
Critical Limb Ischemia Market Report 2026
Global Outlook – By Type (Devices, Drugs, Surgery), By Medication (Antihypertensive Agents, Antiplatelet Drugs, Antithrombotic Agents, Other Medications), By Application (Hospital, Clinic, Other Applications) - Market Size, Trends, And Global Forecast 2026-2035
Critical Limb Ischemia Market Overview
• Critical Limb Ischemia market size has reached to $3.89 billion in 2025 • Expected to grow to $6 billion in 2030 at a compound annual growth rate (CAGR) of 9% • Growth Driver: Increasing Investments In Healthcare Infrastructure Propel Growth Of Critical Limb Ischemia Treatment Market • Market Trend: FDA Approval For Dissolving Stent Esprit BTK • North America was the largest region in 2025.What Is Covered Under Critical Limb Ischemia Market?
Critical limb ischemia treatment refers to re-establishing blood flow to the area affected with critical limb ischemia, a chronic condition where blood flow to one or two of the hands, and legs are severely blocked causing numbness and pain. Treatment includes diagnosis and treating patients by medication, surgery using devices, and others. The main critical limb ischemia types are devices, drugs, and surgery. Embolic protection devices (EPDs) refer to a medical device that traps blood and luminal debris and prevents plaque debris from reaching the distal bed. The various medications are antihypertensive agents, antiplatelet drugs, antithrombotic agents, and others that are used in hospital, clinic, and others.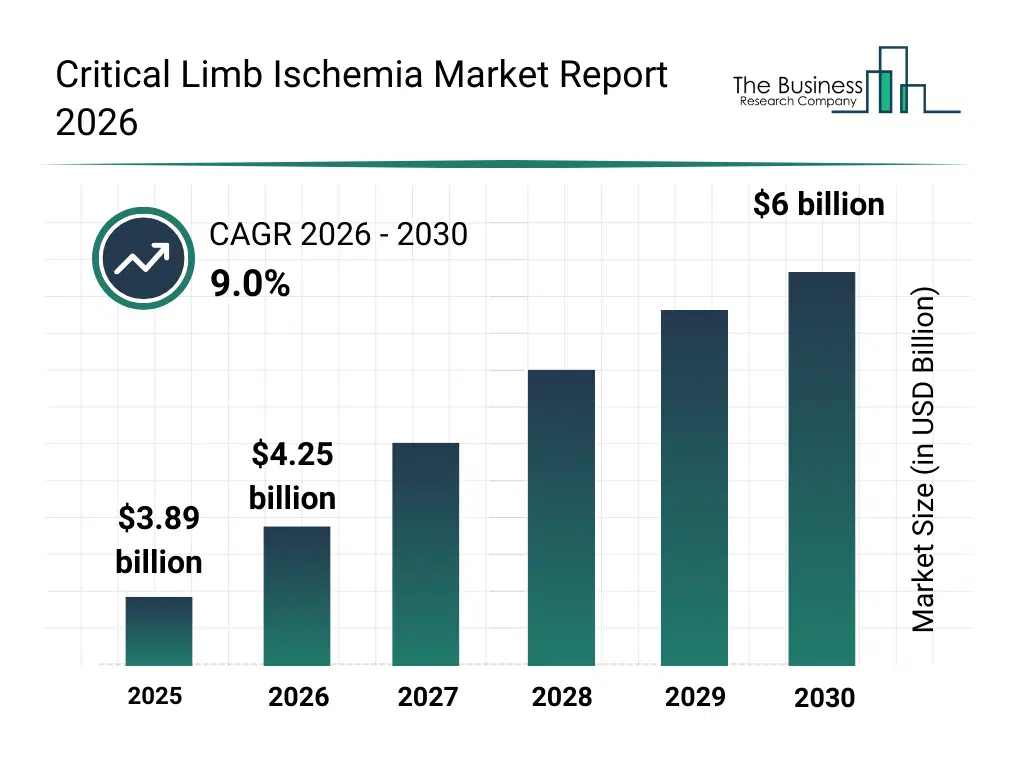
What Is The Critical Limb Ischemia Market Size and Share 2026?
The critical limb ischemia market size has grown strongly in recent years. It will grow from $3.89 billion in 2025 to $4.25 billion in 2026 at a compound annual growth rate (CAGR) of 9.3%. The growth in the historic period can be attributed to high prevalence of peripheral artery disease, increasing diabetes incidence, aging population growth, reliance on surgical revascularization, delayed disease diagnosis.What Is The Critical Limb Ischemia Market Growth Forecast?
