
Cytokine Release Syndrome Management Market Report 2026
Global Outlook – By Treatment Type (Monoclonal Antibodies, Corticosteroids, Intravenous Immunoglobulin (IVIG), Targeted Therapy, Supportive Care, Other Treatment Types), By Indication (Chimeric Antigen Receptor (CAR) T-Cell Therapy, Immune Checkpoint Inhibitors, Hematopoietic Stem Cell Transplantation (HSCT), Autoimmune Diseases, Infections, Other Indications), By Route Of Administration (Intravenous (IV), Subcutaneous (SC), Oral, Intramuscular (IM), Intrathecal, Other Routes Of Administration), By End Users (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Clinics, Research Institutions, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Cytokine Release Syndrome Management Market Overview
• Cytokine Release Syndrome Management market size has reached to $23.76 billion in 2025 • Expected to grow to $32.35 billion in 2030 at a compound annual growth rate (CAGR) of 6.3% • Growth Driver: The Increasing Use Of Targeted Therapies Is Fueling The Market Growth Due To Enhanced Treatment Precision And Reduce Side Effects • Market Trend: Future Prospects Of Host-Directed Small Molecule Drugs In The Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Cytokine Release Syndrome Management Market?
Cytokine release syndrome (CRS) management involves medical strategies to control the excessive immune response caused by an overproduction of cytokines, often seen in conditions such as CAR-T cell therapy or certain infections. It includes the use of medications such as corticosteroids and cytokine inhibitors, supportive care, and sometimes intensive monitoring to prevent severe complications. This management is crucial in reducing the risk of severe organ damage and improving patient outcomes. The main treatment types for cytokine release syndrome management include monoclonal antibodies, corticosteroids, intravenous immunoglobulin (IVIG), targeted therapy, supportive care, and other treatment options. Monoclonal antibodies are lab-engineered molecules that target specific cytokines or their receptors to regulate immune activity and manage CRS. These antibodies are used for various indications, including chimeric antigen receptor (CAR) T-cell therapy, immune checkpoint inhibitors, hematopoietic stem cell transplantation (HSCT), autoimmune diseases, infections, and others, and are administered intravenously (IV), subcutaneously (SC), orally, intramuscularly (IM), intrathecally, and by others. Treatment options are utilized by various end users, including hospital pharmacies, retail pharmacies, online pharmacies, specialty clinics, research institutions, and others.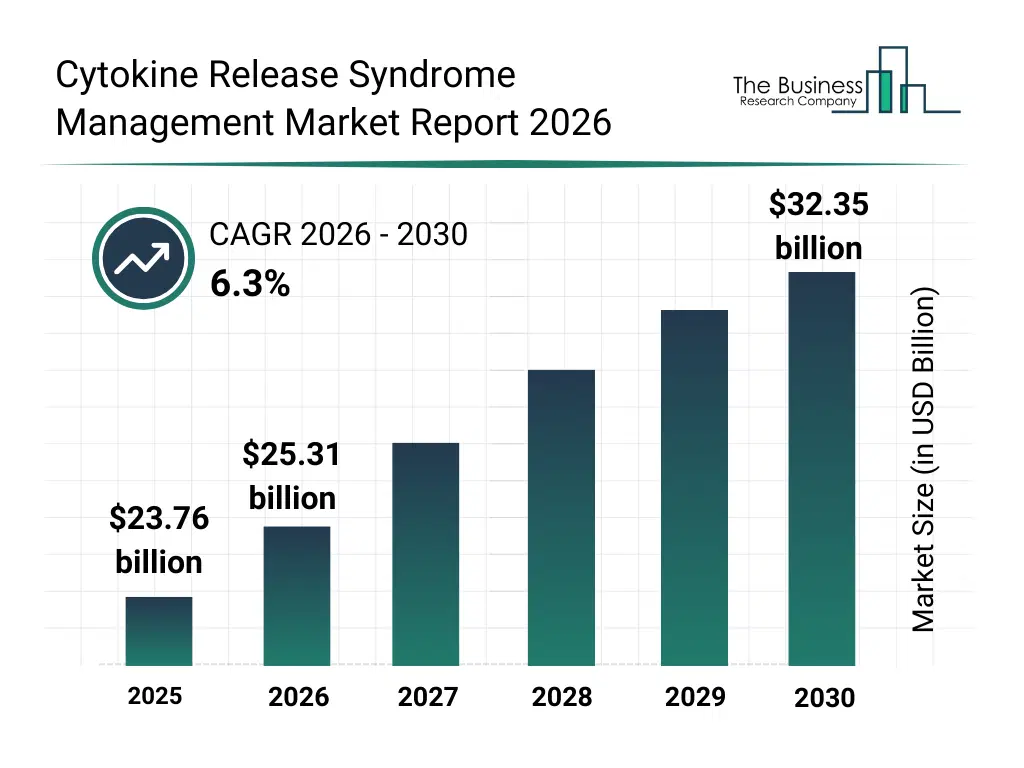
What Is The Cytokine Release Syndrome Management Market Size and Share 2026?
