
Complete Blood Count Device Market Report 2026
Global Outlook – By Device Type (Automated Blood Count Analyzers, Manual Blood Count Analyzers, Point-Of-Care Testing Devices, Hematology Analyzers), By Test Type (White Blood Cell Count (WBC Or Leukocyte Count), White Blood Cell (WBC) Differential Count, Red Blood Cell Count (RBC Or Erythrocyte Count), Hematocrit (Hct), Hemoglobin (Hbg), Platelet Count, Mean Platelet Volume (MPV), Mean Corpuscular Hemoglobin (MCH), Other Test Types), By Technology (Impedance Technology, Laser-Based Technology, Flow Cytometry, Microscopy), By Distribution Channel (Direct Sales, Online Sales, Third-Party Distributors, Laboratory Equipment Suppliers), By End-User (Hospital, Diagnostic Laboratories, Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Complete Blood Count Device Market Overview
• Complete Blood Count Device market size has reached to $4.33 billion in 2025 • Expected to grow to $5.76 billion in 2030 at a compound annual growth rate (CAGR) of 5.9% • Growth Driver: The Growing Prevalence Of Blood Disorders Is Fueling The Growth Of The Market Due To Its Impact On Diagnostic Demand • Market Trend: Advancements In Compact Hematology Analyzers For Point-Of-Care Testing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Complete Blood Count Device Market?
A complete blood count (CBC) device is a medical instrument used to perform a complete blood count, a standard laboratory test that measures the levels and characteristics of various blood components. It automates the blood analysis process, delivering precise and rapid results, which assist healthcare professionals in making well-informed decisions for diagnosis and treatment. The main types of complete blood count devices are automated blood count analyzers, manual blood count analyzers, point-of-care testing devices, and hematology analyzers. Automated blood count analyzers are advanced instruments that use technologies to rapidly and accurately count and classify blood cells, enhancing throughput and reducing manual errors in clinical laboratories. It is utilized for various test types such as white blood cell count (WBC or leukocyte count), white blood cell (WBC) differential count, red blood cell count (RBC or erythrocyte count), hematocrit (HCT), hemoglobin (HBG), platelet count, mean platelet volume (MPV), mean corpuscular hemoglobin (MCH), and others with several technologies, including impedance technology, laser-based technology, flow cytometry, and microscopy. It is distributed through various distribution channels, such as direct sales, online sales, third-party distributors, and retail pharmacies, and is used by several end-users, including hospitals, diagnostic laboratories, research institutes, and others.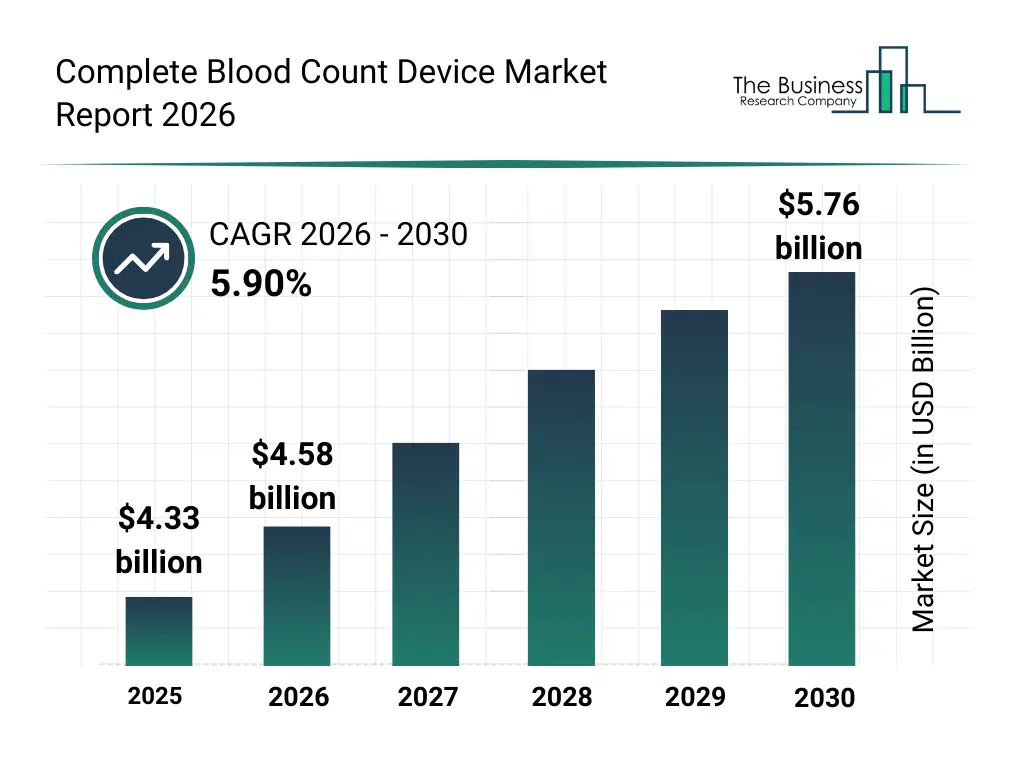
What Is The Complete Blood Count Device Market Size and Share 2026?