The critical limb ischemia market size is expected to see strong growth in the next few years. It will grow to $6 billion in 2030 at a compound annual growth rate (CAGR) of 9.0%. The growth in the forecast period can be attributed to advancements in vascular devices, rising minimally invasive procedures, expansion of specialized vascular centers, growing awareness of limb preservation, improved diagnostic imaging access. Major trends in the forecast period include rising adoption of endovascular revascularization, increased use of drug-eluting devices, growth of limb salvage treatment approaches, expansion of multidisciplinary vascular care, focus on early diagnosis and intervention.Global Critical Limb Ischemia Market Segmentation
1) By Type: Devices, Drugs, Surgery 2) By Medication: Antihypertensive Agents, Antiplatelet Drugs, Antithrombotic Agents, Other Medications 3) By Application: Hospital, Clinic, Other Applications Subsegments: 1) By Devices: Angioplasty Balloons, Stents, Catheters, Atherectomy Devices, Implantable Devices 2) By Drugs: Antiplatelet Agents, Vasodilators, Anticoagulants, Thrombolytics, Growth Factors And Cytokines 3) By Surgery: Bypass Surgery, Endarterectomy, Amputation, Revascularization ProceduresWhat Is The Driver Of The Critical Limb Ischemia Market?
The increasing investments in healthcare infrastructure are expected to propel the growth of the critical limb ischemia treatment market going forward. Healthcare infrastructure investment refers to the allocation of funds towards the development, improvement, and maintenance of essential physical structures, facilities, and systems that support the delivery of healthcare services. Healthcare infrastructure investment plays a significant role in critical limb ischemia (CLI) treatment by providing the necessary facilities, equipment, and resources to diagnose and treat the condition effectively. For instance, in May 2023, according to a report published by the Office for National Statistics, a UK-based government department, between 2022 and 2023, healthcare spending in the UK increased by 5.6% compared with growth of 0.9% in 2022. The UK healthcare expenditure in UK healthcare expenditure was around $317.63 billion (£292 billion) in 2023. Therefore, the increasing investments in healthcare infrastructure are driving the growth of the critical limb ischemia treatment market.Key Players In The Global Critical Limb Ischemia Market
Major companies operating in the critical limb ischemia market are Medtronic Plc, Boston Scientific Corporation, Abbott Laboratories, LimFlow SA, Micro Medical Solutions, Cardiovascular Systems Inc., Shanghai MicroPort Medical (Group) Co Ltd., B. Braun SE, Teva Pharmaceutical Industries Ltd., Cook Medical, Philips, BD, Penumbra Inc., Cardinal Health Inc., Teleflex Incorporated, AngioDynamics Inc., Endologix Inc., Terumo Corporation, MicroPort Scientific, Silk Road Medical, LeMaitre Vascular Inc., Merit Medical Systems Inc., Shockwave Medical, Avinger, Biomerics LLC, Intact Vascular Inc., Bluegrass Vascular Technologies, PQ Bypass, Rex Medical, Aortica CorporationGlobal Critical Limb Ischemia Market Trends and Insights
Major companies operating in the critical limb ischemia (CLI) treatment market are innovating technologically advanced products, such as dissolving stents, to improve treatment outcomes for patients suffering from this severe form of peripheral artery disease. Dissolving stents, also known as bioresorbable stents, are designed to provide temporary support to the affected artery and then gradually dissolve after the vessel has healed. For instance, in April 2024, Abbott Laboratories, a US-based medical device company, received approval from the Food and Drug Administration (FDA), a US-based federal agency, for the Esprit BTK Everolimus Eluting Resorbable Scaffold System. It is a dissolving stent designed for patients suffering from chronic limb-threatening ischemia (CLTI) in arteries below the knee. This marks the first approval of a dissolving stent specifically for this condition in the United States. The stent is made from materials similar to dissolving sutures and is designed to gradually dissolve over approximately three years after implantation. It releases the immunosuppressive drug everolimus, which helps prevent the re-narrowing of arteries and supports vessel healing.What Are Latest Mergers And Acquisitions In The Critical Limb Ischemia Market?
In November 2023, Inari Medical, a US-based medical device company, acquired LimFlow SA for an undisclosed amount. With this acquisition, Inari Medical aims to broaden its portfolio with innovative technology that meets unmet needs in the chronic limb-threatening ischemia (CLTI), thereby improving patient outcomes and fostering growth. LimFlow SA is a Switzerland-based company that provides medical devices for the treatment of chronic limb-threatening ischemia (CLTI).Regional Outlook
North America was the largest region in the critical limb ischemia treatment market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Critical Limb Ischemia Market?
The critical limb ischemia treatment market includes revenues earned by entities by providing angioplasty, stents and laser atherectomy treatment. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Critical Limb Ischemia Market Report 2026?
The critical limb ischemia market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the critical limb ischemia industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Critical Limb Ischemia Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.25 billion |
| Revenue Forecast In 2035 | $6 billion |
| Growth Rate | CAGR of 9.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Medication, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic Plc, Boston Scientific Corporation, Abbott Laboratories, LimFlow SA, Micro Medical Solutions, Cardiovascular Systems Inc., Shanghai MicroPort Medical (Group) Co Ltd., B. Braun SE, Teva Pharmaceutical Industries Ltd., Cook Medical, Philips, BD, Penumbra Inc., Cardinal Health Inc., Teleflex Incorporated, AngioDynamics Inc., Endologix Inc., Terumo Corporation, MicroPort Scientific, Silk Road Medical, LeMaitre Vascular Inc., Merit Medical Systems Inc., Shockwave Medical, Avinger, Biomerics LLC, Intact Vascular Inc., Bluegrass Vascular Technologies, PQ Bypass, Rex Medical, Aortica Corporation |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
