
Decentralized Clinical Trials Market Report 2026
Global Outlook – By Type (Interventional, Observational, Expanded Access), By Technology Used (Wearable Devices, Mobile Health Applications, Telemedicine Platforms, Electronic Data Capture Systems), By Study Phase (Phase I, Phase II, Phase III, Phase IV), By Therapeutic Area (Oncology, Cardiology, Neurology, Infectious Diseases), By End-User (Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations, Regulatory Authorities, Academic And Research Institutions) - Market Size, Trends, And Global Forecast 2026-2035
Decentralized Clinical Trials Market Overview
• Decentralized Clinical Trials market size has reached to $8.77 billion in 2025 • Expected to grow to $19.55 billion in 2030 at a compound annual growth rate (CAGR) of 17.3% • Growth Driver: The Rising Focus On Telemedicine Is Fueling The Growth Of The Market Due To Its Role In Enabling Remote Patient Monitoring • Market Trend: Development In All-In-One No-Code Clinical Trials Software Transforming Trial Efficiency And Patient Engagement • North America was the largest region in 2025 and Asia Pacific is the fastest growing region.What Is Covered Under Decentralized Clinical Trials Market?
Decentralized clinical trials are a modern approach to conducting clinical research that uses digital tools and remote processes to engage participants beyond traditional trial sites. They streamline data collection and monitoring in real time, improving trial efficiency. Decentralized clinical trials enhance accessibility, allowing broader and more diverse participation, and also accelerate research timelines while reducing operational challenges. The main types of decentralized clinical trials are interventional, observational, and expanded access. Interventional decentralized clinical trials involve assigning participants to receive specific interventions to evaluate their effects on health outcomes. The technologies used include wearable devices, mobile health applications, telemedicine platforms, and electronic data capture systems, with various study phases, such as phase I, phase II, phase III, and phase IV. It is applied in several therapeutic areas, including oncology, cardiology, neurology, and infectious diseases, and used by various end-users, including pharmaceutical companies, biotechnology firms, contract research organizations, regulatory authorities, and academic and research institutions.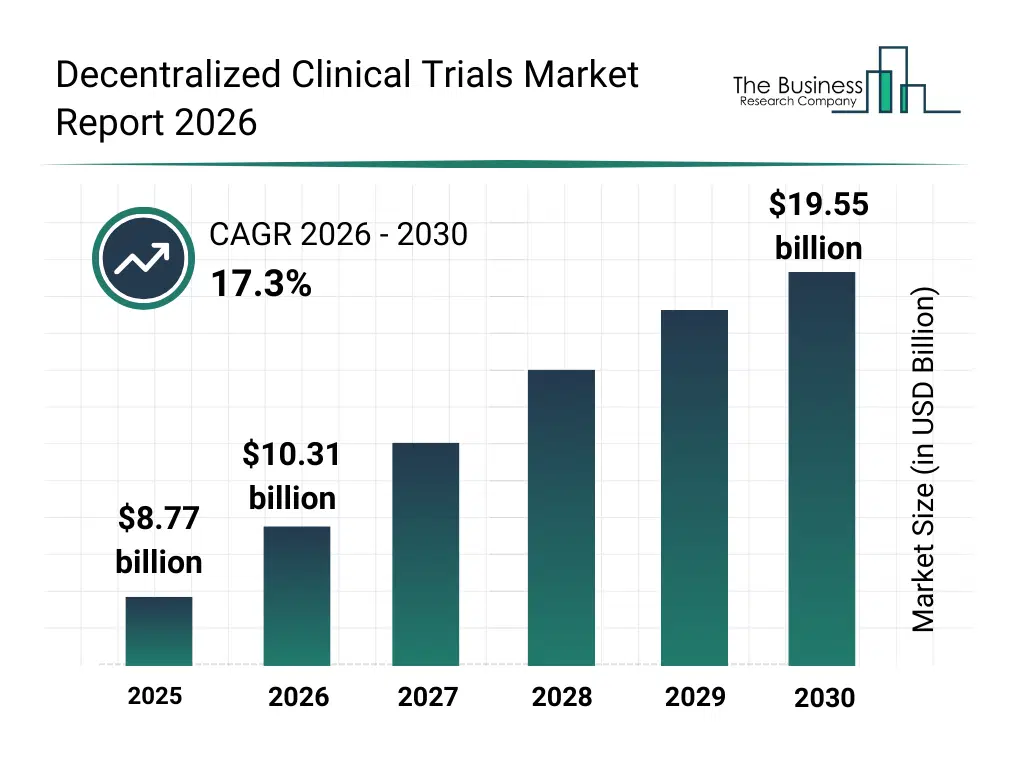
What Is The Decentralized Clinical Trials Market Size and Share 2026?
The decentralized clinical trials market size has grown rapidly in recent years. It will grow from $8.77 billion in 2025 to $10.31 billion in 2026 at a compound annual growth rate (CAGR) of 17.7%. The growth in the historic period can be attributed to increasing complexity of clinical trials, rising need for patient recruitment efficiency, expansion of digital health technologies, growing use of electronic data capture systems, increasing regulatory openness to decentralized models.What Is The Decentralized Clinical Trials Market Growth Forecast?
