
Companion Diagnostics Market Report 2026
Global Outlook – By Product And Service (Assays, Kits And Reagents, Software And Services), By Technology (Polymerase Chain Reaction, Next-Generation Sequencing, In Situ Hybridization, Immunohistochemistry), By Indication (Lung Cancer, Breast Cancer, Colorectal Cancer, Leukemia, Melanoma), By End-User (Pharmaceutical And Biopharmaceutical Companies, Reference Laboratories, Contract Research Organizations) – Market Size, Trends, Strategies, and Forecast to 2035
Companion Diagnostics Market Overview
• Companion Diagnostics market size has reached to $8.69 billion in 2025 • Expected to grow to $22.7 billion in 2030 at a compound annual growth rate (CAGR) of 21.1% • Growth Driver: The Surge In Targeted Therapies Fueling The Expansion Of Companion Diagnostics • Market Trend: Innovative Companion Diagnostics Transforming Precision Medicine In Non-Small Cell Lung Cancer • North America was the largest region in 2025.What Is Covered Under Companion Diagnostics Market?
Companion diagnostics is an in vitro diagnostic medical test used to check the safety and effectivity of a corresponding drug or biological product. The test aids a medical expert to match a patient to a specific drug or therapy. The main products and services of companion diagnostics are assays, kits and reagents and software and services. Assays, kits and reagents are used in various life science, environmental, and research labs. An assay is a procedure for examining a substance to ascertain its composition or quality. Kits are a set or collection of equipment, materials, or instruction for a particular use. A reagent is a chemical used in laboratory testing to make, measure, or detect other compounds. Polymerase chain reaction, next-generation sequencing, in situ hybridization and immunohistochemistry are the technologies used in companion diagnostics. The companion diagnostics are used for identifying lung, breast, colorectal, leukemia and melanoma cancer diseases. Pharmaceutical and biopharmaceutical companies, reference laboratories and contract research organizations use companion diagnostics.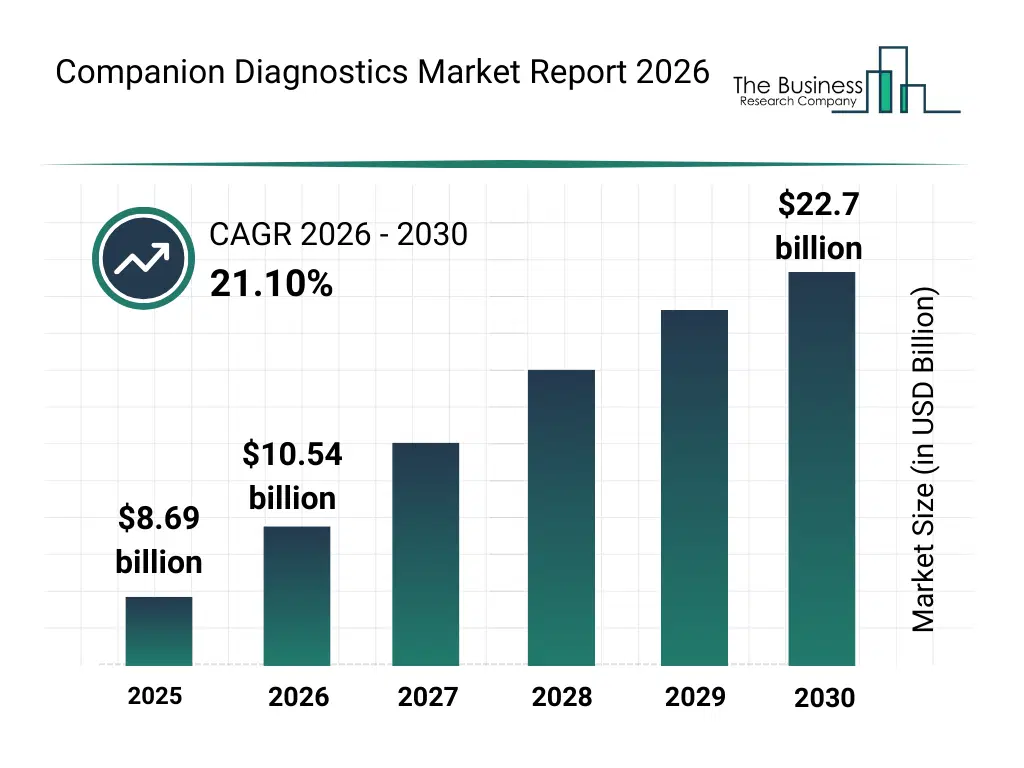
What Is The Companion Diagnostics Market Size and Share 2026?
The companion diagnostics market size has grown exponentially in recent years. It will grow from $8.69 billion in 2025 to $10.54 billion in 2026 at a compound annual growth rate (CAGR) of 21.3%. The growth in the historic period can be attributed to growth in targeted cancer therapies, expansion of oncology clinical trials, increased collaboration between pharma and diagnostics firms, rising cancer incidence rates, regulatory approvals for companion tests.What Is The Companion Diagnostics Market Growth Forecast?
