma. Its unique features include convenience (single blood draw), potential to increase screening uptake, and a pathway to Medicare coverage, and its applications are population screening and triage to diagnostic colonoscopy for positives, offering earlier detection with fewer invasive procedures.What Are Latest Mergers And Acquisitions In The Colorectal Cancer Molecular Diagnostics Market?
In July 2023, New Day Diagnostics LLC, a US-based medical equipment manufacturing company, acquired the assets of Epigenomics AG for an undisclosed amount. With this acquisition, New Day Diagnostics broadens its offerings within cancer diagnostics, overtaking intellectual property belonging to Epigenomics AG, including Epi ProColon, a screening technology aimed at the non-invasive identification of colorectal cancer. Epigenomics AG is a Germany-based colorectal cancer molecular diagnostics company.Regional Outlook
North America was the largest region in the colorectal cancer molecular diagnostics market in 2025. Asia Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Colorectal Cancer Molecular Diagnostics Market?
The colorectal cancer molecular diagnostics market consists of revenues earned by entities by providing diagnostic genetic mutation analysis, biomarker profiling, and epigenetic analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. The colorectal cancer molecular diagnostics market consists of sales of molecular diagnostic biochips, liquid biopsy kits, and bioinformatics software. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Colorectal Cancer Molecular Diagnostics Market Report 2026?
The colorectal cancer molecular diagnostics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the colorectal cancer molecular diagnostics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Colorectal Cancer Molecular Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.02 billion |
| Revenue Forecast In 2035 | $4.15 billion |
| Growth Rate | CAGR of 8.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Technology, End Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Bayer AG, Abbott Laboratories, Danaher Corporation, Becton Dickinson and Company, Biocartis Group NV, Agilent Technologies Inc., Grifols S.A, Hologic Inc., Biomérieux SA, Quidel Corporation, Bio-Rad Laboratories Inc., Qiagen N.V., Exact Sciences Corporation, Illumina Inc., Cepheid, Myriad Genetics Inc., Guardant Health, Luminex Corporation, GenMark Diagnostics, Amoy Diagnostics Co. Ltd., MDxHealth Inc., HTG Molecular Diagnostics Inc., GenPath Diagnostics, geneOmbio Technologies Pvt. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
