
COVID-19 Detection Test Kits and Consumables Market Report 2026
Global Outlook – By Kits (Viral Load Testing Kits (qPCR and RT-PCR), Virus Neutralizing Assay Kits, Antibody Detection Kits (Elisa), Viral Antigen Detection Test Kits, Other Kits), By Consumables (Swabs, Tubes, Viral Transfer Media, Reagents, Other Consumables), By Specimen Type (Nose & Throat Swab, Blood, Sputum, Nasal Aspirate), By End Use (Hospitals, Clinics, Public Health Labs, Private And Commercial Labs, Physicians Labs, Research Institutes, Other End Uses) – Market Size, Trends, Strategies, and Forecast to 2035
COVID-19 Detection Test Kits and Consumables Market Overview
• COVID-19 Detection Test Kits and Consumables market size has reached to $8.35 billion in 2025 • Expected to grow to $13.23 billion in 2030 at a compound annual growth rate (CAGR) of 10.3% • Growth Driver: Government Initiatives Fueling Growth In The COVID-19 Detection Test Kits And Consumables Market • Market Trend: Advancements In At-Home Molecular Diagnostics For COVID-19 And Influenza Detection • North America was the largest region in 2025.What Is Covered Under COVID-19 Detection Test Kits and Consumables Market?
The COVID-19 detection test kits & consumables are used for the identification of novel COVID-19 (SARS-CoV-2). These COVID-19 test kits and consumables use a laboratory procedure called reverse transcription polymerase chain reaction to find the virus' genetic material (RT-PCR). The COVID-19 detection test kits and consumables market covered in this report is segmented by kits into viral load testing kits (qPCR and RT-PCR), virus-neutralizing assay kits, antibody detection kits (ELISA), viral antigen detection test kits, and others. It is also segmented by consumables into swabs, tubes, viral transfer media, reagents, and other consumables and by specimen type into nose and throat swabs, blood, sputum, and nasal aspirate. The various end-uses of COVID-19 detection test kits and consumables are hospitals, clinics, public health labs, private and commercial labs, physicians' labs, research institutes, and others.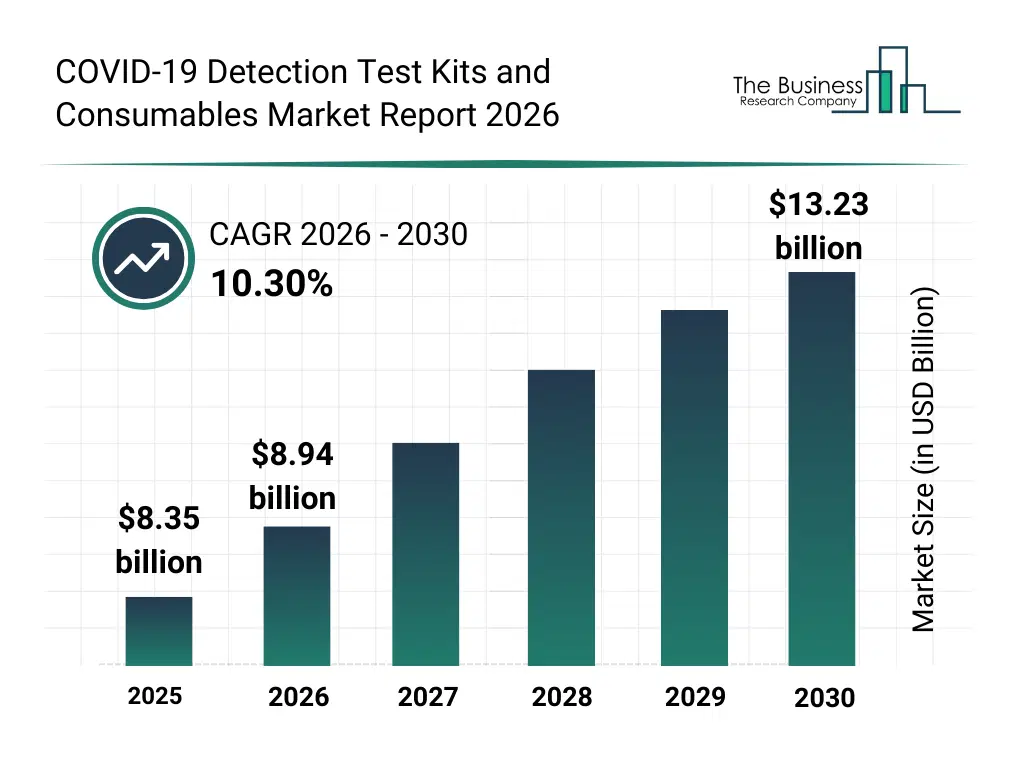
What Is The COVID-19 Detection Test Kits and Consumables Market Size and Share 2026?
