
Controlled Release Drug Delivery Market Report 2026
Global Outlook – By Technology (Wurster Technique, Coacervation, Micro Encapsulation, Implants, Transdermal, Targeted Delivery, Other Technologies), By Release Mechanism (Polymer Based Systems, Micro Reservoir Partition Controlled Drug Delivery Systems, Feedback Regulated Drug Delivery Systems, Activation-Modulated Drug Delivery Systems, Chemically Activated,), By Application (Metered Dose Inhalers, Injectable, Transdermal And Ocular Patches, Other Applications), By End User (Hospitals, Clinics, Personal, Research, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Controlled Release Drug Delivery Market Overview
• Controlled Release Drug Delivery market size has reached to $72.34 billion in 2025 • Expected to grow to $133.82 billion in 2030 at a compound annual growth rate (CAGR) of 13.3% • Growth Driver: The Role Of Rising Healthcare Expenditure In Driving The Controlled Release Drug Delivery Market • Market Trend: Hovione And Ripple Therapeutics Inc. Pave The Way For Broadening Ophthalmic Applications Of Epidel Technology • North America was the largest region in 2025.What Is Covered Under Controlled Release Drug Delivery Market?
Controlled release drug delivery refers to a dosage form that administers a medicine or medication in a specified way. This drug delivery method continuously delivers pharmaceuticals or medications in predictable kinetics for the predetermined time. The main types of technology used in controlled release drug delivery are the Wurster technique, coacervation, micro encapsulation, implants, transdermal, targeted delivery, and others. The Wurster technique, often referred to as fluid bed microencapsulation is the encapsulation of discrete particles in a fluidized bed employing differential air flow to produce cyclic movement of material. It involves several release mechanisms such as polymer-based systems, micro-reservoir partition-controlled drug delivery systems, feedback-regulated drug delivery systems, activation-modulated drug delivery systems, and chemically activated, which are used in applications such as metering dose inhalers, injectables, transdermal and ocular patches, and others. Major end-users include hospitals, clinics, personal, research, and others.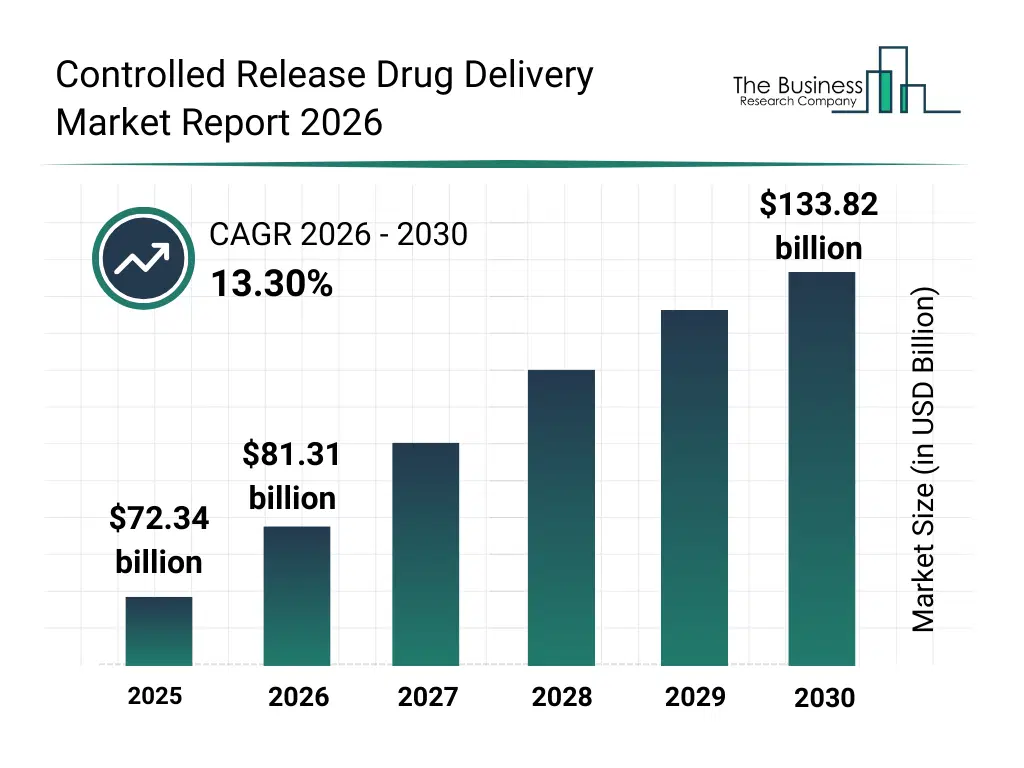
What Is The Controlled Release Drug Delivery Market Size and Share 2026?
