Difficile-Associated Diarrhea Treatment Market Report 2026

Difficile-Associated Diarrhea Treatment Market Report 2026
Global Outlook – By Type (Narrow Spectrum Antibiotics, Broad Spectrum Antibiotics), By Distribution Channel (Hospital Pharmacy, Online Pharmacy, Retail Pharmacy), By End User (Hospitals, Homecare, Specialty Clinics, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Difficile-Associated Diarrhea Treatment Market Overview
• Difficile-Associated Diarrhea Treatment market size has reached to $3.69 billion in 2025 • Expected to grow to $6.57 billion in 2030 at a compound annual growth rate (CAGR) of 12.3% • Growth Driver: Elevated Prevalence Of Digestive Disorders Drives Growth In The Clostridium Difficile-Associated Diarrhea Treatment Market • Market Trend: Innovative Drug Approvals Transforming Clostridium Difficile-Associated Diarrheal Treatment Market • North America was the largest region in 2025.What Is Covered Under Difficile-Associated Diarrhea Treatment Market?
Difficile-associated diarrhea is a bacterium that causes an infection of the large intestine (colon) and often infects people who have recently taken antibiotics. Difficile-associated diarrhea treatment is used to control the overgrowth of C. difficile bacteria (Clostridium) in the colon and reduce the production of toxins. Common symptoms include watery diarrhea, loss of appetite, nausea, abdominal pain and more. The main types of difficile-associated diarrhea treatments are narrow-spectrum antibiotics and broad-spectrum antibiotics. Narrow-spectrum antibiotics refer to a class of antimicrobial agents that target a specific group of bacteria or subgroups of bacteria. These are sold through several distribution channels, including hospital pharmacies, online pharmacies and retail pharmacies, to various end users, such as hospitals, homecare, specialty clinics and others.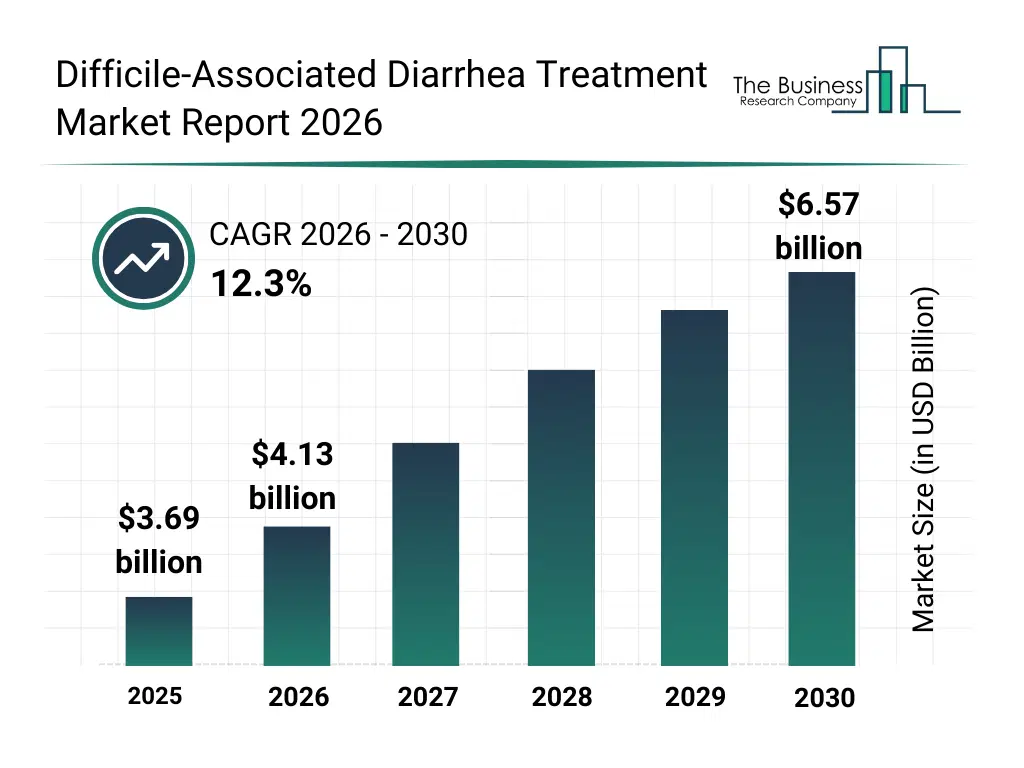
What Is The Difficile-Associated Diarrhea Treatment Market Size and Share 2026?