The covid-19 detection test kits and consumables market size has grown strongly in recent years. It will grow from $8.35 billion in 2025 to $8.94 billion in 2026 at a compound annual growth rate (CAGR) of 7.1%. The growth in the historic period can be attributed to initial shortage of covid-19 test kits, rising demand in hospitals and public health labs, reliance on conventional rt-pcr testing, limited supply of swabs and reagents, increasing awareness of pandemic monitoring.What Is The COVID-19 Detection Test Kits and Consumables Market Growth Forecast?
The covid-19 detection test kits and consumables market size is expected to see rapid growth in the next few years. It will grow to $13.23 billion in 2030 at a compound annual growth rate (CAGR) of 10.3%. The growth in the forecast period can be attributed to development of rapid and high-throughput testing kits, expansion of private and commercial labs, integration of digital reporting and cloud-based data management, adoption of multiplex and combination test kits, growing investment in research and diagnostic infrastructure. Major trends in the forecast period include rising demand for rapid and accurate covid-19 detection kits, expansion of consumables such as swabs, reagents, and viral transport media, increased adoption of multiplex and combination test kits, growing use of point-of-care and rapid antigen testing, focus on standardization and quality compliance across laboratories.Global COVID-19 Detection Test Kits and Consumables Market Segmentation
1) By Kits: Viral Load Testing Kits (qPCR and RT-PCR), Virus Neutralizing Assay Kits, Antibody Detection Kits (Elisa), Viral Antigen Detection Test Kits, Other Kits 2) By Consumables: Swabs, Tubes, Viral Transfer Media, Reagents, Other Consumables 3) By Specimen Type: Nose & Throat Swab, Blood, Sputum, Nasal Aspirate 4) By End Use: Hospitals, Clinics, Public Health Labs, Private And Commercial Labs, Physicians Labs, Research Institutes, Other End Uses Subsegments: 1) By Viral Load Testing Kits (QPCR And RT-PCR): QPCR Kits, RT-PCR Kits 2) By Virus Neutralizing Assay Kits: Neutralization Test Kits 3) By Antibody Detection Kits (Elisa): IgG Detection Kits, IgM Detection Kits 4) By Viral Antigen Detection Test Kits: Rapid Antigen Test Kits 5) By Other Kits: Combination Test Kits, Multiplex Test KitsWhat Is The Driver Of The COVID-19 Detection Test Kits and Consumables Market?
The increasing government initiatives are expected to propel the growth of the COVID-19 detection test kits and consumables market going forward. Government initiatives refer to actions and programs undertaken by government authorities to address specific issues, achieve objectives, or promote the well-being of the population. Government initiatives are instrumental in creating an enabling environment for the effective deployment of COVID-19 detection test kits and consumables, contributing to the global effort to manage and mitigate the impact of the pandemic. For instance, in 2025, according to the American Medical Association, a US-based association reported that, health expenditures in the U.S. rose by 7.5% in 2023, reaching $4.9 trillion, or about $14,570 per person. Therefore, increasing government initiatives are driving the growth of the COVID-19 detection test kits and consumables industry.Key Players In The Global COVID-19 Detection Test Kits and Consumables Market
Major companies operating in the covid-19 detection test kits and consumables market are Cepheid, BGI Genomics, Abbott Laboratories, bioMérieux SA, Bio-Rad Laboratories, F. Hoffmann-La Roche, GenMark Diagnostics, Mylab Discovery Solutions, Qiagen, Quidel Corporation, Randox Laboratories, SD Biosensor, Seegene Inc., Shenzhen Bioeasy Biotechnology, Thermo Fisher Scientific inc., BioMednomics, Getein Biotech, Sensing Self Ltd., Hangzhou Biotest Biotech Co. Ltd, AmonMed Biotechnology Co., Beijing Tigsun Diagnostics Co Ltd., BioMaxima S.A., CTK Biotech, Hunan Lituo Biotechnology Co., Vivacheck Lab, MD Solutions, FastSense Diagnostics, Altona Diagnostics, Siemens AG, Agilent Technologies Inc., Seasun Biomaterials, BTNX Inc., Rapiim, Canon Medical Systems Corporation, Cellspect Co. Ltd., iHealth Labs Inc., InBios International Inc., iXensor Co. Ltd., Jiangsu Medomics medical technology Co Ltd., Maxim Biomedical Inc., Mologic Inc., OraSure Technologies Inc., OSANG LLC, PHASE Scientific International Ltd., Watmind USA, Xiamen Boson Biotech Co. Ltd.Global COVID-19 Detection Test Kits and Consumables Market Trends and Insights