The decentralized clinical trials market size is expected to see rapid growth in the next few years. It will grow to $19.55 billion in 2030 at a compound annual growth rate (CAGR) of 17.3%. The growth in the forecast period can be attributed to increasing adoption of hybrid trial designs, rising demand for faster drug development timelines, expansion of global virtual trials, growing use of ai-driven trial optimization, increasing focus on diverse patient participation. Major trends in the forecast period include increasing adoption of remote patient monitoring tools, growing use of virtual trial platforms, expansion of wearable-based data collection, rising implementation of real-time trial analytics, enhanced focus on patient-centric trial design.Global Decentralized Clinical Trials Market Segmentation
1) By Type: Interventional, Observational, Expanded Access 2) By Technology Used: Wearable Devices, Mobile Health Applications, Telemedicine Platforms, Electronic Data Capture Systems 3) By Study Phase: Phase I, Phase II, Phase III, Phase IV 4) By Therapeutic Area: Oncology, Cardiology, Neurology, Infectious Diseases 5) By End-User: Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations, Regulatory Authorities, Academic And Research Institutions Subsegments: 1) By Interventional: Randomized Controlled Trials (RCTs), Non-Randomized Trials, Adaptive Clinical Trials, Pragmatic Clinical Trials 2) By Observational: Cohort Studies, Case-Control Studies, Cross-Sectional Studies, Prospective Studies, Retrospective Studies, Longitudinal Studies 3) By Expanded Access: Treatment Investigational New Drug (IND) Applications, Intermediate-Size Patient Population Investigational New Drug (IND) Applications, Single-Patient Investigational New Drug (IND) Applications, Emergency Use Investigational New Drug (IND) ApplicationsWhat Is The Driver Of The Decentralized Clinical Trials Market?
The rising focus on telemedicine is expected to propel growth in the decentralized clinical trials market going forward. Telemedicine is the delivery of healthcare services remotely through digital communication technologies, enabling medical consultations and monitoring without requiring physical presence at healthcare facilities. The growing focus on telemedicine is primarily driven by the need for accessible healthcare delivery in response to global health challenges and social distancing requirements. Decentralized clinical trials use telemedicine to remotely engage and monitor patients, minimizing in-person visits while enabling real-time data collection, greater access, and continuous oversight. For instance, in September 2024, according to the Australian Bureau of Statistics (ABS), the national statistical agency of Australia, in 2023–24, 83.5% of males reported using telehealth services, an increase from 78.6% in 2022–23. Therefore, rising focus on telemedicine is driving the growth of the decentralized clinical trials industry.Key Players In The Global Decentralized Clinical Trials Market
Major companies operating in the decentralized clinical trials market are Walgreens Boots Alliance Inc., Pfizer Inc., Oracle Corporation, Thermo Fisher Scientific Inc., IQVIA Holdings Inc., Labcorp Holdings Inc., ICON Public Limited Company, Dassault Systèmes SE, LEO Pharma A/S, Verily Life Sciences LLC, ProPharma, Signant Health Holding Corp., PCM Trials, Huma Therapeutics Limited, Medable Inc., Science 37 Inc., Obvio Health, EmVenio Research Inc., Grove AI Inc., Phesi Inc.Global Decentralized Clinical Trials Market Trends and Insights
Major companies operating in the decentralized clinical trials market are focusing on developing advanced solutions such as all-in-one, no-code clinical trials software to simplify trial setup and management and enhance overall efficiency and patient engagement. An all-in-one, no-code clinical trials software empowers researchers to design, manage, and run trials without coding while seamlessly integrating patient recruitment, data capture, monitoring, and analytics into a single user-friendly platform. For instance, in August 2024, Medable, a US-based clinical technology company, launched Medable Studio, an all-in-one, no-code software solution designed to simplify and accelerate the configuration, translation, validation, and launch of electronic Clinical Outcome Assessments (eCOA), eConsent, Televisit, and sensors Plus into clinical trials. It enables biopharmaceutical companies to create protocol-fit study designs in hours instead of months, giving users greater control, transparency, and speed, while offering flexible self-service, full-service, or hybrid options that significantly reduce study startup timelines and costs. This platform aims to revolutionize trial execution by making the study build process faster, more efficient, and less dependent on professional services.What Are Latest Mergers And Acquisitions In The Decentralized Clinical Trials Market?
In July 2023, Signant Health, a US-based provider of evidence generation for modern clinical trials, acquired DSG Inc. for an undisclosed amount. Through this acquisition, Signant Health aims to enhance its eClinical solution suite by integrating DSG’s unified EDC and DDC platform, enabling more flexible and comprehensive support for both traditional and decentralized clinical trials. This deal strengthens operational efficiency, simplifies trial processes, and improves digital solutions for sponsors, sites, and CROs. DSG Inc. is a US-based provider of electronic data capture (EDC) and direct data capture (DDC) technology within a unified eClinical suite for site-based and decentralized clinical trials.Regional Outlook
North America was the largest region in the decentralized clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Decentralized Clinical Trials Market?
The decentralized clinical trials market consists of revenues earned by entities by providing services such as patient recruitment and retention, site management and monitoring, data management and analytics, regulatory compliance support, and telemedicine or virtual care solutions. The market value includes the value of related goods sold by the service provider or included within the service offering. The decentralized clinical trials market also includes sales of wearable health monitoring devices, electronic data capture systems, biosensors, remote patient monitoring equipment, and clinical trial management software. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities, wholesalers, distributors, and retailers or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Decentralized Clinical Trials Market Report 2026?
The decentralized clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the decentralized clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Decentralized Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $10.31 billion |
| Revenue Forecast In 2035 | $19.55 billion |
| Growth Rate | CAGR of 17.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Technology Used, Study Phase, Therapeutic Area, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Walgreens Boots Alliance Inc., Pfizer Inc., Oracle Corporation, Thermo Fisher Scientific Inc., IQVIA Holdings Inc., Labcorp Holdings Inc., ICON Public Limited Company, Dassault Systèmes SE, LEO Pharma A/S, Verily Life Sciences LLC, ProPharma, Signant Health Holding Corp., PCM Trials, Huma Therapeutics Limited, Medable Inc., Science 37 Inc., Obvio Health, EmVenio Research Inc., Grove AI Inc., Phesi Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