The complete blood count device market size has grown strongly in recent years. It will grow from $4.33 billion in 2025 to $4.58 billion in 2026 at a compound annual growth rate (CAGR) of 6.0%. The growth in the historic period can be attributed to expansion of clinical diagnostic testing volumes, increased hospital laboratory automation, growing burden of chronic diseases, availability of standardized blood testing protocols, rising demand for routine health screenings.What Is The Complete Blood Count Device Market Growth Forecast?
The complete blood count device market size is expected to see strong growth in the next few years. It will grow to $5.76 billion in 2030 at a compound annual growth rate (CAGR) of 5.9%. The growth in the forecast period can be attributed to increasing adoption of decentralized diagnostics, rising investments in smart laboratory infrastructure, growing demand for rapid diagnostic results, expansion of preventive healthcare testing, continuous advancements in hematology technology. Major trends in the forecast period include increasing adoption of automated hematology analyzers, rising demand for point-of-care cbc devices, growing integration of ai-based diagnostics, expansion of high-throughput laboratory systems, enhanced focus on diagnostic accuracy and speed.Global Complete Blood Count Device Market Segmentation
1) By Device Type: Automated Blood Count Analyzers, Manual Blood Count Analyzers, Point-Of-Care Testing Devices, Hematology Analyzers 2) By Test Type: White Blood Cell Count (WBC Or Leukocyte Count), White Blood Cell (WBC) Differential Count, Red Blood Cell Count (RBC Or Erythrocyte Count), Hematocrit (Hct), Hemoglobin (Hbg), Platelet Count, Mean Platelet Volume (MPV), Mean Corpuscular Hemoglobin (MCH), Other Test Types 3) By Technology: Impedance Technology, Laser-Based Technology, Flow Cytometry, Microscopy 4) By Distribution Channel: Direct Sales, Online Sales, Third-Party Distributors, Laboratory Equipment Suppliers 5) By End-User: Hospital, Diagnostic Laboratories, Research Institutes, Other End-Users Subsegments: 1) By Automated Blood Count Analyzers: 3-Part Differential Analyzers, 5-Part Differential Analyzers, 6-Part And Above Differential Analyzers, Integrated Hematology Systems, High-Throughput Lab Analyzers 2) By Manual Blood Count Analyzers: Hemocytometers, Microscopes For Manual Differential, Manual Cell Counters, Staining Kits And Reagents 3) By Point-of-Care Testing Devices: Portable Hematology Analyzers, Handheld Complete Blood Count (CBC) Devices, Fingerstick Blood Test Devices, Rapid Diagnostic Complete Blood Count (CBC) Kits 4) By Hematology Analyzers: Low-Volume Hematology Analyzers, Mid-Volume Hematology Analyzers, High-Volume Hematology Analyzers, Veterinary Hematology Analyzers, Reagent-Free AnalyzersWhat Is The Driver Of The Complete Blood Count Device Market?