The controlled release drug delivery market size has grown rapidly in recent years. It will grow from $72.34 billion in 2025 to $81.31 billion in 2026 at a compound annual growth rate (CAGR) of 12.4%. The growth in the historic period can be attributed to rising prevalence of chronic diseases, increasing r&d in pharmaceutical formulations, demand for sustained-release medicines, advancements in polymer and biomaterial science, growing awareness of drug adherence benefits.What Is The Controlled Release Drug Delivery Market Growth Forecast?
The controlled release drug delivery market size is expected to see rapid growth in the next few years. It will grow to $133.82 billion in 2030 at a compound annual growth rate (CAGR) of 13.3%. The growth in the forecast period can be attributed to growth of personalized medicine, expansion of nanotechnology applications, integration of ai in drug delivery design, increasing geriatric population, regulatory incentives for controlled release therapies. Major trends in the forecast period include personalized drug release systems, nanotechnology-enabled delivery, biodegradable and eco-friendly carriers, advanced oral and injectable formulations, patient-centric compliance solutions.Global Controlled Release Drug Delivery Market Segmentation
1) By Technology: Wurster Technique, Coacervation, Micro Encapsulation, Implants, Transdermal, Targeted Delivery, Other Technologies 2) By Release Mechanism: Polymer Based Systems, Micro Reservoir Partition Controlled Drug Delivery Systems, Feedback Regulated Drug Delivery Systems, Activation-Modulated Drug Delivery Systems, Chemically Activated, 3) By Application: Metered Dose Inhalers, Injectable, Transdermal And Ocular Patches, Other Applications 4) By End User: Hospitals, Clinics, Personal, Research, Other End Users Subsegments: 1) By Wurster Technique: Spray Coating, Fluid Bed Coating 2) By Coacervation: Simple Coacervation, Complex Coacervation 3) By Micro Encapsulation: Polymer-Based Micro Encapsulation, Lipid-Based Micro Encapsulation 4) By Implants: Biodegradable Implants, Non-Biodegradable Implants 5) By Transdermal: Microneedle Patches, Reservoir Systems, Matrix Systems 6) By Targeted Delivery: Nanoparticles, Antibody-Drug Conjugates, Liposomes 7) By Other Technologies: Osmotic Pumps, In Situ Gelling SystemsWhat Is The Driver Of The Controlled Release Drug Delivery Market?
The increasing healthcare spending is expected to propel the growth of the controlled release drug delivery market in the coming years. Healthcare expenditure is the entire number of financial resources, both public and private, devoted to the healthcare sector within a certain geographic region, country, or economy during a specified period. Controlled release healthcare spending on medicinal delivery is a multidimensional investment that encompasses a medicinal product's full lifespan, from early research to post-market surveillance, to improve treatment results, patient adherence, and overall healthcare efficiency. For instance, in May 2024, the Office for National Statistics, a UK-based statistics authority, in 2023, healthcare expenditure in the UK was approximately £292 billion, reflecting a nominal increase of 5.6%. Additionally, long-term health and social care expenditure rose by 2.8% in real terms in 2022. Therefore, increasing healthcare spending is driving the controlled release drug delivery industry.Key Players In The Global Controlled Release Drug Delivery Market
Major companies operating in the controlled release drug delivery market are Johnson & Johnson Services Inc., Merck & Co. Inc., Alkermes plc, Coating Place Inc., Corium International Inc., Depomed Inc., Pfizer Inc., Aradigm Corporation, Capsugel LLC, GlaxoSmithKline plc, Bayer Healthcare LLC, Collegium Pharmaceuticals Inc., GE HealthCare Technologies Inc., Adare Pharmaceuticals Inc., Allergan plc, Novartis AG, Lonza Group AG, Colorcon Inc., SKY Pharmaceuticals Pvt. Ltd., AstraZeneca plc, Biogen Inc., Orbis Biosciences Inc., Alza Corporation, MicroCHIPS Inc., Endo International plc, Heron Therapeutics Inc., Intellipharmaceutics International Inc., Ipsen Biopharmaceuticals Inc., Luye Pharma Group Ltd., Medtronic plc, Nektar Therapeutics, Novo Nordisk A/S, Ocular Therapeutix Inc., Pacira BioSciences Inc., Progenity Inc., Sanofi S.A., Teva Pharmaceutical Industries Ltd., Titan Pharmaceuticals Inc., Vectura Group plc, West Pharmaceutical Services Inc., Zosano Pharma Corporation, Zynerba Pharmaceuticals Inc.Global Controlled Release Drug Delivery Market Trends and Insights