The difficile-associated diarrhea treatment market size has grown rapidly in recent years. It will grow from $3.69 billion in 2025 to $4.13 billion in 2026 at a compound annual growth rate (CAGR) of 11.6%. The growth in the historic period can be attributed to high antibiotic usage rates, hospital-acquired infection prevalence, limited treatment options, aging population vulnerability, prolonged hospital stays.What Is The Difficile-Associated Diarrhea Treatment Market Growth Forecast?
The difficile-associated diarrhea treatment market size is expected to see rapid growth in the next few years. It will grow to $6.57 billion in 2030 at a compound annual growth rate (CAGR) of 12.3%. The growth in the forecast period can be attributed to development of novel antibiotics, infection control initiatives, recurrence reduction strategies, improved diagnostics adoption, antimicrobial stewardship programs. Major trends in the forecast period include increasing use of narrow-spectrum antibiotics, growing focus on recurrence prevention, expansion of hospital-based treatment protocols, rising awareness of antibiotic stewardship, development of targeted cdi therapies.Global Difficile-Associated Diarrhea Treatment Market Segmentation
1) By Type: Narrow Spectrum Antibiotics, Broad Spectrum Antibiotics 2) By Distribution Channel: Hospital Pharmacy, Online Pharmacy, Retail Pharmacy 3) By End User: Hospitals, Homecare, Specialty Clinics, Other End-Users Subsegments: 1) By Narrow Spectrum Antibiotics: Vancomycin, Fidaxomicin, Metronidazole 2) By Broad Spectrum Antibiotics: Ampicillin, Ciprofloxacin, ClindamycinWhat Are The Drivers Of The Difficile-Associated Diarrhea Treatment Market?
The increasing prevalence of digestive disorders is expected to propel the growth of the difficile-associated diarrhea treatment market going forward. Digestive disorders refer to a group of medical conditions that affect the normal functioning of the digestive system, which is responsible for breaking down food and absorbing nutrients. Clostridium difficile (C. diff) infection is associated with digestive disorders, specifically diarrhea and colitis. Clostridium difficile is a bacterium that can infect the bowel by producing toxins that cause inflammation and damage the intestinal lining. With more people being diagnosed with digestive disorders, there is an increasing need for effective treatments to manage associated symptoms like difficile-associated diarrhea. For instance, in June 2023, according to Crohn's and Colitis Canada, a Canada-based non-profit organization, over 322,600 Canadians were estimated to live with inflammatory bowel diseases (IBD) in 2023, accounting for approximately 0.82% of the population. Further, it is estimated that around 470,000 Canadians will be living with IBD by 2035, about 1.1% of the population, or 1 in every 91 people in the country. Therefore, the increasing prevalence of digestive disorders is expected to propel the growth of the difficile-associated diarrhea treatment industry. The increasing prevalence of clostridium difficile infection (CDI) is expected to drive the growth of the difficile-associated diarrhea treatment market. Clostridium difficile infection (CDI), or C. difficile infection is caused by the bacterium clostridium difficile, which primarily affects the colon, produces toxins damaging the colon lining, and results in symptoms including diarrhea, abdominal pain, and potentially life-threatening complications. Clostridium difficile infection (CDI) is the primary cause of Difficile-Associated Diarrhea (DAD), and the successful treatment of CDI is crucial for managing and resolving DAD. For instance, in January 2023, according to the National Library of Medicine, a US-based medical library operated by the federal government, the Centers for Disease Control (CDC) estimates indicate that C. difficile infects about half a million Americans annually. Among these cases, roughly 29,000 patients face fatal outcomes within one month of diagnosis, with 15,000 of these deaths directly linked to C. difficile infection. Additionally, approximately 83,000 patients experience at least one recurrence of the disease, and 29,000 succumb to the initial diagnosis within 30 days. Therefore, the rise in minimally invasive procedures drives the growth of the difficile-associated diarrhea treatment industry.Key Players In The Global Difficile-Associated Diarrhea Treatment Market