Major companies operating in the COVID-19 Detection Test Kits and Consumables market are increasingly focusing on at-home molecular diagnostic platforms, such as Molecular tests, to enhance testing accuracy, improve user convenience, and support timely detection of infections. Molecular tests are diagnostic tests that detect specific genetic material, such as DNA or RNA, of microorganisms, human cells, or other biological markers. For instance, in April 2023, Lucira Health, a US-based medical technology company, launched the Lucira COVID-19 & Flu Home Test, which gained FDA Emergency Use Authorization (EUA) as the first OTC, at-home combination molecular test for SARS-CoV-2 and influenza A/B. This single-use kit uses a shallow nasal swab that is swirled in a vial, which is then inserted into a battery-powered test unit; in about 30 minutes, it delivers separate readouts for COVID-19, Flu A, and Flu B, with performance comparable to lab-based PCR in clinical trials. Key benefits include the speed, the ability to differentiate between COVID-19 and flu (which have overlapping symptoms but require different treatment), and the convenience of use at home with one simple device.What Are Latest Mergers And Acquisitions In The COVID-19 Detection Test Kits and Consumables Market?
In December 2023, Danaher Corporation, a US-based provider of life-sciences instruments, diagnostics and advanced scientific tools, acquired Abcam plc for an undisclosed amount. With this acquisition, Danaher aims to integrate Abcam’s offerings into its life-sciences and diagnostics platform to deepen its consumables portfolio and better support research and therapeutic discovery. Abcam plc is a UK-based company that specializes in manufacturing COVID-19 detection test kits and related consumables, including multiple SARS CoV 2 antibody ELISA kits.Regional Outlook
North America was the largest region in the COVID-19 detection test kits and consumables market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the COVID-19 Detection Test Kits and Consumables Market?
The COVID-19 detection test kits & consumables market consists of sales of nucleic acid amplification tests (NAATs) and antigen tests. Values in this market are factory gate values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.COVID-19 Detection Test Kits and Consumables Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $8.94 billion |
| Revenue Forecast In 2035 | $13.23 billion |
| Growth Rate | CAGR of 7.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Kits, Consumables, Specimen Type, End Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cepheid, BGI Genomics, Abbott Laboratories, bioMérieux SA, Bio-Rad Laboratories, F. Hoffmann-La Roche, GenMark Diagnostics, Mylab Discovery Solutions, Qiagen, Quidel Corporation, Randox Laboratories, SD Biosensor, Seegene Inc., Shenzhen Bioeasy Biotechnology, Thermo Fisher Scientific inc., BioMednomics, Getein Biotech, Sensing Self Ltd., Hangzhou Biotest Biotech Co. Ltd, AmonMed Biotechnology Co., Beijing Tigsun Diagnostics Co Ltd., BioMaxima S.A., CTK Biotech, Hunan Lituo Biotechnology Co., Vivacheck Lab, MD Solutions, FastSense Diagnostics, Altona Diagnostics, Siemens AG, Agilent Technologies Inc., Seasun Biomaterials, BTNX Inc., Rapiim, Canon Medical Systems Corporation, Cellspect Co. Ltd., iHealth Labs Inc., InBios International Inc., iXensor Co. Ltd., Jiangsu Medomics medical technology Co Ltd., Maxim Biomedical Inc., Mologic Inc., OraSure Technologies Inc., OSANG LLC, PHASE Scientific International Ltd., Watmind USA, Xiamen Boson Biotech Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