Major companies operating in the chocolate syrup market are adopting a strategic partnership approach, aiming to expand ophthalmic applications. Strategic partnerships refer to a process in which companies leverage each other's strengths and resources to achieve mutual benefits and success. For instance, in March 2023, Hovione, a Portugal-based contract drug manufacturing company, announced a partnership with Ripple Therapeutics Inc. to broaden the usage of Ripple's Epidel technology beyond ophthalmic applications. Collaboration with Hovione is an exciting next step in the expansion of the Epidel platform beyond the ophthalmology area due to the mix of technical synergy, innovative vision, and cultural fit. Hovione's expertise in regulated, sustained drug administration, along with their knowledge of chemical synthesis and pharmaceutical manufacture, makes for an excellent collaboration. Ripple Therapeutics Inc. is a Canada-based pharmaceutical company.What Are Latest Mergers And Acquisitions In The Controlled Release Drug Delivery Market?
In April 2023, DifGen Pharmaceuticals LLC, a U.S.-based specialty pharmaceutical company developing complex generics and advanced dosage forms, acquired Aveva Drug Delivery Services, Inc. for an undisclosed amount. With this acquisition, DifGen strategically enhanced its R&D and manufacturing capability for complex controlled-release formats, particularly transdermal patches and oral dissolvable films. Aveva, a U.S.-based developer and manufacturer, specializes in Transdermal Delivery Systems (TDS) and Oral Dissolvable Films (ODF), including controlled-release formulations.Regional Insights
North America was the largest region in the controlled release drug delivery market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Controlled Release Drug Delivery Market?
The controlled release drug delivery market consists of revenues earned by entities by providing services such as mucoadhesive drug delivery, implantable drug delivery, and gastro-retentive drug delivery. The market value includes the value of related goods sold by the service provider or included within the service offering. The controlled release drug delivery market also includes sales of nebulizers, infusion pumps, and other systems used to provide controlled release drug delivery. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Controlled Release Drug Delivery Market Report 2026?
The controlled release drug delivery market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the controlled release drug delivery industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Controlled Release Drug Delivery Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $81.31 billion |
| Revenue Forecast In 2035 | $133.82 billion |
| Growth Rate | CAGR of 12.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Release Mechanism, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson Services Inc., Merck & Co. Inc., Alkermes plc, Coating Place Inc., Corium International Inc., Depomed Inc., Pfizer Inc., Aradigm Corporation, Capsugel LLC, GlaxoSmithKline plc, Bayer Healthcare LLC, Collegium Pharmaceuticals Inc., GE HealthCare Technologies Inc., Adare Pharmaceuticals Inc., Allergan plc, Novartis AG, Lonza Group AG, Colorcon Inc., SKY Pharmaceuticals Pvt. Ltd., AstraZeneca plc, Biogen Inc., Orbis Biosciences Inc., Alza Corporation, MicroCHIPS Inc., Endo International plc, Heron Therapeutics Inc., Intellipharmaceutics International Inc., Ipsen Biopharmaceuticals Inc., Luye Pharma Group Ltd., Medtronic plc, Nektar Therapeutics, Novo Nordisk A/S, Ocular Therapeutix Inc., Pacira BioSciences Inc., Progenity Inc., Sanofi S.A., Teva Pharmaceutical Industries Ltd., Titan Pharmaceuticals Inc., Vectura Group plc, West Pharmaceutical Services Inc., Zosano Pharma Corporation, Zynerba Pharmaceuticals Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