Major companies operating in the difficile-associated diarrhea treatment market are Pfizer Inc., F. Hoffmann-La Roche AG, Merck & Co. Inc., AbbVie Inc., Sanofi S.A., Teva Pharmaceutical Industries Ltd., Mylan N.V., Sun Pharmaceutical Industries Ltd., Perrigo Company PLC, Dr. Reddy's Laboratories Ltd., Aurobindo Pharma Limited, Cipla Limited, Hikma Pharmaceuticals plc, Amneal Pharmaceuticals LLC, Zydus Lifesciences Ltd., Lupin Limited, Torrent Pharmaceuticals Ltd., Salix Pharmaceuticals Inc., Seres Therapeutics, Summit Therapeutics, Vedanta Biosciences, Finch Therapeutics GroupGlobal Difficile-Associated Diarrhea Treatment Market Trends and Insights
Major companies operating in the difficile-associated diarrhea treatment market are focused on developing innovative drugs and getting them approved to sustain their position in the market. Drug approvals for difficile-associated diarrhea treatment are increasing due to the need for more effective and safer treatments, such as SER-109, which provides a new therapeutic option for difficile-associated diarrhea treatment. For instance, in April 2023, the U.S. Food and Drug Administration, a US-based federal agency, approved SER-109, the first orally administered fecal microbiota product to prevent the recurrence of Clostridioides difficile diarrhea developed by Seres Therapeutics, a US-based biotech company. SER-109 is a live, spore-form probiotic administered orally in capsule form. It is composed of Firmicutes bacteria naturally found in the human gut. SER-109 works by restoring the balance of bacteria in the stomach, which can help prevent C. diff infections from recurring. This medication helps prevent the recurrence of Clostridioides difficile (C. diff) infection in individuals aged 18 and above following antibacterial treatment.Regional Outlook
North America was the largest region in the difficile-associated diarrhea treatment market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Difficile-Associated Diarrhea Treatment Market?
The difficile-associated diarrhea treatment market consists of revenues earned by entities by providing conservative therapy, laboratory tests, colonoscopy and imaging tests. The market value includes the value of related goods sold by the service provider or included within the service offering. The difficile-associated diarrhea treatment market also includes sales of oral rehydration solutions, electrolyte supplements and probiotics. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Difficile-Associated Diarrhea Treatment Market Report 2026?
The difficile-associated diarrhea treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the difficile-associated diarrhea treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Difficile-Associated Diarrhea Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.13 billion |
| Revenue Forecast In 2035 | $6.57 billion |
| Growth Rate | CAGR of 11.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Distribution Channel, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., F. Hoffmann-La Roche AG, Merck & Co. Inc., AbbVie Inc., Sanofi S.A., Teva Pharmaceutical Industries Ltd., Mylan N.V., Sun Pharmaceutical Industries Ltd., Perrigo Company PLC, Dr. Reddy's Laboratories Ltd., Aurobindo Pharma Limited, Cipla Limited, Hikma Pharmaceuticals plc, Amneal Pharmaceuticals LLC, Zydus Lifesciences Ltd., Lupin Limited, Torrent Pharmaceuticals Ltd., Salix Pharmaceuticals Inc., Seres Therapeutics, Summit Therapeutics, Vedanta Biosciences, Finch Therapeutics Group |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Difficile-Associated Diarrhea Treatment market was valued at $3.69 billion in 2025, increased to $4.13 billion in 2026, and is projected to reach $6.57 billion by 2030.
request a sample hereThe global Difficile-Associated Diarrhea Treatment market is expected to grow at a CAGR of 12.3% from 2026 to 2035 to reach $6.57 billion by 2035.
request a sample hereSome Key Players in the Difficile-Associated Diarrhea Treatment market Include, Pfizer Inc., F. Hoffmann-La Roche AG, Merck & Co. Inc., AbbVie Inc., Sanofi S.A., Teva Pharmaceutical Industries Ltd., Mylan N.V., Sun Pharmaceutical Industries Ltd., Perrigo Company PLC, Dr. Reddy's Laboratories Ltd., Aurobindo Pharma Limited, Cipla Limited, Hikma Pharmaceuticals plc, Amneal Pharmaceuticals LLC, Zydus Lifesciences Ltd., Lupin Limited, Torrent Pharmaceuticals Ltd., Salix Pharmaceuticals Inc., Seres Therapeutics, Summit Therapeutics, Vedanta Biosciences, Finch Therapeutics Group .
request a sample hereMajor trend in this market includes: Innovative Drug Approvals Transforming Clostridium Difficile-Associated Diarrheal Treatment Market. For further insights on this market.
request a sample hereNorth America was the largest region in the difficile-associated diarrhea treatment market in 2025. The regions covered in the difficile-associated diarrhea treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here