The cytokine release syndrome management market size has grown strongly in recent years. It will grow from $23.76 billion in 2025 to $25.31 billion in 2026 at a compound annual growth rate (CAGR) of 6.5%. The growth in the historic period can be attributed to growth of car-t therapies, limited crs treatment options, reliance on corticosteroids, hospital-based management, high mortality risk.What Is The Cytokine Release Syndrome Management Market Growth Forecast?
The cytokine release syndrome management market size is expected to see strong growth in the next few years. It will grow to $32.35 billion in 2030 at a compound annual growth rate (CAGR) of 6.3%. The growth in the forecast period can be attributed to expansion of cell therapies, advancements in immunomodulators, improved early detection, standardized treatment guidelines, increasing oncology adoption. Major trends in the forecast period include rising use of il-6 inhibitors, expansion of crs management in car-t therapy, increased focus on early intervention protocols, growth of intensive care monitoring, development of targeted immunomodulators.Global Cytokine Release Syndrome Management Market Segmentation
1) By Treatment Type: Monoclonal Antibodies, Corticosteroids, Intravenous Immunoglobulin (IVIG), Targeted Therapy, Supportive Care, Other Treatment Types 2) By Indication: Chimeric Antigen Receptor (CAR) T-Cell Therapy, Immune Checkpoint Inhibitors, Hematopoietic Stem Cell Transplantation (HSCT), Autoimmune Diseases, Infections, Other Indications 3) By Route Of Administration: Intravenous (IV), Subcutaneous (SC), Oral, Intramuscular (IM), Intrathecal, Other Routes Of Administration 4) By End Users: Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Clinics, Research Institutions, Other End Users Subsegments: 1) By Monoclonal Antibodies: Tocilizumab, Siltuximab, Infliximab, Rituximab 2) By Corticosteroids: Dexamethasone, Prednisone, Methylprednisolone, Hydrocortisone 3) By Intravenous Immunoglobulin (IVIG): Immunoglobulin G (IgG)-Based Intravenous Immunoglobulin, Immunoglobulin A (IgA)-Based Intravenous Immunoglobulin, Immunoglobulin M (IgM)-Based Intravenous Immunoglobulin, Polyvalent Intravenous Immunoglobulin 4) By Targeted Therapy: Janus Kinase (JAK) Inhibitors, Interleukin-6 (IL-6) Inhibitors, Tumor Necrosis Factor-Alpha (TNF-α) Inhibitors, Bruton's Tyrosine Kinase (BTK) Inhibitors 5) By Supportive Care: Antipyretic Medications, Analgesic Medications, Intravenous Fluids And Electrolyte Solutions, Oxygen Therapy, Vasopressor Agents 6) By Other Treatment Types: Plasmapheresis (Therapeutic Plasma Exchange), Antihistamine Medications, Leukapheresis, Experimental Therapies, Nutritional SupportWhat Is The Driver Of The Cytokine Release Syndrome Management Market?