The companion diagnostics market size is expected to see exponential growth in the next few years. It will grow to $22.7 billion in 2030 at a compound annual growth rate (CAGR) of 21.1%. The growth in the forecast period can be attributed to increasing demand for personalized treatment selection, rising investments in precision medicine, expansion of immuno-oncology pipelines, growing adoption of ai-driven diagnostic interpretation, increasing regulatory emphasis on diagnostic-drug co-development. Major trends in the forecast period include increasing adoption of precision oncology diagnostics, rising integration of companion diagnostics in drug development, growing use of ngs-based diagnostic platforms, expansion of biomarker-driven therapies, enhanced focus on personalized medicine.Global Companion Diagnostics Market Segmentation
1) By Product And Service: Assays, Kits And Reagents, Software And Services 2) By Technology: Polymerase Chain Reaction, Next-Generation Sequencing, In Situ Hybridization, Immunohistochemistry 3) By Indication: Lung Cancer, Breast Cancer, Colorectal Cancer, Leukemia, Melanoma 4) By End-User: Pharmaceutical And Biopharmaceutical Companies, Reference Laboratories, Contract Research Organizations Subsegments: 1) By Assays: Polymerase Chain Reaction (PCR) Assays, Immunohistochemistry (IHC) Assays, Next-Generation Sequencing (NGS) Assays, In Situ Hybridization (ISH) Assays 2) By Kits And Reagents: Diagnostic Kits, Detection Reagents, Control Reagents 3) By Software And Services: Data Analysis Software, Interpretation Software, Custom Development Services, Consulting and Support ServicesWhat Is The Driver Of The Companion Diagnostics Market?
The increasing number of targeted therapies is expected to propel the growth of the companion diagnostics market. Targeted therapy is a pharmacological therapy that targets particular characteristics of cancer cells to inhibit the growth and spread of the disease. The medications circulate throughout the body but have a more targeted effect than chemotherapy and frequently have fewer adverse effects. Biological marker-based companion diagnostic testing for targeted cancer therapy is becoming an essential part of personalized cancer care. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based hospital and health care organization, in 2023, the FDA approved 16 new personalized treatments for rare disease patients, up from 6 in 2022. Therefore, the surge in targeted therapies drives growth in the companion diagnostics industry.Key Players In The Global Companion Diagnostics Market
Major companies operating in the companion diagnostics market are F Hoffmann-La Roche Ltd., Agilent Technologies Inc., Qiagen NV, Thermo Fisher Scientific Inc., Abbott Laboratories Inc., Amoy Diagnostics Co. Ltd., Danaher Corporation, Guardant Health Inc., Illumina Inc., Myriad Genetics Inc., Siemens Healthcare GmbH, Almac Group, Caris Life Sciences, Cepheid Inc., Diaceutics plc, Epic Sciences Inc., Foundation Medicine Inc., Genomic Health Inc., Grail Inc., Hologic Inc., HTG Molecular Diagnostics Inc., Inivata Ltd., Invivoscribe Inc., MolecularMD Corporation, Natera Inc., NeoGenomics Laboratories Inc., Personal Genome Diagnostics Inc., Precision Therapeutics Inc., Prometheus Laboratories Inc., Sysmex Corporation, Ventana Medical SystemsGlobal Companion Diagnostics Market Trends and Insights
Major companies operating in the companion diagnostics market are collaborating to develop innovative treatment solutions to enhance personalized medicine, improve patient outcomes, and optimize the effectiveness of targeted therapies. Collaboration of the companies will accelerate the development and commercialization of companion diagnostics (CDx) for hematology and oncology treatments. For instance, in January 2024, Agilent Technologies, Inc., a US-based company that provides instruments, software, services, and consumables for laboratories, announced an agreement with Incyte, a US-based pharmaceutical company. This will bring together Agilent’s expertise and proven track record in the development of companion diagnostics (CDx) to support the development and commercialization of Incyte’s hematology and oncology portfolio. This collaboration allows Agilent to expand its CDx portfolio with new biomarkers, while Incyte benefits from Agilent's expertise in assay development, regulatory approvals, and commercialization to support clinical trials and CDx registration in the U.S. and Europe.What Are Latest Mergers And Acquisitions In The Companion Diagnostics Market?
In September 2023, Exact Sciences, a US-based provider of molecular diagnostics and precision oncology tests, acquired Resolution Bioscience for an undisclosed amount. Through this acquisition, Exact Sciences aims to enhance its companion diagnostic capabilities by integrating Resolution Bioscience’s ctDx FIRST liquid biopsy platform and expanding its portfolio of precision oncology assays. Resolution Bioscience is a US-based company that provides next-generation sequencing (NGS)-based liquid biopsy tests to detect actionable tumor mutations and guide targeted therapy.Regional Outlook
North America was the largest region in the companion diagnostics market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Companion Diagnostics Market?
The companion diagnostics market includes revenues earned by theranostics and monitoring tests. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Companion Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $10.54 billion |
| Revenue Forecast In 2035 | $22.7 billion |
| Growth Rate | CAGR of 21.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product And Service, Technology, Indication, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F Hoffmann-La Roche Ltd., Agilent Technologies Inc., Qiagen NV, Thermo Fisher Scientific Inc., Abbott Laboratories Inc., Amoy Diagnostics Co. Ltd., Danaher Corporation, Guardant Health Inc., Illumina Inc., Myriad Genetics Inc., Siemens Healthcare GmbH, Almac Group, Caris Life Sciences, Cepheid Inc., Diaceutics plc, Epic Sciences Inc., Foundation Medicine Inc., Genomic Health Inc., Grail Inc., Hologic Inc., HTG Molecular Diagnostics Inc., Inivata Ltd., Invivoscribe Inc., MolecularMD Corporation, Natera Inc., NeoGenomics Laboratories Inc., Personal Genome Diagnostics Inc., Precision Therapeutics Inc., Prometheus Laboratories Inc., Sysmex Corporation, Ventana Medical Systems |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