The growing prevalence of blood disorders is expected to propel the growth of the complete blood count device market going forward. Blood disorders are conditions that disrupt the production, function, or structure of blood cells, resulting in impaired blood function. The rise in blood disorders is linked to the growing occurrence of lifestyle diseases, where poor diet, inactivity, and stress lead to conditions such as anemia and clotting issues. A complete blood count (CBC) device aids in diagnosing blood disorders by analyzing key blood components and detecting abnormalities, allowing healthcare professionals to identify clotting disorders and ensuring prompt and precise treatment. For instance, in May 2024, according to the Canadian Cancer Society, a Canada-based organization, 4,100 individuals were diagnosed with multiple myeloma (a type of blood cancer), while 1,750 succumbed to the disease. Therefore, the growing prevalence of blood disorders is driving the growth of the complete blood count device industry.Key Players In The Global Complete Blood Count Device Market
Major companies operating in the complete blood count device market are Abbott Laboratories, Siemens Healthineers, Mindray Medical International Limited, Beckman Coulter Inc., Sysmex Corporation, Horiba Ltd., Nihon Kohden Corporation, Agappe Diagnostics Ltd., Trivitron Healthcare Pvt. Ltd., HemoCue AB, EKF Diagnostics Holdings plc, Erba Mannheim, Boule Diagnostics AB, Biosystems S.A., Diatron MI Zrt., Labtron Equipment Ltd., Microlab Instruments Co. Pvt. Ltd., URIT Medical Electronic Co. Ltd., Roche Diagnostics, Ortho Clinical Diagnostics, BioSystems Diagnostics Pvt. Ltd., Drew Scientific (subsidiary of EKF), Analyticon Biotechnologies GmbH, Rayto Life and Analytical Sciences Co. Ltd.Global Complete Blood Count Device Market Trends and Insights
Major companies operating in the complete blood count device market are focusing on developing advanced products, such as compact hematology analyzers, to provide more accurate, faster, and cost-effective solutions for point-of-care testing. Compact hematology analyzers are portable, user-friendly devices that provide quick, accurate, complete blood count (CBC) results with minimal sample volume, making them ideal for point-of-care use, enhancing diagnostic accuracy, efficiency, and ease of use while being cost-effective and easy to maintain. For instance, in June 2024, HORIBA, a Japan-based analytical and measurement technology company, launched the Yumizen H550E and H500E hematology analyzers. These compact devices integrate erythrocyte sedimentation rate (ESR) testing with complete blood count (CBC) and 5-part differential (DIFF) analysis, utilizing HORIBA's patented CoRA (correlated rouleaux analysis) technology to provide comprehensive results from whole blood samples in just 60 seconds.What Are Latest Mergers And Acquisitions In The Complete Blood Count Device Market?
In February 2024, Sysmex Corporation, a Japan-based manufacturer of hematology analyzers, partnered with CellaVision AB to advance hematology through innovation in digital cell morphology. The partnership aims to enhance efficiency, precision, and standardization in hematology testing by combining Sysmex’s hematology expertise with CellaVision’s AI-driven imaging technologies, enabling more accurate diagnoses and improved clinical outcomes. CellaVision AB is a Sweden-based provider of digital hematology microscopy solutions.Regional Outlook
North America was the largest region in the complete blood count device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Complete Blood Count Device Market?
The complete blood count (CBC) device market consists of sales of automated hematology analyzers, point-of-care CBC devices, reagents and consumables. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Complete Blood Count Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.58 billion |
| Revenue Forecast In 2035 | $5.76 billion |
| Growth Rate | CAGR of 6.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Device Type, Test Type, Technology, Distribution Channel, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Siemens Healthineers, Mindray Medical International Limited, Beckman Coulter Inc., Sysmex Corporation, Horiba Ltd., Nihon Kohden Corporation, Agappe Diagnostics Ltd., Trivitron Healthcare Pvt. Ltd., HemoCue AB, EKF Diagnostics Holdings plc, Erba Mannheim, Boule Diagnostics AB, Biosystems S.A., Diatron MI Zrt., Labtron Equipment Ltd., Microlab Instruments Co. Pvt. Ltd., URIT Medical Electronic Co. Ltd., Roche Diagnostics, Ortho Clinical Diagnostics, BioSystems Diagnostics Pvt. Ltd., Drew Scientific (subsidiary of EKF), Analyticon Biotechnologies GmbH, Rayto Life and Analytical Sciences Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