The increasing use of targeted therapies is expected to propel the growth of the cytokine release syndrome management market going forward. Targeted therapies are drugs or treatments designed to specifically block the growth and spread of cancer by interfering with molecular targets involved in tumor development. The increasing use of targeted therapies is primarily due to their ability to precisely target cancer cells while minimizing damage to healthy cells, improving treatment efficacy, and reducing side effects compared to traditional therapies. Cytokine release syndrome management is essential in optimizing targeted therapies by controlling intense inflammatory reactions, which enhances the safety and effectiveness of treatments such as CAR-T cell therapy and immune checkpoint inhibitors. For instance, in January 2024, according to the American Society of Gene and Cell Therapy, a US-based organization for gene and cell therapy, in the fourth quarter of 2023, the number of gene therapies in Phase III clinical trials increased by 10%, marking the first quarterly rise of this kind since the third quarter of 2022. Therefore, the increasing use of targeted therapies is driving the growth of the cytokine release syndrome management industry.Key Players In The Global Cytokine Release Syndrome Management Market
Major companies operating in the cytokine release syndrome management market are Pfizer Inc., Johnson & Johnson Private Limited, F. Hoffmann-La Roche Ltd., AbbVie Inc., Sanofi S.A., Bristol Myers Squibb Company, Novartis AG, GlaxoSmithKline plc, Amgen Inc., Gilead Sciences Inc., Regeneron Pharmaceuticals Inc., Genentech Inc., Incyte Corporation, Swedish Orphan Biovitrum AB, Biocon Limited, MacroGenics Inc., Legend Biotech Corporation, Bluebird Bio Inc., InflaRx N.V., Humanigen Inc.Global Cytokine Release Syndrome Management Market Trends and Insights
Major companies operating in the cytokine release syndrome management market are focusing on conducting clinical trials to develop innovative therapies, such as host-directed small molecule drugs, aimed at modulating the immune response and reducing the severity of cytokine storms without compromising the body’s ability to fight infections or cancer. A host-directed small molecule drug is a low molecular weight compound that targets the host’s immune signaling pathways to suppress excessive cytokine production, thereby helping manage cytokine release syndrome (CRS) by preventing immune system overactivation. For instance, in April 2025, CytoAgents Inc., a US-based biotechnology company, announced the successful completion of the first dose-escalation cohort in its Phase 1b/2a clinical trial of CTO1681, a host-directed small molecule drug developed to prevent and treat CRS in patients receiving CAR T-cell therapy. The investigational therapy demonstrated a favorable safety profile, with no dose-limiting toxicities or disruption to CAR T-cell efficacy. The trial, utilizing a rolling six dose-escalation design, is being conducted to assess the safety, tolerability, and preliminary efficacy of CTO1681, as well as to establish the recommended phase 2 dose.What Are Latest Mergers And Acquisitions In The Cytokine Release Syndrome Management Market?
In March 2024, Merck & Co., Inc., a US-based biopharmaceutical company, acquired Harpoon Therapeutics for $680 million. With this acquisition, Merck aims to strengthen its oncology pipeline by integrating Harpoon’s T-cell engager technology and lead candidate, MK-6070, currently in a Phase 1/2 trial (NCT04471727) for advanced cancers, enhancing its position in immuno-oncology and expanding its next-generation cancer therapies. Harpoon Therapeutics Inc. is a US-based clinical-stage biopharmaceutical company specializing in T-cell engagers for cancer treatment, focused on its TriTAC-XR platform to reduce cytokine release syndrome and improve efficacy.Regional Outlook
North America was the largest region in the cytokine release syndrome management market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Cytokine Release Syndrome Management Market?
The cytokine release syndrome management market consists of revenues earned by entities by providing services such as diagnostic testing, immunosuppressive therapy, supportive care, clinical trial services, and post-treatment monitoring to manage and treat patients affected by CRS. The market value includes the value of related goods sold by the service provider or included within the service offering. The cytokine release syndrome management market also includes sales of tocilizumab (actemra), siltuximab (sylvant), dexamethasone, ruxolitinib (jakafi), and ivig (intravenous immunoglobulin). Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Cytokine Release Syndrome Management Market Report 2026?
The cytokine release syndrome management market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the cytokine release syndrome management industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Cytokine Release Syndrome Management Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $25.31 billion |
| Revenue Forecast In 2035 | $32.35 billion |
| Growth Rate | CAGR of 6.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Type, Indication, Route Of Administration, End Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Johnson & Johnson Private Limited, F. Hoffmann-La Roche Ltd., AbbVie Inc., Sanofi S.A., Bristol Myers Squibb Company, Novartis AG, GlaxoSmithKline plc, Amgen Inc., Gilead Sciences Inc., Regeneron Pharmaceuticals Inc., Genentech Inc., Incyte Corporation, Swedish Orphan Biovitrum AB, Biocon Limited, MacroGenics Inc., Legend Biotech Corporation, Bluebird Bio Inc., InflaRx N.V., Humanigen Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
